Advanced Synthesis of Para-Alkenyl Halobenzenes for High-Performance Liquid Crystal Manufacturing
The landscape of electronic materials is constantly evolving, driven by the demand for higher performance liquid crystal displays. Patent CN101555192B introduces a significant breakthrough in the synthesis of para-alkenyl bromobenzene, para-alkenyl iodobenzene, and para-alkenyl chlorobenzene, which serve as critical precursors for alkenyl benzoic ether liquid crystal monomers. These monomers are essential for mixed liquid crystal formulations that enhance threshold voltage, response speed, and temperature stability in high-quality display devices. The patent discloses a novel synthetic pathway that addresses the historical instability and complexity associated with producing these specific intermediates. By leveraging a refined Grignard coupling strategy, the technology enables the production of high-purity compounds with yields ranging from 60% to 80% for the intermediates and up to 99.8% for the final esterified products. This technical advancement represents a pivotal shift for manufacturers seeking reliable liquid crystal intermediate supplier partnerships, as it simplifies the supply chain for next-generation optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyl phenylformic acid and its derivatives has been plagued by significant technical hurdles that impede efficient commercial scale-up of complex liquid crystal monomers. Traditional routes often suffer from unstable reaction conditions, requiring rigorous and often difficult-to-maintain parameters that lead to inconsistent batch quality. The complexity of the process frequently results in the formation of difficult-to-remove impurities, which negatively impact the electro-optical properties of the final liquid crystal mixture. Furthermore, conventional methods may involve multiple protection and de-protection steps or harsh reaction conditions that degrade the sensitive alkenyl functionality, leading to lower overall yields and increased waste generation. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for procurement teams aiming to secure cost reduction in electronic chemical manufacturing. The lack of a generalized, robust synthesis mode for formula (I) compounds has previously limited the availability of high-quality raw materials for the display industry.
The Novel Approach
The methodology outlined in CN101555192B offers a transformative solution by streamlining the synthesis into a more direct and controllable sequence. This novel approach utilizes a Grignard reaction between a para-halobenzyl halide and an alkenyl halide, facilitated by magnesium and a catalytic amount of iodine. The process is characterized by its operational simplicity and stability, allowing for temperature controls that are easily manageable in standard industrial reactors, specifically maintaining exotherms below 45°C during the critical coupling phase. By eliminating the need for complex multi-step sequences, this route drastically reduces the potential for side reactions and impurity formation. The result is a robust process that consistently delivers high-purity para-alkenyl halobenzenes, which can be subsequently converted into alkenyl benzoic ethers with exceptional purity profiles. This stability and simplicity are key drivers for enhancing supply chain reliability, ensuring that downstream manufacturers receive consistent quality material without the variability associated with older synthetic technologies.
Mechanistic Insights into Grignard Coupling and Esterification
The core of this synthetic innovation lies in the precise execution of the Grignard coupling reaction, which forms the carbon-carbon bond necessary to construct the alkenyl benzene skeleton. The mechanism initiates with the formation of the Grignard reagent from a para-halobenzyl halide (Formula II) in an ether or THF solvent system, activated by iodine to ensure rapid initiation at moderate temperatures. Once the organomagnesium species is generated, it acts as a potent nucleophile that attacks the alkenyl halide (Formula III) in a substitution reaction. 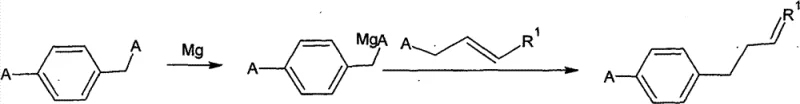 Critical to this mechanism is the strict temperature control during the addition of the alkenyl halide, which must be kept below 45°C to prevent Wurtz-type coupling side reactions or degradation of the Grignard reagent. The subsequent hydrolysis step quenches the reaction mixture, releasing the desired para-alkenyl halobenzene (Formula I) into the organic phase. This mechanistic pathway is superior because it avoids the use of transition metal catalysts that often require expensive removal steps, thereby simplifying the downstream purification process and enhancing the overall economic viability of the synthesis.
Critical to this mechanism is the strict temperature control during the addition of the alkenyl halide, which must be kept below 45°C to prevent Wurtz-type coupling side reactions or degradation of the Grignard reagent. The subsequent hydrolysis step quenches the reaction mixture, releasing the desired para-alkenyl halobenzene (Formula I) into the organic phase. This mechanistic pathway is superior because it avoids the use of transition metal catalysts that often require expensive removal steps, thereby simplifying the downstream purification process and enhancing the overall economic viability of the synthesis.
Following the formation of the intermediate, the pathway proceeds to the synthesis of alkenyl benzoic acid via carboxylation with carbon dioxide, followed by esterification to form the final liquid crystal monomer. The conversion of the para-alkenyl halobenzene to the corresponding acid involves a second Grignard formation followed by quenching with dry ice or gaseous CO2, a reaction that must be carefully managed to control the exotherm and ensure complete conversion. The final esterification step utilizes DCC (dicyclohexylcarbodiimide) and DMAP (4-dimethylaminopyridine) as coupling agents to react the acid with a phenol or alcohol derivative. 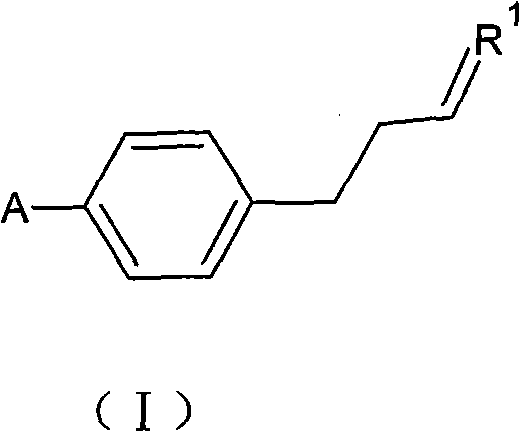 This specific esterification protocol is designed to minimize racemization or isomerization of the alkenyl chain, preserving the geometric integrity required for liquid crystal performance. The rigorous purification protocols, including acid-base washes and recrystallization, ensure that the final product meets the stringent purity specifications of 99.8% GC, which is essential for achieving the high contrast and response speeds demanded by modern display technologies.
