Scalable Synthesis of Gadoxetate Disodium Intermediates for Commercial MRI Contrast Agents
The pharmaceutical landscape for magnetic resonance imaging (MRI) contrast agents is undergoing a significant transformation driven by the need for higher purity intermediates and more robust manufacturing processes. Patent CN103420862A introduces a groundbreaking methodology for synthesizing the key metal salt intermediates of Gadoxetate Disodium, a liver-specific contrast agent critical for early diagnosis of hepatic lesions. This innovation addresses the longstanding challenges associated with the stability and scalability of the (4S)-4-(4-ethoxybenzyl)-3,6,9-tris(carboxymethyl)-3,6,9-triazaundecanedioic acid metal salts. By shifting from amorphous, hygroscopic materials to well-defined crystalline structures such as Li5-EOB-DTPA, Na5-EOB-DTPA, and K5-EOB-DTPA, the technology enables a more reliable supply chain for high-purity pharmaceutical intermediates. The strategic implementation of this crystallization technique not only enhances the chemical stability of the intermediate but also streamlines the downstream chelation process with gadolinium oxide. For global procurement teams, this represents a pivotal opportunity to secure a reliable Gadoxetate Disodium intermediate supplier capable of meeting stringent regulatory standards without the bottlenecks of traditional purification methods. The ability to produce these intermediates with purity exceeding 98% directly correlates to the quality of the final active pharmaceutical ingredient, ensuring patient safety and diagnostic accuracy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Gadoxetate Disodium intermediates has been plagued by significant technical hurdles that impede large-scale commercialization and increase overall manufacturing costs. Prior art methods, such as those described in US5695739A, often rely on the direct synthesis from protected precursors followed by complex preparative liquid chromatography to achieve acceptable purity levels. This reliance on chromatographic separation is inherently inefficient for industrial scale-up, as it introduces substantial solvent consumption, extended processing times, and high operational expenditures that erode profit margins. Furthermore, the intermediates produced via these traditional routes frequently exist in an amorphous state, which renders them highly hygroscopic and prone to becoming viscous or even liquefying upon prolonged storage. This physical instability creates severe logistical challenges for supply chain heads, as it necessitates specialized storage conditions and increases the risk of material degradation during transit. The inability to effectively remove impurities without chromatography also means that batch-to-batch consistency is difficult to maintain, posing a risk to the quality control protocols required by major regulatory bodies. Consequently, the conventional approach limits the availability of cost reduction in MRI contrast agent manufacturing and restricts the ability of suppliers to meet growing global demand.
The Novel Approach
The methodology disclosed in CN103420862A offers a transformative solution by utilizing a direct alkaline hydrolysis and controlled crystallization strategy that bypasses the need for chromatographic purification entirely. By reacting the protected precursor Compound II with specific alkali metal hydroxides in alcohol solvents under reflux conditions, the process facilitates the simultaneous deprotection and salt formation of the intermediate. This one-pot reaction design significantly simplifies the operational workflow, allowing for the direct isolation of the product through filtration after cooling the reaction mixture. The resulting crystalline salts exhibit superior physical properties, including high melting points above 300°C and excellent stability against moisture absorption, which resolves the storage and handling issues associated with amorphous forms. This novel approach not only ensures that the intermediate compound product has a purity of more than 98% but also enables the further synthesis of high-purity Gadoxetate Disodium with yields exceeding 88%. For procurement managers, this translates to a drastic simplification of the supply chain, reducing lead time for high-purity pharmaceutical intermediates and minimizing the risk of production delays. The robustness of this crystallization process makes it ideally suited for the commercial scale-up of complex pharmaceutical intermediates, providing a sustainable pathway for mass production.
