Optimizing Meptazinol Hydrochloride Production: A Technical & Commercial Analysis
The pharmaceutical industry continuously seeks robust manufacturing pathways for analgesic agents, and Patent CN1271057C presents a significant advancement in the synthesis of 1-methyl-3-ethyl-3-(3-hydroxyphenyl)-hexahydro-1H-azepine hydrochloride, commonly known as Meptazinol Hydrochloride. This specific intellectual property outlines a method that diverges from traditional high-cost routes by leveraging a Willgerodt rearrangement followed by a strategic cyclization and reduction sequence. For R&D Directors and Procurement Managers, the value proposition lies in the substitution of hazardous and expensive reagents with more industrially friendly alternatives, such as replacing liquefied ammonia with sodium amide in non-polar solvents. The patent explicitly details a multi-step process that begins with m-methoxyacetophenone, transforming it through acylation and condensation into a key amide intermediate before closing the seven-membered azepine ring. This technical breakthrough not only addresses the purity concerns often associated with older synthetic methods but also aligns with modern green chemistry principles by reducing the need for extreme cryogenic conditions. As a reliable pharmaceutical intermediates supplier, understanding the nuances of this patent is crucial for evaluating the long-term viability and cost-efficiency of the supply chain for this potent analgesic compound.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Meptazinol Hydrochloride has been plagued by significant operational hurdles that impact both cost reduction in pharmaceutical manufacturing and overall process safety. Prior art, such as the methods described in GB1285025, necessitates the use of liquefied ammonia at low temperatures, creating a complex infrastructure requirement for cryogenic storage and handling that is often impractical for standard chemical plants. Furthermore, alternative routes like those found in US4197239 rely heavily on butyllithium, a pyrophoric reagent that demands stringent safety protocols and drives up the cost of goods sold due to the expense of the reagent itself and the specialized equipment needed to manage it. These conventional pathways often suffer from reproducibility issues, where the acylation cyclization steps fail to yield consistent results on a larger scale, leading to batch failures and extended lead times for high-purity pharmaceutical intermediates. The reliance on such harsh conditions also complicates the impurity profile, often requiring extensive downstream purification which further erodes profit margins and delays time-to-market for generic formulations.
The Novel Approach
The methodology disclosed in Patent CN1271057C offers a transformative solution by re-engineering the synthetic route to prioritize mild reaction conditions and commercially available reagents. Instead of cryogenic ammonia, the novel approach utilizes sodium amide as a base for the critical cyclization step, conducted in non-polar solvents which are easier to handle and recover on an industrial scale. This shift eliminates the need for specialized low-temperature reactors, thereby drastically simplifying the capital expenditure required for commercial scale-up of complex pharmaceutical intermediates. Additionally, the ethylation step employs potassium tert-butoxide and ethyl iodide, a combination that significantly reduces the formation of oxygenated by-products compared to previous methods, enhancing the overall yield and purity of the intermediate. By streamlining the reduction phases through a combination of Wolff-Kishner-Huang and lithium aluminum hydride reactions, the process ensures a high degree of conversion while maintaining a manageable impurity spectrum. This approach represents a paradigm shift towards more sustainable and economically viable manufacturing, appealing directly to supply chain heads looking for continuity and reliability.
Mechanistic Insights into Sodium Amide-Catalyzed Cyclization
The core of this synthetic innovation lies in the cyclization of the N-methyl-N-(3-methoxycarbonyl-propyl)-(3-methoxyphenyl)acetamide intermediate, a step that dictates the formation of the seven-membered azepine ring. In this mechanism, sodium amide acts as a strong base to deprotonate the active methylene group, initiating an intramolecular nucleophilic attack that closes the ring to form 1-methyl-3-(3-methoxyphenyl)-hexahydro-1H-azepine-2,4-dione. The choice of a non-polar solvent environment is critical here, as it stabilizes the reactive intermediates and prevents side reactions that could lead to polymerization or ring-opening. This specific mechanistic pathway allows for precise control over the regioselectivity of the cyclization, ensuring that the nitrogen atom is correctly positioned within the ring structure. For R&D teams, understanding this mechanism is vital for troubleshooting potential scale-up issues, as the stoichiometry of the sodium amide and the dryness of the solvent are key parameters that influence the reaction kinetics and final yield.
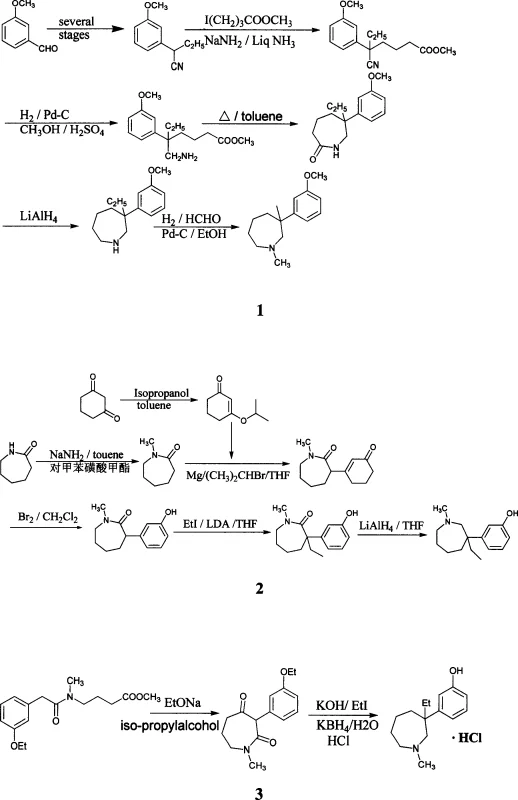
Following the ring closure, the subsequent ethylation and reduction steps are equally critical for defining the final impurity profile of the API. The ethylation reaction, utilizing potassium tert-butoxide, is designed to minimize O-alkylation, a common side reaction that can be difficult to separate from the desired N-alkylated product. The patent highlights that this specific base and solvent combination reduces the oxygenation ratio, thereby simplifying the purification workload. Subsequent reduction via the Wolff-Kishner-Huang method effectively removes the carbonyl oxygens, while the final lithium aluminum hydride reduction ensures the complete conversion of the remaining ketone to the methylene group. This two-stage reduction strategy is superior to single-step reductions which often struggle with selectivity, ensuring that the final 1-methyl-3-ethyl-3-(3-hydroxyphenyl)-hexahydro-1H-azepine structure is obtained with high fidelity. The final salt formation with hydrogen chloride gas in isopropanol yields the stable hydrochloride salt, ready for formulation, demonstrating a complete and robust chemical lifecycle from raw material to finished API intermediate.
