Advanced Low-Temperature Synthesis of Thiotepa for High-Purity Pharmaceutical Intermediates
Advanced Low-Temperature Synthesis of Thiotepa for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust manufacturing processes for critical oncology intermediates, and the synthesis of Thiotepa (CAS 52-24-4) represents a significant area of innovation. According to patent CN108484669B, a novel preparation method has been developed that addresses longstanding challenges in yield, purity, and environmental safety. This technology leverages a precise low-temperature control strategy, maintaining the reaction environment between 15°C and 17°C, coupled with a sophisticated acid scavenging protocol. By optimizing the molar ratios of ethyleneimine, thiophosphoryl halides, and a dual-component acid binding agent, the process achieves exceptional conversion rates. The breakthrough lies not just in the reagents, but in the temporal control of their addition, ensuring that the highly reactive aziridine rings remain intact while effectively neutralizing acidic byproducts. This approach offers a reliable pharmaceutical intermediate supplier with a pathway to produce high-purity alkylating agents suitable for stringent regulatory environments.
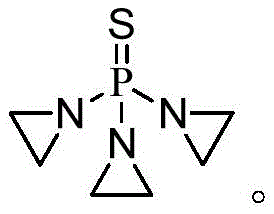
Furthermore, the method utilizes a specific mixed solvent system comprising tetrahydrofuran and cyclohexane, which plays a pivotal role in the downstream processing and purification stages. The interaction between these solvents modulates the solubility of the reaction salts, allowing for efficient separation without the need for complex extraction procedures. This results in a final product with purity levels reaching up to 99.3%, a critical specification for API manufacturing where impurity profiles must be tightly controlled. The process is designed for industrial scalability, utilizing cheap and easily obtainable raw materials while minimizing the generation of hazardous three wastes. For procurement and supply chain leaders, this translates to a more sustainable and cost-effective sourcing strategy for this vital chemotherapy agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Thiotepa has been plagued by significant safety and efficiency drawbacks that hinder large-scale production. Early methods, such as those disclosed in US2670347, relied heavily on anhydrous benzene as a solvent, a known carcinogen that poses severe health risks and requires expensive containment and disposal protocols. These legacy processes typically operated at sub-zero temperatures, demanding high energy consumption for cooling, yet still suffered from low yields and difficult product isolation. The formation of triethylamine hydrochloride salts in benzene often led to cumbersome filtration steps, and the subsequent recrystallization from petroleum ether was inefficient. Moreover, alternative aqueous routes described in literature, such as US4928199, attempted to bypass organic solvents but resulted in extremely poor yields, often less than 10%, due to the hydrolysis of the intermediate aziridine in water. These conventional limitations created a bottleneck for cost reduction in API manufacturing, forcing producers to accept low throughput and high waste generation.
The Novel Approach
In stark contrast, the methodology outlined in CN108484669B introduces a paradigm shift by replacing hazardous solvents with a safer, optimized mixed solvent system and refining the reaction kinetics through precise dosing. Instead of relying on a single solvent or water, the new process employs a volume ratio of 11-12:5 of tetrahydrofuran to cyclohexane. This specific combination ensures that while the reaction proceeds rapidly in the polar THF phase, the resulting amine salts precipitate out due to the presence of cyclohexane, simplifying the workup. Furthermore, the reaction temperature is maintained at a mild 15-17°C, which is significantly easier to control industrially compared to cryogenic conditions. The introduction of a composite acid scavenger, consisting of potassium carbonate and triethylamine, allows for fine-tuned neutralization that prevents the degradation of the sensitive product. This holistic redesign of the synthetic route eliminates the use of benzene entirely, drastically simplifies the operation, and boosts the yield to over 90%, representing a substantial improvement over prior art.