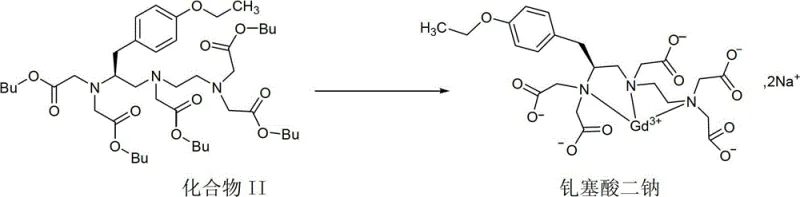
Mechanistic Insights into Alkaline Hydrolysis and Crystallization
The core of this technological advancement lies in the precise control of the deprotection reaction and the subsequent crystallization kinetics, which dictate the purity and physical form of the final intermediate. The reaction mechanism involves the nucleophilic attack of hydroxide ions on the tert-butoxycarbonyl and tert-butyl ester groups of Compound II, cleaving the protecting groups to reveal the free carboxylic acids which immediately form salts with the alkali metal cations. The choice of solvent, typically methanol, ethanol, or isopropanol, plays a critical role in modulating the solubility of the intermediate during the reaction and cooling phases, thereby influencing the crystal lattice formation. By maintaining specific molar ratios of the hydroxide to the precursor, ranging from 1:5 to 1:15, the process ensures complete conversion while minimizing the formation of side products that could contaminate the crystal lattice. The crystallization step is particularly vital, as it acts as a purification mechanism where impurities remain in the mother liquor while the pure salt precipitates out in a defined crystalline structure. This mechanism effectively eliminates the need for external purification columns, leveraging thermodynamic principles to achieve high chemical purity. For R&D directors, understanding this mechanism is crucial for optimizing process parameters such as reflux time and cooling rates to maximize yield and ensure the consistent formation of the desired polymorph. The ability to tune the crystal form by selecting different alkali metals (Li, Na, K) provides additional flexibility in tailoring the physical properties of the intermediate for specific downstream processing requirements.
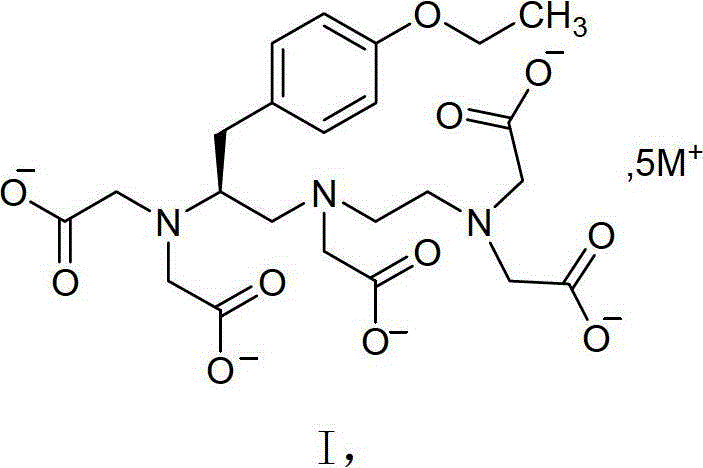
Impurity control is another critical aspect of this mechanistic pathway, as the removal of residual starting materials and by-products is essential for meeting the stringent specifications of MRI contrast agents. The crystalline nature of the intermediate inherently rejects impurities during the lattice growth phase, a phenomenon known as crystal purification, which is far more efficient than adsorption-based methods. The specific X-ray powder diffraction patterns observed for the Li, Na, and K salts confirm the formation of distinct, ordered crystal structures that are free from the disorder characteristic of amorphous materials. This structural order contributes to the chemical stability of the molecule, preventing degradation pathways that might be accelerated in a disordered, hygroscopic matrix. Furthermore, the use of cation exchange resin in the subsequent step to convert the intermediate to the final gadolinium complex is facilitated by the high purity of the input salt, reducing the load on the resin and extending its operational life. This integrated approach to impurity management ensures that the final Gadoxetate Disodium product achieves purity levels of 99% or higher, meeting the rigorous demands of the pharmaceutical industry. The mechanistic robustness of this process provides a solid foundation for regulatory filings, as the consistency of the crystal form can be reliably demonstrated through standard analytical techniques.
How to Synthesize EOB-DTPA Intermediate Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions and crystallization parameters to fully realize the benefits of the patented technology. The process begins with the dissolution of the protected precursor in an organic solvent, followed by the addition of the alkali hydroxide and heating to reflux for a defined period to ensure complete deprotection. Detailed standardized synthesis steps are provided below to guide process engineers in replicating this high-efficiency pathway.
- Dissolve the protected precursor Compound II in an organic alcohol solvent such as methanol or ethanol at room temperature.
- Add the appropriate alkali metal hydroxide (LiOH, NaOH, or KOH) and heat the mixture to reflux for 5 to 8 hours to ensure complete deprotection.
