Scalable Synthesis of 3-Methyl-7,8-Dihydroxyisochromanone-4 for Commercial Pharmaceutical Applications
Scalable Synthesis of 3-Methyl-7,8-Dihydroxyisochromanone-4 for Commercial Pharmaceutical Applications
The pharmaceutical industry constantly seeks reliable sources for bioactive natural products that can be transitioned from limited extraction to robust chemical synthesis. Patent CN101016289A details a groundbreaking methodology for the total synthesis of 3-methyl-7,8-dihydroxyisochromanone-4, a novel natural product originally isolated from banana peels with significant antihypertensive activity comparable to captopril. This technical insight report analyzes the proprietary synthetic route disclosed in the patent, highlighting its potential for industrial scale-up and supply chain stabilization. By shifting from agricultural extraction to a defined chemical pathway starting from 2-methyl-3,4-dimethoxybenzoic acid, manufacturers can secure a consistent supply of this high-value pharmaceutical intermediate. The following analysis dissects the reaction engineering, mechanistic nuances, and commercial implications for R&D and procurement leaders seeking to integrate this molecule into their cardiovascular drug pipelines.
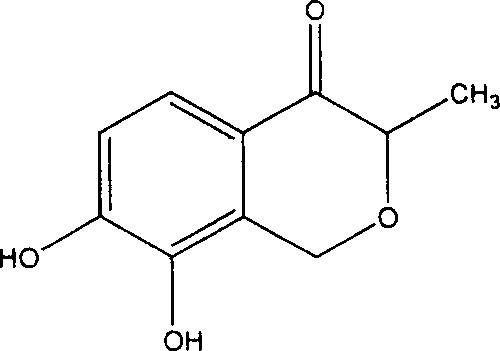
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the sourcing of 3-methyl-7,8-dihydroxyisochromanone-4 relied entirely on the extraction of effective parts from banana peels, a process fraught with inherent supply chain vulnerabilities and quality inconsistencies. Natural extraction is heavily dependent on agricultural seasons, geographic origin, and the variable concentration of the active ingredient within the plant matrix, leading to unpredictable yields and fluctuating market availability. Furthermore, the purification of natural extracts often requires extensive chromatographic separation to remove complex biological impurities, resulting in high operational costs and significant solvent waste generation. For a pharmaceutical application requiring stringent purity specifications and regulatory compliance, relying on a botanical source introduces unacceptable risks regarding batch-to-batch reproducibility and the potential presence of unknown allergens or contaminants that are difficult to characterize and control.
The Novel Approach
The synthetic methodology outlined in patent CN101016289A represents a paradigm shift by establishing a fully synthetic route that bypasses the limitations of natural sourcing. This approach utilizes readily available commodity chemicals, specifically 2-methyl-3,4-dimethoxybenzoic acid, as the starting material, ensuring a stable and cost-effective raw material base. The process employs a logical sequence of esterification, bromination, etherification, and cyclization reactions that are well-understood in organic synthesis, allowing for precise control over reaction parameters such as temperature, pressure, and stoichiometry. By constructing the isochromanone core chemically, manufacturers can achieve superior purity profiles and eliminate the variability associated with biological systems. This transition from extraction to synthesis not only secures the supply chain but also opens the door for structural analogs and derivatives, providing a robust platform for medicinal chemistry optimization and cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Dieckmann and Perkin Cyclization Strategies
The core innovation of this synthesis lies in the construction of the isochromanone ring system, which is achieved through two distinct intramolecular cyclization pathways: the Dieckmann condensation and the intramolecular Perkin condensation. In the Dieckmann route, the diester intermediate undergoes base-catalyzed intramolecular Claisen condensation, where an enolate formed at the alpha-position of one ester group attacks the carbonyl carbon of the adjacent ester moiety. This nucleophilic acyl substitution results in the formation of a beta-keto ester cyclic intermediate, which subsequently undergoes hydrolysis and decarboxylation to yield the desired ketone functionality within the ring. This mechanism is highly efficient for forming five- and six-membered rings and allows for the introduction of the methyl group at the 3-position with high regioselectivity, driven by the thermodynamic stability of the resulting enolate.
Alternatively, the patent describes a pathway involving hydrolysis of the diester to a keto-acid followed by an intramolecular Perkin-type condensation. In this mechanism, the carboxylic acid moiety is activated, typically via anhydride formation in situ, facilitating nucleophilic attack by the enolizable position on the adjacent carbonyl. This route offers a valuable contingency for process optimization, particularly if the direct Dieckmann conditions lead to specific side reactions or impurity profiles that are difficult to purge. Both pathways converge on the formation of 3-methyl-7,8-dimethoxyisochromanone-4, which is then subjected to demethylation using Lewis acids such as aluminum trichloride or strong inorganic acids to reveal the critical catechol motif essential for biological activity. Understanding these mechanistic details is crucial for R&D teams to optimize reaction conditions and minimize impurity formation during scale-up.