How to Synthesize Meptazinol Hydrochloride Efficiently
Implementing this synthesis route requires a disciplined approach to reaction monitoring and parameter control to ensure the theoretical yields described in the patent are realized in a production environment. The process begins with the preparation of the key amide intermediate, followed by the critical cyclization step where moisture control is paramount to prevent the decomposition of sodium amide. Detailed standardized synthesis steps are essential for maintaining batch-to-batch consistency, particularly during the ethylation and reduction phases where temperature gradients can affect product quality. Operators must be trained to handle the specific work-up procedures, such as the acidification and extraction steps, to maximize recovery and minimize waste. For facilities looking to adopt this technology, the following guide outlines the critical operational milestones.
- Preparation of key intermediate N-methyl-N-(3-methoxycarbonyl-propyl)-(3-methoxyphenyl)acetamide via Willgerodt rearrangement and acylation.
- Cyclization using sodium amide in non-polar solvents to form the azepine-2,4-dione core structure.
- Sequential ethylation, Wolff-Kishner-Huang reduction, and lithium aluminum hydride reduction to yield the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthesis method described in Patent CN1271057C offers substantial strategic benefits for procurement managers and supply chain directors tasked with optimizing the cost structure of analgesic production. The primary advantage stems from the elimination of cryogenic reagents and pyrophoric organometallics, which not only reduces the direct cost of raw materials but also lowers the indirect costs associated with safety compliance and specialized equipment maintenance. By shifting to reagents like sodium amide and potassium tert-butoxide, manufacturers can achieve significant cost savings in pharmaceutical manufacturing without compromising on the quality or purity of the final product. This transition also mitigates supply chain risks, as the new reagents are more widely available and less subject to the logistical constraints that often plague hazardous chemical shipments. Consequently, this leads to enhanced supply chain reliability, ensuring that production schedules are met without unexpected delays caused by reagent shortages or safety incidents.
- Cost Reduction in Manufacturing: The replacement of expensive butyllithium and liquefied ammonia with more common industrial bases drastically lowers the variable cost per kilogram of the produced API. This chemical substitution removes the need for energy-intensive cooling systems, resulting in lower utility costs and a reduced carbon footprint for the manufacturing facility. Furthermore, the improved selectivity of the ethylation step reduces the volume of waste solvents and by-products that require disposal, contributing to overall operational efficiency. These cumulative effects create a leaner cost structure that allows for more competitive pricing in the generic pharmaceutical market while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Utilizing readily available reagents ensures that the production of Meptazinol Hydrochloride is not vulnerable to the supply disruptions often associated with specialized or hazardous chemicals. The mild reaction conditions allow for production in a wider range of standard chemical plants, increasing the potential pool of qualified suppliers and reducing dependency on single-source vendors. This diversification strengthens the supply chain against geopolitical or logistical shocks, ensuring a continuous flow of high-purity pharmaceutical intermediates to downstream formulation partners. Additionally, the simplified safety profile reduces the likelihood of production stoppages due to regulatory inspections or safety audits, further stabilizing the supply timeline.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily managed in large-scale reactors without the need for exotic materials of construction. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, minimizing the risk of compliance penalties and facilitating smoother permitting processes for capacity expansion. The ability to run these reactions under atmospheric pressure and moderate temperatures also reduces the energy intensity of the process, supporting corporate sustainability goals. This scalability ensures that as market demand for the analgesic grows, the manufacturing capacity can be expanded rapidly and efficiently to meet commercial needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity for stakeholders evaluating its adoption. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details is essential for assessing the feasibility of integrating this process into existing manufacturing portfolios.
Q: What are the primary cost drivers in conventional Meptazinol synthesis?
A: Conventional methods often rely on cryogenic conditions using liquefied ammonia or expensive organolithium reagents like butyllithium, which significantly increase operational costs and safety risks.
Q: How does the new process improve impurity control?
A: By utilizing sodium amide in non-polar solvents for cyclization and specific ethylation conditions with potassium tert-butoxide, the process minimizes oxygenation by-products and simplifies purification.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method employs mild reaction conditions, avoids extreme low temperatures, and uses readily available raw materials, making it highly adaptable for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Meptazinol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes like Patent CN1271057C to maintain competitiveness in the global pharmaceutical market. Our CDMO expertise allows us to translate complex laboratory protocols into robust industrial processes, ensuring that the technical advantages of this method are fully realized in commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of Meptazinol Hydrochloride meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and leverage the cost benefits of this innovative synthesis method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your API sourcing strategy. By partnering with us, you gain access to a reliable supply of high-quality intermediates backed by deep technical expertise and a commitment to continuous improvement.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
