Advanced Benzoxazole Carbazole Diamine Monomers for High Performance Fluorescent Polyimide Manufacturing
Advanced Benzoxazole Carbazole Diamine Monomers for High Performance Fluorescent Polyimide Manufacturing
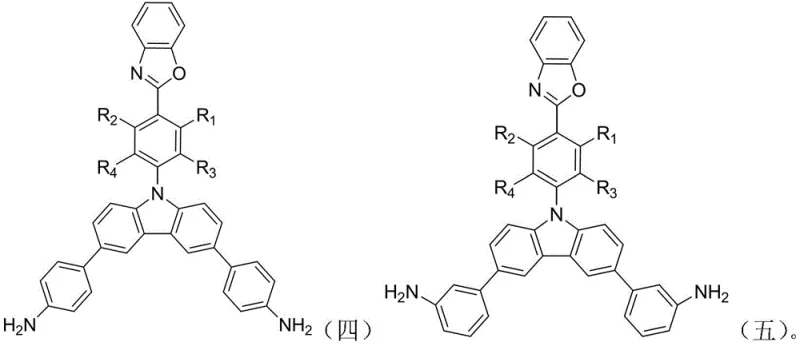
The rapid evolution of the optoelectronics industry demands materials that combine exceptional thermal stability with superior luminescent properties. Patent CN111138424B introduces a groundbreaking class of diamine monomers featuring integrated benzoxazole and carbazole structural motifs, specifically engineered to overcome the limitations of traditional polyimide precursors. This innovation addresses the critical challenge of charge transfer complex formation which often quenches fluorescence in conventional aromatic polyimides. By strategically incorporating these heterocyclic units, the resulting polymers exhibit not only high thermal decomposition temperatures exceeding 480°C but also distinct fluorescence characteristics with high luminous intensity. For R&D directors and procurement specialists in the electronic materials sector, this technology represents a significant leap forward in designing next-generation display and optical components.
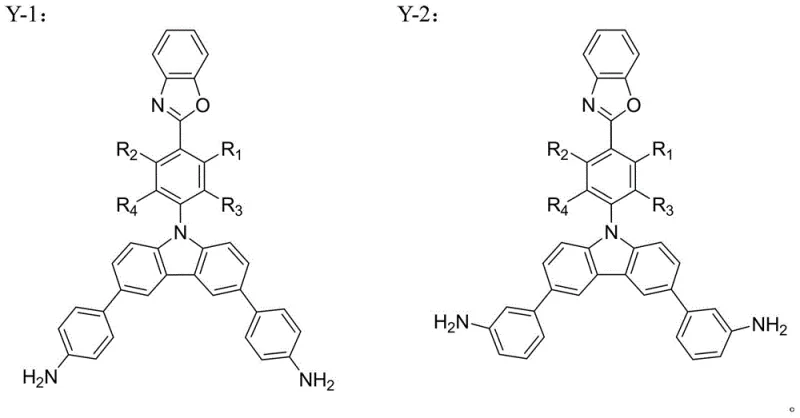
The structural versatility of these monomers allows for precise tuning of electronic properties, making them ideal candidates for reliable electronic chemical supplier portfolios focused on high-end display technologies. The synthesis pathway described in the patent utilizes well-established cross-coupling methodologies, ensuring that the transition from laboratory scale to commercial production is both feasible and economically viable. This report provides a deep technical analysis of the synthesis mechanism, compares it against conventional methods, and outlines the substantial commercial advantages for supply chain stakeholders seeking cost reduction in display materials manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional diamine monomers used in polyimide synthesis often rely on simple aromatic backbones which, while thermally stable, suffer from strong intermolecular interactions. These interactions facilitate the formation of charge transfer complexes (CTCs) between polymer chains, which acts as a quenching mechanism for fluorescence, thereby limiting their utility in light-emitting applications. Furthermore, existing methods to introduce fluorescent groups often result in poor solubility or compromised thermal stability due to the disruption of the rigid polymer backbone. The inability to effectively decouple the charge transfer effect from the main chain dynamics has long been a bottleneck in developing high-efficiency luminescent polyimides. Consequently, manufacturers have struggled to find monomers that offer a balanced profile of processability, thermal resistance, and optical performance without resorting to prohibitively expensive or unstable chemical architectures.
The Novel Approach
The novel approach detailed in the patent leverages the unique electronic properties of benzoxazole and carbazole units to disrupt these detrimental intermolecular interactions. By grafting a benzoxazole-containing moiety onto a carbazole core, the resulting diamine monomer introduces steric hindrance and electronic modulation that significantly reduces the density of charge transfer complexes. This structural design preserves the inherent thermal stability of the polyimide while unlocking strong fluorescence capabilities. The synthesis strategy employs a modular Suzuki coupling protocol, allowing for the introduction of various substituents to fine-tune the solubility and electronic bandgap of the final polymer. This method ensures that the commercial scale-up of complex polymer additives is achievable with high purity and consistent quality, directly addressing the needs of the advanced materials market.