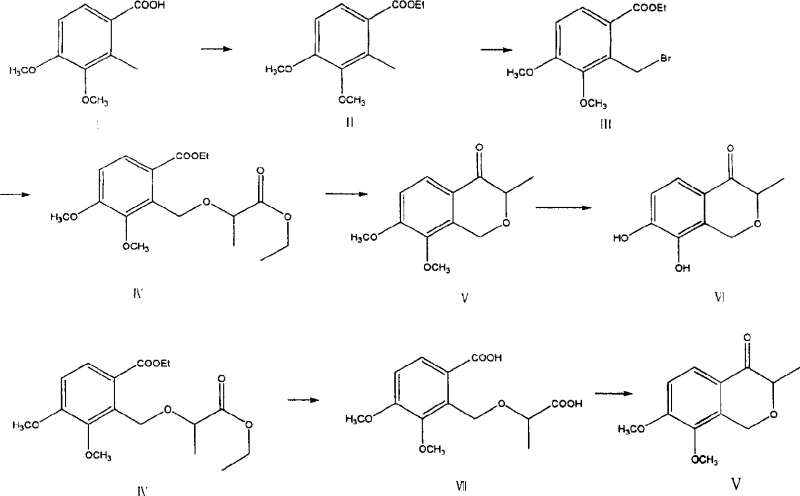
How to Synthesize 3-Methyl-7,8-Dihydroxyisochromanone-4 Efficiently
The synthesis of this antihypertensive intermediate requires careful attention to reaction stoichiometry and purification techniques at each stage to ensure high overall yield and purity. The process begins with the activation of the benzoic acid starting material, followed by functionalization of the benzylic position to enable coupling with the lactate side chain. The critical cyclization step demands anhydrous conditions and precise temperature control to favor intramolecular ring closure over intermolecular polymerization. Following the formation of the protected isochromanone core, the final demethylation step must be managed to prevent over-reaction or degradation of the sensitive catechol system. For detailed operational parameters, reagent grades, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process steps derived from the patent examples.
- Esterification of 2-methyl-3,4-dimethoxybenzoic acid with ethanol using thionyl chloride or sulfuric acid catalyst to form the ethyl ester.
- Radical bromination of the methyl group using NBS or bromine to generate the bromomethyl intermediate.
- Etherification reaction between the bromomethyl intermediate and ethyl lactate under basic conditions to link the side chain.
- Intramolecular cyclization via Dieckmann condensation or Perkin condensation to form the isochromanone core structure.
- Final demethylation using Lewis acids like aluminum trichloride or inorganic acids to yield the target dihydroxy compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this synthetic route offers profound strategic advantages beyond mere technical feasibility. By decoupling the production of this key intermediate from agricultural cycles, organizations can achieve a level of supply security that is impossible with natural extraction. The use of commodity starting materials like dimethoxybenzoic acid and ethyl lactate ensures that raw material costs remain stable and predictable, shielding the project from the volatility of crop yields and climate-related disruptions. Furthermore, the synthetic pathway is designed using standard unit operations common in fine chemical manufacturing, such as reflux, filtration, and distillation, which facilitates seamless technology transfer to existing multipurpose production facilities without the need for specialized equipment investments.
- Cost Reduction in Manufacturing: The synthetic route eliminates the massive solvent volumes and low-efficiency extraction steps associated with processing tons of biomass to obtain grams of active ingredient. By utilizing high-yielding chemical transformations, the overall material throughput is drastically improved, leading to substantial cost savings per kilogram of finished product. Additionally, the ability to recycle solvents like toluene and ethanol in the esterification and cyclization steps further enhances the economic viability of the process, making it highly competitive against extraction-based sourcing models while maintaining rigorous quality standards.
- Enhanced Supply Chain Reliability: Relying on a chemical synthesis route fundamentally transforms the supply risk profile from high to low. Unlike natural products which are subject to harvest failures and geopolitical trade restrictions on agricultural goods, the precursors for this synthesis are produced by established global chemical suppliers with redundant capacity. This ensures continuous availability of raw materials and allows for long-term supply agreements with fixed pricing structures. The robustness of the synthetic method also means that production can be ramped up rapidly in response to market demand spikes, providing a agile response capability that natural extraction simply cannot match.
- Scalability and Environmental Compliance: The process is inherently scalable, moving efficiently from gram-scale laboratory synthesis to multi-ton commercial production without fundamental changes to the reaction chemistry. From an environmental perspective, the synthetic route generates a more defined waste stream compared to the complex organic sludge produced by plant extraction, simplifying wastewater treatment and disposal compliance. The elimination of biomass processing also reduces the carbon footprint associated with the transportation and handling of bulky agricultural raw materials, aligning the manufacturing process with modern sustainability goals and green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-methyl-7,8-dihydroxyisochromanone-4. These answers are derived directly from the technical specifications and experimental data provided in the underlying patent literature, ensuring accuracy and relevance for decision-makers evaluating this technology for their pipeline.
Q: What are the advantages of synthesizing 3-methyl-7,8-dihydroxyisochromanone-4 over natural extraction?
A: Synthetic production eliminates the dependency on seasonal agricultural sources like banana peels, ensuring consistent supply continuity, higher purity profiles, and significantly reduced batch-to-batch variability compared to natural extraction methods.
Q: Which cyclization method is preferred for large-scale manufacturing?
A: While both Dieckmann and Perkin condensations are viable, the Dieckmann route offers a more direct path from the diester intermediate, potentially reducing unit operations, whereas the Perkin route via hydrolysis provides an alternative if specific impurity profiles need to be managed differently.
Q: How is the purity of the final antihypertensive intermediate controlled?
A: Purity is rigorously controlled through optimized crystallization steps after the cyclization and demethylation reactions, alongside standard silica gel column chromatography during process development to remove regio-isomers and unreacted starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Methyl-7,8-Dihydroxyisochromanone-4 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing high-quality intermediates for cardiovascular drug development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-methyl-7,8-dihydroxyisochromanone-4 meets the highest international standards. Our commitment to technical excellence allows us to navigate the complexities of the Dieckmann and Perkin cyclization steps with precision, delivering a product that is ready for downstream formulation.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume needs, helping you optimize your budget without compromising on quality. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex pharmaceutical intermediate synthesis.