This specific esterification protocol is designed to minimize racemization or isomerization of the alkenyl chain, preserving the geometric integrity required for liquid crystal performance. The rigorous purification protocols, including acid-base washes and recrystallization, ensure that the final product meets the stringent purity specifications of 99.8% GC, which is essential for achieving the high contrast and response speeds demanded by modern display technologies.
How to Synthesize Para-Alkenyl Halobenzene Efficiently
Implementing this synthesis route requires careful attention to reagent quality and thermal management to maximize yield and safety. The process begins with the activation of magnesium turnings, often requiring a small amount of iodine to break the oxide layer and initiate the Grignard formation in anhydrous ether or THF. Once the Grignard reagent is formed, the addition of the alkenyl halide must be performed dropwise to manage the heat of reaction, ensuring the temperature does not exceed the critical 45°C threshold which could lead to byproduct formation. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding stoichiometry and workup procedures.
- Prepare Grignard Reagent: React para-halobenzyl halide (Formula II) with Magnesium bits and Iodine catalyst in diethyl ether or THF under controlled temperature (<20°C initiation, then reflux).
- Coupling Reaction: Add alkenyl halide (Formula III) to the Grignard solution while maintaining temperature below 45°C to form the para-alkenyl halobenzene (Formula I).
- Workup and Purification: Hydrolyze with dilute hydrochloric acid, extract with toluene, dry, and distill to achieve 90-96% GC purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical feasibility. The simplification of the synthetic pathway directly correlates to a reduction in manufacturing complexity, which lowers the barrier for entry for production and reduces the risk of batch failures. By utilizing common and readily available starting materials such as para-halobenzyl halides and simple alkenyl halides, the supply chain becomes more resilient against raw material shortages. The elimination of expensive transition metal catalysts and the reduction in purification steps significantly lower the operational expenditure associated with producing these high-value intermediates. Furthermore, the robust nature of the reaction conditions allows for easier commercial scale-up of complex liquid crystal monomers, ensuring that supply can meet the fluctuating demands of the consumer electronics market without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The process eliminates the need for costly transition metal catalysts and complex purification columns often required in cross-coupling reactions, leading to substantial cost savings. By relying on magnesium, a cheap and abundant reagent, and simplifying the workup to basic acid-base extractions, the overall cost of goods sold is significantly optimized. The high yields reported in the patent, ranging from 60% to 80% for intermediates and up to 90% for acids, further contribute to economic efficiency by maximizing raw material utilization. This economic model supports a competitive pricing strategy for high-purity OLED material and liquid crystal precursors in the global market.
- Enhanced Supply Chain Reliability: The use of stable and commercially available raw materials ensures a consistent supply flow, reducing the risk of production stoppages due to material scarcity. The robustness of the Grignard reaction, when properly controlled, allows for predictable production schedules, which is critical for reducing lead time for high-purity liquid crystal intermediates. The simplified process flow also means fewer unit operations, reducing the potential for equipment bottlenecks and maintenance downtime. This reliability is paramount for long-term contracts with major display panel manufacturers who require uninterrupted material flow to maintain their own production lines.
- Scalability and Environmental Compliance: The reaction conditions are amenable to large-scale batch processing, with temperature controls that are easily managed in standard industrial reactors. The waste stream is primarily composed of magnesium salts and organic solvents which can be recovered and recycled, aligning with modern environmental compliance standards. The high purity of the final product reduces the need for extensive downstream processing, thereby lowering the overall environmental footprint of the manufacturing process. This scalability ensures that the technology can grow with market demand, supporting the production of tons of material annually without significant re-engineering of the process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the purity levels achievable with this synthesis method?
A: According to patent data, the intermediate para-alkenyl halobenzene achieves 90-96% GC purity, while the final alkenyl benzoic ether reaches 99.8% purity after recrystallization.
Q: Why is this Grignard route preferred for liquid crystal intermediates?
A: This method offers a simpler and more stable process compared to conventional routes, avoiding complex purification steps and ensuring consistent quality for high-performance display materials.
Q: Can this process be scaled for industrial production?
A: Yes, the process uses common solvents like toluene and ether, with manageable exothermic controls (<45°C), making it highly suitable for commercial scale-up of complex liquid crystal monomers.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Para-Alkenyl Halobenzene Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to translate complex patent routes like CN101555192B into commercial reality. Our R&D team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced GC-MS and HPLC systems to guarantee that every batch of para-alkenyl halobenzene meets the exacting standards required for liquid crystal applications. Our commitment to quality ensures that our clients receive materials that consistently perform in their final formulations, minimizing the risk of downstream failures.
We invite global partners to collaborate with us to optimize their supply chains and achieve significant competitive advantages. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential clients to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your partner in driving innovation and efficiency in the electronic materials sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