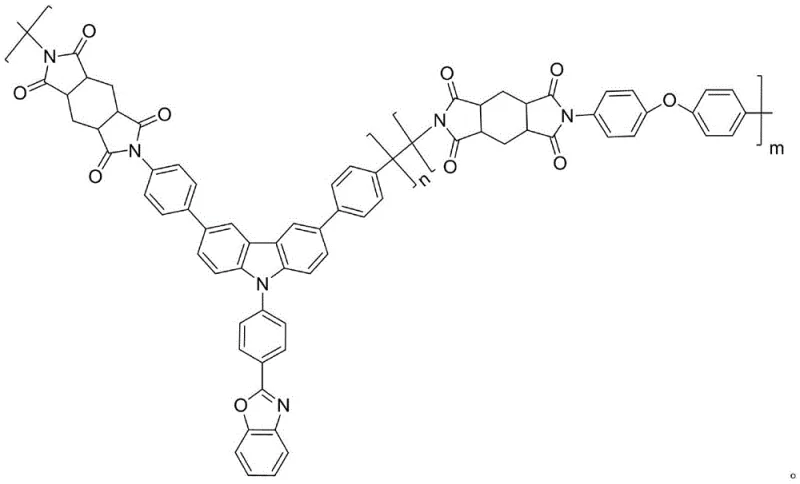
Mechanistic Insights into Suzuki-Catalyzed Assembly of Functional Diamines
The core of this synthesis lies in a sophisticated three-step sequence dominated by palladium-catalyzed Suzuki-Miyaura cross-coupling reactions, known for their high tolerance to functional groups and excellent regioselectivity. The first step involves the coupling of 2-chlorobenzoxazole with substituted p-fluorobenzeneboronic acid. In this transformation, the palladium tetratriphenylphosphine catalyst facilitates the oxidative addition into the carbon-chlorine bond of the benzoxazole, followed by transmetallation with the boronic acid species activated by the base, typically potassium carbonate. This step is critical as it establishes the electron-deficient benzoxazole unit which is essential for the subsequent electronic properties of the monomer. The reaction conditions, typically maintained at 90°C in a THF-water biphasic system, ensure complete conversion while minimizing side reactions such as homocoupling of the boronic acid.
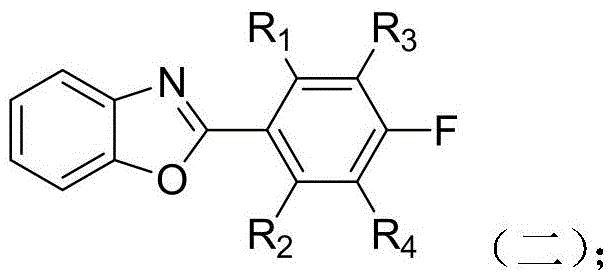
Following the formation of the benzoxazole intermediate, the second mechanistic phase involves the nucleophilic aromatic substitution or coupling with a dihalogenated carbazole monomer. This step grafts the active hydrogen of the carbazole nitrogen or utilizes the halogen positions on the carbazole ring to attach the benzoxazole unit. The use of cesium carbonate in anhydrous DMF at elevated temperatures (150°C) drives this reaction to completion, forming a robust C-N or C-C bond depending on the specific derivative used. This creates a sterically bulky intermediate that prevents close packing of polymer chains in the final material. The final step is another Suzuki coupling, this time introducing the terminal amine functionality via aminophenylboronic acid. This reaction installs the polymerizable amine groups necessary for polycondensation with dianhydrides, completing the monomer architecture with high fidelity.
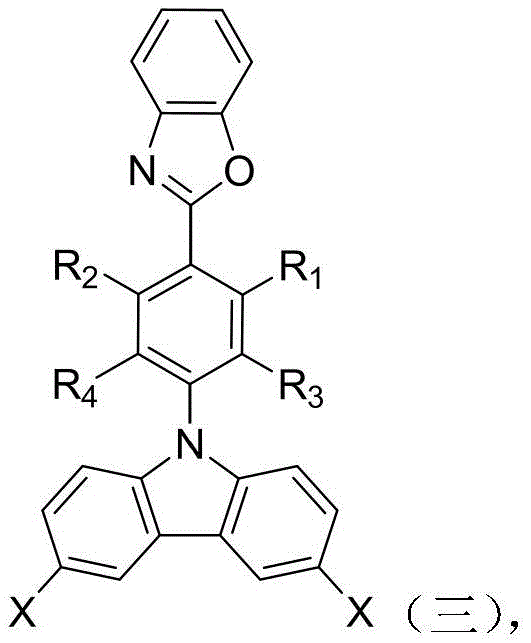
Impurity control in this multi-step synthesis is managed through careful selection of stoichiometry and purification techniques. The patent specifies the use of column chromatography and recrystallization from solvent pairs like ethyl acetate and petroleum ether to remove residual palladium catalysts and unreacted starting materials. This rigorous purification is vital for electronic grade materials, where trace metal impurities can degrade device performance. The mechanistic pathway ensures that the final diamine monomer possesses the precise structural integrity required to impart high thermal stability and obvious fluorescence characteristics to the resulting polyimide, solving the technical problems associated with strong charge transfer effects in prior art materials.