- Cool the reaction mixture to induce crystallization, filter the solid product, and dry to obtain the high-purity crystalline metal salt intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel crystallization technology offers profound commercial benefits that extend beyond mere technical superiority, directly impacting the bottom line and operational resilience of pharmaceutical manufacturers. By eliminating the need for chromatographic purification, the process drastically reduces the consumption of expensive solvents and stationary phases, leading to substantial cost savings in raw material procurement and waste disposal. The simplified workflow also shortens the overall production cycle time, allowing facilities to increase throughput and respond more agilely to market fluctuations without the need for significant capital investment in new equipment. For supply chain heads, the enhanced stability of the crystalline intermediate mitigates the risks associated with material degradation during storage and transportation, ensuring that inventory remains viable for longer periods. This reliability is crucial for maintaining continuous supply to downstream customers, particularly in the highly regulated medical imaging sector where interruptions can have significant clinical consequences. The ability to source high-purity intermediates from a reliable Gadoxetate Disodium intermediate supplier who utilizes this technology provides a competitive edge in terms of both cost and quality assurance.
- Cost Reduction in Manufacturing: The elimination of chromatographic separation steps removes a major cost driver from the manufacturing process, as it avoids the high expenses associated with silica gel, resins, and large volumes of organic solvents. This simplification allows for a more streamlined operation where labor and utility costs are significantly reduced due to the shorter processing times and fewer unit operations required. Additionally, the high yield of the reaction minimizes the loss of valuable starting materials, further enhancing the economic efficiency of the production line. The qualitative reduction in waste generation also contributes to lower environmental compliance costs, making the process more sustainable and economically viable in the long term. These factors combine to create a manufacturing profile that is highly attractive for cost-sensitive markets while maintaining premium quality standards.
- Enhanced Supply Chain Reliability: The physical stability of the crystalline intermediate ensures that the material can be stored and transported under standard conditions without the risk of liquefaction or degradation, which simplifies logistics and reduces the need for specialized climate-controlled shipping. This robustness allows for the maintenance of larger safety stocks without the fear of inventory spoilage, providing a buffer against supply chain disruptions and demand spikes. The consistent quality of the intermediate also reduces the likelihood of batch rejections during quality control testing, ensuring a smoother flow of materials through the supply chain. For procurement managers, this reliability translates to reduced lead time for high-purity pharmaceutical intermediates and greater confidence in meeting delivery commitments to global clients. The ability to scale this process easily further supports supply continuity, as production capacity can be increased without encountering the bottlenecks typical of chromatography-dependent methods.
- Scalability and Environmental Compliance: The use of common alcohol solvents and simple reflux conditions makes this process highly scalable from laboratory to industrial production volumes without the need for complex engineering modifications. The reduction in solvent usage and waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations across different jurisdictions. The solid nature of the product simplifies handling and packaging, reducing the risk of spills and exposure during manufacturing operations. This environmental and operational efficiency positions the technology as a sustainable choice for long-term production, appealing to stakeholders who prioritize corporate social responsibility and regulatory adherence. The scalability ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly to meet global demand for MRI contrast agents.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial implications of this intermediate synthesis technology, providing clarity for potential partners and stakeholders. These answers are derived directly from the patent data and technical analysis to ensure accuracy and relevance to your procurement decisions.
Q: Why is the crystalline form of the EOB-DTPA intermediate superior to the amorphous form?
A: The crystalline form offers significantly improved stability, preventing moisture absorption and viscosity issues common in amorphous salts, which ensures consistent quality during long-term storage and transport.
Q: Does this synthesis method require chromatographic purification?
A: No, the novel process eliminates the need for expensive and time-consuming chromatographic separation by utilizing controlled crystallization to remove impurities effectively.
Q: What is the purity level achievable with this intermediate synthesis route?
A: The process consistently yields intermediate compounds with purity exceeding 98%, which facilitates the production of final Gadoxetate Disodium with purity greater than 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gadoxetate Disodium Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced technologies like the crystalline intermediate synthesis described in CN103420862A to deliver exceptional value to our global partners. Our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensures that we can meet your volume requirements with consistent quality and reliability. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of Gadoxetate Disodium intermediate meets the highest industry standards. Our team of experts is dedicated to optimizing every step of the synthesis process, from raw material selection to final packaging, to ensure maximum efficiency and cost-effectiveness for our clients. By partnering with us, you gain access to a supply chain that is robust, compliant, and capable of supporting your long-term strategic goals in the medical imaging sector.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project needs and drive value for your organization. Request a Customized Cost-Saving Analysis to understand the potential economic benefits of switching to our optimized synthesis route. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and transparency. Let us help you secure a stable supply of high-quality intermediates for your MRI contrast agent production.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
