Advanced PTC-Catalyzed Synthesis of 3,4-Dimethoxyphenylacetaldehyde for Commercial API Production
Advanced PTC-Catalyzed Synthesis of 3,4-Dimethoxyphenylacetaldehyde for Commercial API Production
The pharmaceutical industry constantly seeks robust, scalable, and safe synthetic routes for critical intermediates, particularly those serving high-volume therapeutic areas such as cardiovascular and neurological disorders. Patent CN100540519C introduces a transformative methodology for the synthesis of 3,4-dimethoxyphenylacetaldehyde, a pivotal building block for major drugs like Verapamil Hydrochloride and Levodopa. This innovation shifts the paradigm from hazardous, high-cost traditional methods to a sophisticated Phase Transfer Catalysis (PTC) approach. By utilizing inexpensive inorganic bases and mild reaction conditions, this technology addresses the longstanding challenges of safety, cost, and environmental compliance in fine chemical manufacturing. For R&D directors and procurement strategists, understanding this shift is crucial for securing a reliable supply chain for these essential pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-dimethoxyphenylacetaldehyde has been plagued by significant operational hazards and economic inefficiencies. Early literature methods, such as those reported by K. Ogura, required high-pressure hydrogenation with palladium carbon catalysts, creating substantial capital expenditure barriers and safety risks associated with high-pressure vessels. Other approaches, like the one by K. Haridas, necessitated instantaneous high temperatures reaching 240°C, a condition that is energetically prohibitive and difficult to control on a multi-ton scale, often leading to thermal degradation of sensitive functional groups. Furthermore, methods described in USP5057624 and subsequent improvements by researchers like Jia Shuxiang relied heavily on potassium tert-butoxide or metallic sodium. The use of metallic sodium introduces severe fire and explosion hazards, requiring specialized inert atmosphere handling and generating large volumes of hazardous waste. Additionally, the reliance on toxic methanol and expensive organic bases like potassium 2-butoxide drastically inflates raw material costs, making these routes economically unviable for competitive cost reduction in API manufacturing.
The Novel Approach
The patented methodology presented in CN100540519C offers a decisive break from these constraints by employing a Phase Transfer Catalysis (PTC) strategy. Instead of dangerous alkali metals, the process utilizes benign inorganic bases such as potassium carbonate or sodium carbonate, which are not only significantly cheaper but also far easier to handle and dispose of safely. The reaction proceeds efficiently in common organic solvents like tetrahydrofuran (THF) or dimethylformamide (DMF) at mild temperatures ranging from 10°C to 40°C. This elimination of extreme thermal and pressure requirements simplifies reactor design and reduces energy consumption. By replacing expensive reagents like potassium 2-butoxide with cost-effective alternatives and optimizing the molar ratios of reactants, the new route achieves a total yield of 70-75% with exceptional stability. This represents a major leap forward in process chemistry, enabling the production of high-purity 3,4-dimethoxyphenylacetaldehyde with a much smaller environmental footprint and lower operational risk profile.
Mechanistic Insights into PTC-Catalyzed Darzens Condensation
The core of this technological advancement lies in the efficient execution of the Darzens condensation under phase transfer conditions. In this mechanism, the phase transfer catalyst, typically benzyltriethylammonium chloride (TEBA) or tetrabutylammonium bromide (TBAB), plays a critical role in shuttling the carbonate anion from the solid or aqueous phase into the organic phase where the reaction occurs. This facilitates the deprotonation of methyl chloroacetate to form the reactive enolate species without the need for strong, hazardous bases in the organic medium. The enolate then attacks the carbonyl carbon of 3,4-dimethoxybenzaldehyde, forming the glycidic ester intermediate. The precise control of the catalyst loading, optimized at a molar ratio of 1:0.03 to 1:0.048 relative to the aldehyde, ensures rapid reaction kinetics while minimizing side reactions. This mechanistic elegance allows the reaction to proceed smoothly at room temperature, preserving the integrity of the methoxy groups on the aromatic ring which might otherwise be susceptible to cleavage under harsher alkaline conditions.
Following the condensation, the process involves a carefully controlled hydrolysis and decarboxylation sequence. The glycidic ester is hydrolyzed using alcoholic potassium hydroxide to form the corresponding potassium salt. This step is crucial for impurity control, as the formation of the salt allows for effective purification through crystallization or extraction before the final decarboxylation. The final step involves acidification and thermal decarboxylation in a mixed solvent system, releasing carbon dioxide and yielding the target aldehyde. The mild conditions throughout this three-step sequence prevent the polymerization of the aldehyde, a common issue in aldehyde synthesis, thereby ensuring the final product meets stringent purity specifications (HPLC ≥99%). This level of control over the reaction pathway is essential for producing commercial scale-up of complex pharmaceutical intermediates that meet global regulatory standards.