How to Synthesize Benzoxazole Carbazole Diamine Efficiently
The synthesis of these high-value diamine monomers requires precise control over reaction parameters to ensure high yields and purity suitable for electronic applications. The process begins with the preparation of the benzoxazole building block, followed by its conjugation to the carbazole core, and concludes with the installation of amine termini. Each step utilizes standard organic synthesis equipment but demands strict adherence to inert atmosphere conditions to protect the palladium catalyst and sensitive intermediates. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles that have been optimized for reproducibility.
- Perform Suzuki coupling between substituted p-fluorobenzeneboronic acid and 2-chlorobenzoxazole using palladium tetratriphenylphosphine catalyst.
- Graft the resulting benzoxazole intermediate onto a dihalogenated carbazole monomer via nucleophilic substitution in anhydrous DMF.
- Execute a final Suzuki coupling reaction with aminophenylboronic acid to introduce the terminal amine groups, followed by purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers significant strategic benefits beyond mere technical performance. The reliance on commercially available starting materials such as chlorobenzoxazole and various boronic acids ensures a stable supply chain with reduced risk of raw material shortages. The synthetic pathway avoids the use of exotic or highly toxic reagents, simplifying waste management and regulatory compliance. Furthermore, the purification methods described, primarily precipitation and recrystallization, are inherently scalable and do not require complex distillation setups, which translates to lower capital expenditure for manufacturing facilities. This streamlined process flow supports the goal of cost reduction in electronic chemical manufacturing by minimizing unit operations and maximizing throughput.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts other than standard palladium systems, combined with the use of common solvents like THF and DMF, significantly lowers the raw material cost profile. The high yields reported in the examples, often exceeding 70-80% per step, reduce the overall material loss and waste generation. Additionally, the ability to purify intermediates via simple precipitation rather than extensive chromatography on a large scale drastically reduces processing time and solvent consumption. These factors collectively contribute to a more economical production model, allowing for competitive pricing in the high-performance polymer market without compromising on quality.
- Enhanced Supply Chain Reliability: The modular nature of the Suzuki coupling chemistry allows for flexibility in sourcing. If a specific boronic acid becomes unavailable, the synthetic route can often be adapted to use alternative precursors without redesigning the entire process. The robustness of the reaction conditions, which tolerate a range of temperatures and concentrations, ensures consistent output even with minor variations in feedstock quality. This resilience is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream electronics manufacturers. The use of stable intermediates also allows for inventory buffering, further securing the supply chain against disruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction vessels and conditions that are standard in the fine chemical industry. The absence of harsh reagents like strong mineral acids or cryogenic conditions simplifies the engineering requirements for scale-up. Moreover, the aqueous workup steps and the potential for solvent recovery align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This compliance with environmental standards is increasingly important for suppliers aiming to serve global markets with strict regulatory frameworks, ensuring long-term viability and market access.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized diamine monomers. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is essential for evaluating the feasibility of integrating this material into your specific product development pipeline.
Q: What are the key advantages of this diamine monomer for polyimide synthesis?
A: The monomer incorporates benzoxazole and carbazole structures which effectively reduce charge transfer complexes, resulting in polyimides with high thermal stability and enhanced fluorescence intensity suitable for optical devices.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the patent describes a robust three-step route using standard reagents and solvents like THF and DMF, with purification methods such as precipitation and recrystallization that are amenable to large-scale manufacturing.
Q: What applications are targeted for polymers made from this monomer?
A: The resulting polyimides are designed for luminescent layer materials, photoluminescent materials, and flexible electroluminescent devices in advanced optical equipment due to their superior electronic transmission and fluorescence properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Carbazole Diamine Supplier
As the demand for high-performance optical materials continues to surge, partnering with an experienced CDMO is critical for success. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of analyzing trace impurities down to ppm levels, guaranteeing that every batch of diamine monomer meets the exacting standards required for electronic grade polyimides. We understand the nuances of handling sensitive organometallic reactions and can replicate the patented Suzuki coupling protocols with high fidelity.
We invite you to contact our technical procurement team to discuss your specific requirements. We offer a Customized Cost-Saving Analysis to help you optimize your supply chain and reduce overall manufacturing expenses. By collaborating with us, you gain access to specific COA data and route feasibility assessments tailored to your project goals. Let us help you accelerate the development of next-generation fluorescent polyimides with our reliable supply of high-purity benzoxazole carbazole diamine monomers.
