Advanced Biocatalytic Synthesis of Chiral Beta-Amino Acid Esters for Commercial Scale-Up
Advanced Biocatalytic Synthesis of Chiral Beta-Amino Acid Esters for Commercial Scale-Up
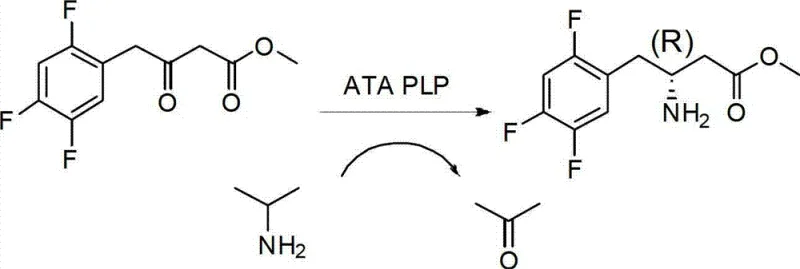
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more efficient synthetic methodologies, particularly for the production of complex chiral intermediates. Patent CN103014081A introduces a groundbreaking biological preparation method for synthesizing 3-amino-4-(2,4,5-trifluorophenyl)methyl butyrate, a critical chiral building block. This innovation leverages recombinant transaminase technology to convert 3-carbonyl-4-(2,4,5-trifluorophenyl)methyl butyrate into its corresponding amino derivative with exceptional stereocontrol. Unlike traditional chemical routes that often rely on harsh conditions and precious metal catalysts, this biocatalytic approach operates in a mild aqueous buffer system at neutral pH. For R&D directors and process chemists, this represents a significant advancement in accessing high-value beta-amino acid derivatives with reduced environmental impact and simplified purification workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-amino acids and their esters has relied heavily on asymmetric hydrogenation using transition metal catalysts, such as ruthenium complexes. While effective, these conventional chemical methods present substantial challenges for large-scale manufacturing and supply chain management. The requirement for high-pressure hydrogen gas introduces significant safety hazards, necessitating specialized high-pressure reactors and rigorous safety protocols that increase capital expenditure. Furthermore, the use of heavy metal catalysts creates a persistent risk of metal contamination in the final product, mandating complex and costly downstream purification steps to meet stringent pharmaceutical regulatory limits. Additionally, achieving high enantioselectivity often requires expensive chiral ligands and precise control over reaction parameters, leading to variable yields and increased production costs.
The Novel Approach
In stark contrast, the novel biocatalytic route described in the patent utilizes a recombinant transaminase to facilitate the asymmetric amination of the keto-ester substrate under ambient pressure and mild temperatures. This enzymatic transformation proceeds in a water-phase buffer solution containing a cosolvent like DMSO to enhance substrate solubility, eliminating the need for volatile organic solvents typically associated with chemical synthesis. The use of isopropylamine as an amino donor drives the equilibrium towards the desired product while generating acetone as a benign byproduct that can be easily removed. This approach not only simplifies the reaction setup by removing the need for high-pressure equipment but also inherently ensures high optical purity due to the enzyme's natural stereoselectivity. By shifting from a metal-catalyzed process to a biocatalytic one, manufacturers can achieve a cleaner reaction profile with fewer by-products.
Mechanistic Insights into Recombinant Transaminase Catalysis
The core of this technological breakthrough lies in the mechanism of the pyridoxal phosphate (PLP)-dependent transaminase, which acts as a highly specific biocatalyst. The enzyme facilitates the transfer of an amino group from the donor, isopropylamine, to the prochiral ketone substrate through a ping-pong bi-bi mechanism involving the formation of a Schiff base intermediate. Initially, the internal aldimine formed between PLP and a lysine residue in the enzyme active site reacts with the amino donor to form a quinonoid intermediate, which subsequently collapses to release acetone and generate the external aldimine (PMP form of the enzyme). This PMP-enzyme complex then reacts with the 3-carbonyl substrate, transferring the amino group to form the chiral amine product and regenerating the PLP-enzyme complex. The strict spatial arrangement of the enzyme's active site ensures that only the (R)-enantiomer is produced, providing the high optical activity observed in the experimental data.
From an impurity control perspective, this enzymatic mechanism offers distinct advantages over chemical catalysis. Because the enzyme is highly specific for the ketone functionality and the specific steric environment of the 2,4,5-trifluorophenyl group, side reactions such as over-reduction or non-specific alkylation are virtually eliminated. The reaction conditions, specifically the pH range of 6.0 to 9.0 and temperature of 25-45°C, are optimized to maintain enzyme stability while maximizing turnover frequency. The patent data indicates that maintaining the pH at 7.0 provides the optimal balance for enzyme activity and substrate stability. Furthermore, the use of a recombinant enzyme allows for potential protein engineering to further enhance stability or activity, offering a pathway for continuous process improvement that is not feasible with static chemical catalysts.