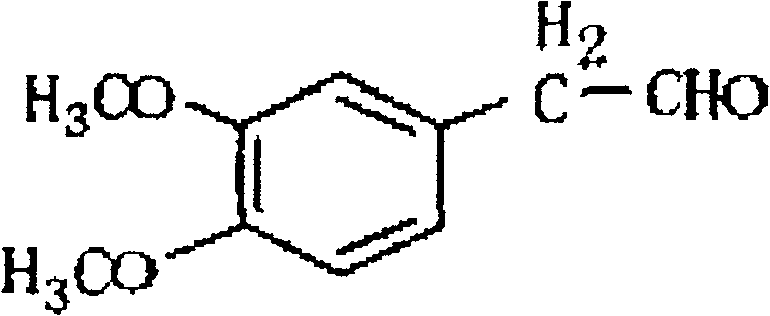
How to Synthesize 3,4-Dimethoxyphenylacetaldehyde Efficiently
Implementing this synthesis route requires precise adherence to the optimized parameters defined in the patent to ensure maximum yield and safety. The process begins with the preparation of the reaction mixture containing 3,4-dimethoxybenzaldehyde, methyl chloroacetate, and the inorganic base in the chosen solvent system, followed by the addition of the phase transfer catalyst. Maintaining the temperature within the 10-40°C window is critical during the 8 to 72-hour reaction period to balance reaction rate and selectivity. Subsequent workup involves extraction, hydrolysis, and decarboxylation steps that must be monitored closely to prevent product degradation. The detailed standardized synthesis steps, including specific reagent quantities and workup procedures for laboratory and pilot scale, are outlined below for technical reference.
- Conduct Darzens condensation of 3,4-dimethoxybenzaldehyde with methyl chloroacetate using TEBA/TBAB catalyst and inorganic base in THF/DMF at 10-40°C.
- Extract the reaction mixture with dichloromethane, dry, and hydrolyze the glycidic ester intermediate using alcoholic potassium hydroxide.
- Acidify and decarboxylate the resulting potassium salt in a mixed solvent system to isolate high-purity 3,4-dimethoxyphenylacetaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this PTC-based synthesis route offers profound strategic advantages beyond mere technical feasibility. The primary benefit is the drastic simplification of the supply chain for raw materials. By eliminating the dependency on metallic sodium and potassium tert-butoxide, manufacturers remove the need for specialized hazardous material logistics and storage facilities. This transition to stable, commodity-grade inorganic bases like potassium carbonate significantly reduces raw material procurement costs and mitigates the risk of supply disruptions caused by the limited availability of specialty reagents. Furthermore, the use of common solvents like THF and DMF, which are widely available in the global chemical market, ensures a resilient supply chain capable of withstanding regional shortages. This robustness is vital for maintaining continuous production schedules for high-demand APIs like Verapamil.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost reagents with low-cost alternatives. The removal of expensive organic bases and the avoidance of pyrophoric metals eliminate significant line items from the bill of materials. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, leading to lower utility costs per kilogram of product. The simplified waste stream, devoid of heavy metal residues or reactive metal wastes, lowers the cost of effluent treatment and disposal. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, enhancing the competitiveness of the final drug product in price-sensitive markets.
- Enhanced Supply Chain Reliability: Safety is a cornerstone of supply chain reliability. By removing flammable and explosive reagents from the process, the risk of plant shutdowns due to safety incidents is markedly reduced. The operational simplicity of the PTC method allows for more flexible production scheduling and easier staffing, as it does not require personnel with specialized training in handling hazardous pyrophoric materials. This ease of operation translates to higher plant uptime and more consistent delivery performance. For buyers, this means a more dependable source of high-purity 3,4-dimethoxyphenylacetaldehyde, reducing the need for excessive safety stock and freeing up working capital.
- Scalability and Environmental Compliance: The scalability of this process is inherent in its design. The absence of high-pressure steps and extreme temperatures means that scaling from pilot plant to commercial production (100 MT scale) requires standard glass-lined or stainless steel reactors, avoiding the need for custom-engineered high-pressure vessels. From an environmental perspective, the process aligns with green chemistry principles by using safer solvents and generating less hazardous waste. This compliance with increasingly strict environmental regulations future-proofs the supply chain against regulatory tightening, ensuring long-term viability and reducing the risk of production bans or fines related to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,4-dimethoxyphenylacetaldehyde based on the patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on why this method is becoming the industry standard for this critical intermediate. Understanding these details helps stakeholders make informed decisions regarding supplier qualification and process validation.
Q: Why is the PTC method superior to traditional sodium methoxide routes for this intermediate?
A: The PTC method eliminates the need for hazardous metallic sodium and toxic methanol, significantly improving operational safety and reducing waste treatment costs while maintaining yields above 70%.
Q: What are the critical quality parameters for 3,4-dimethoxyphenylacetaldehyde in API synthesis?
A: High purity (HPLC ≥99%) is essential to prevent downstream impurities in Verapamil and Levodopa synthesis. The patented process ensures stable content through controlled low-temperature reaction conditions.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the use of mild temperatures (10-40°C) and common solvents like THF makes the process highly scalable and suitable for large-scale industrial manufacturing without high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dimethoxyphenylacetaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the purity and consistency of its precursors. As a leading CDMO and manufacturer, we have mastered the PTC-catalyzed synthesis of 3,4-dimethoxyphenylacetaldehyde, leveraging the innovations described in CN100540519C to deliver superior value to our global partners. Our facilities are equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in clinical trials or full-scale commercialization. Our stringent purity specifications and rigorous QC labs guarantee that every batch of 3,4-dimethoxyphenylacetaldehyde meets the exacting standards required for the synthesis of life-saving cardiovascular and neurological medications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how our advanced process can improve your margins. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us be your partner in delivering high-quality pharmaceutical solutions to the market with speed, safety, and efficiency.