How to Synthesize 3-amino-4-(2,4,5-trifluorophenyl)methyl butyrate Efficiently
The implementation of this biocatalytic process is straightforward and designed for scalability, making it an attractive option for contract development and manufacturing organizations (CDMOs). The protocol involves preparing a reaction mixture containing the substrate, amino donor, cofactor, and enzyme in a buffered aqueous system, followed by incubation under controlled conditions. The simplicity of the workup procedure, which involves pH adjustment and standard solvent extraction, underscores the practicality of this method for industrial application. For detailed operational parameters and specific reagent ratios, please refer to the standardized synthesis guide below.
- Prepare the reaction system by adding pH 7.0 phosphate buffer, substrate, isopropylamine, DMSO cosolvent, PLP cofactor, and recombinant transaminase into a reactor.
- Maintain the reaction mixture at 30°C with mechanical stirring under nitrogen purging for 24 hours to ensure high conversion efficiency.
- Adjust the pH to 2-3 with hydrochloric acid, filter through diatomite, extract with ethyl acetate, and evaporate solvent to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of high-pressure hydrogenation equipment reduces the barrier to entry for manufacturing this intermediate, allowing for a broader base of qualified suppliers and reducing dependency on specialized facilities. Moreover, the removal of ruthenium catalysts from the process flow significantly lowers raw material costs and eliminates the need for expensive metal scavenging resins or activated carbon treatments during purification. This simplification of the downstream processing train results in shorter batch cycles and higher overall equipment effectiveness, directly contributing to cost reduction in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Cost Reduction in Manufacturing: The transition from a noble metal-catalyzed process to an enzymatic one fundamentally alters the cost drivers of production. By removing the requirement for ruthenium catalysts and high-pressure reactors, the capital intensity of the manufacturing process is drastically reduced. Additionally, the high conversion rates (>95%) and excellent selectivity minimize waste generation and maximize atom economy, leading to substantial cost savings in raw material consumption. The ability to operate at ambient pressure and moderate temperatures also reduces energy consumption associated with heating, cooling, and compression, further enhancing the economic viability of the process for large-scale commercial production.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly improved by the use of commercially available and stable reagents such as isopropylamine and phosphate buffers, which are less susceptible to geopolitical supply disruptions compared to specialized chiral ligands or precious metals. The robust nature of the recombinant transaminase, which can be produced via fermentation, ensures a consistent and scalable supply of the biocatalyst. This reliability allows for better production planning and inventory management, reducing lead time for high-purity pharmaceutical intermediates and ensuring continuity of supply for downstream API synthesis even in volatile market conditions.
- Scalability and Environmental Compliance: The aqueous nature of the reaction medium aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The process generates minimal hazardous waste, primarily consisting of aqueous streams that are easier to treat than organic solvent waste from traditional chemical synthesis. This environmental compatibility simplifies the permitting process for new manufacturing lines and reduces the long-term liability associated with waste disposal. Furthermore, the demonstrated scalability from gram to 100-gram scales in the patent examples suggests that the process can be readily adapted to multi-kilogram or ton-scale production with minimal re-optimization, facilitating rapid commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the biocatalytic production of this chiral intermediate. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What are the advantages of using transaminase over traditional chemical hydrogenation for this substrate?
A: The biocatalytic method eliminates the need for high-pressure hydrogen and expensive ruthenium catalysts, significantly improving operational safety and reducing heavy metal contamination risks in the final API.
Q: What optical purity can be achieved with this recombinant transaminase process?
A: The patented process demonstrates exceptional stereoselectivity, consistently achieving optical purity greater than 99% ee, which is critical for downstream pharmaceutical applications.
Q: Is this enzymatic process scalable for industrial production?
A: Yes, the patent explicitly describes scaling from gram-scale laboratory experiments to 100g+ pilot batches with consistent yields above 95%, indicating robust scalability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-amino-4-(2,4,5-trifluorophenyl)methyl butyrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of biocatalysis in modern pharmaceutical manufacturing and are well-positioned to support your development needs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like this transaminase route can be successfully translated into robust industrial operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced chiral HPLC and GC-MS instrumentation to guarantee that every batch of 3-amino-4-(2,4,5-trifluorophenyl)methyl butyrate meets the highest quality standards required for global regulatory submissions.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green route can optimize your bill of materials. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical chiral intermediates.