Mechanistic Insights into Low-Temperature Nucleophilic Substitution
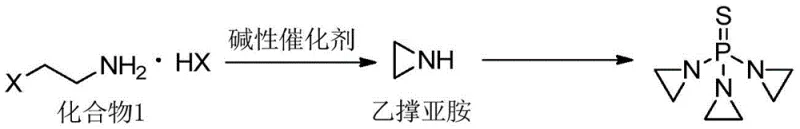
The core chemical transformation involves the nucleophilic attack of ethyleneimine on the phosphorus center of thiophosphoryl chloride, a reaction that is highly exothermic and sensitive to acidic conditions. The mechanism relies on the generation of the aziridine nucleophile, which can be synthesized in situ from precursors like 2-bromoethylamine under basic conditions, as illustrated in related schemes. However, the critical innovation in this patent is the management of the hydrogen chloride byproduct generated during the substitution. If the HCl is not neutralized immediately, it can protonate the unreacted ethyleneimine or the product Thiotepa, leading to ring-opening polymerization or hydrolysis. Conversely, if the base is too strong or present in excess at the start, it can catalyze the decomposition of the reactants. The patent data reveals that maintaining a specific molar excess of ethyleneimine (ratio of 3.01:1 relative to phosphorus reagent) drives the equilibrium forward, while the delayed addition of the scavenger prevents premature side reactions.
Impurity control is achieved through the unique solubility dynamics of the THF-cyclohexane mixture. In pure THF, the triethylamine hydrochloride salt remains dissolved, contaminating the filtrate and requiring extensive washing. By introducing cyclohexane, the polarity of the medium is lowered, causing the salt to precipitate as a solid cake that can be easily filtered off before solvent removal. This physical separation mechanism is far more efficient than chemical quenching and washing. Additionally, the purification step involves dissolving the crude residue in ethyl acetate and inducing crystallization with cyclohexane at temperatures between -7.5°C and -6.5°C. This low-temperature crystallization selectively excludes structurally similar impurities, ensuring the final solid meets the stringent purity specifications required for oncology drugs. The result is a clean impurity profile that minimizes the burden on downstream analytical testing and regulatory filing.
How to Synthesize Thiotepa Efficiently
The synthesis of Thiotepa via this advanced protocol requires strict adherence to temperature and dosing parameters to replicate the high yields reported in the patent literature. The process begins with the preparation of the reaction vessel, where ethyleneimine and the organic solvent mixture are cooled to the critical 15-17°C range before any reagents are introduced. The thiophosphoryl chloride is then added dropwise, but the key operational step occurs when exactly 49% of the theoretical amount has been delivered; at this precise moment, the acid scavenger solution is introduced at a rate roughly 3.06 times that of the phosphorus reagent. This synchronized addition ensures that the reaction mixture never becomes too acidic nor too basic, preserving the integrity of the aziridine rings throughout the 30-45 minute reaction window. Detailed standardized synthesis steps see the guide below.
- Cool ethyleneimine and a mixed organic solvent (THF: Cyclohexane) to a strict temperature range of 15-17°C.
- Dropwise add thiophosphoryl chloride solution; when 49% of the reagent is added, begin dropwise addition of the potassium carbonate/triethylamine acid scavenger mixture.
- Filter the reaction mixture, remove solvents under reduced pressure, and purify the residue via crystallization in ethyl acetate and cyclohexane at -7°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis method offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of benzene, a regulated carcinogen, removes the need for specialized hazardous waste disposal contracts and reduces the regulatory compliance burden associated with worker safety. This shift to safer solvents like THF and cyclohexane, which are commodity chemicals with stable supply chains, enhances the reliability of raw material sourcing. Furthermore, the simplified workup procedure, which relies on precipitation and filtration rather than complex extractions, reduces the cycle time per batch. This efficiency gain allows manufacturing facilities to increase throughput without expanding their physical footprint, effectively lowering the cost of goods sold through better asset utilization. The high purity of the crude product also reduces the load on purification units, saving energy and solvent consumption in the final crystallization steps.
- Cost Reduction in Manufacturing: The process achieves a dramatic reduction in production costs by eliminating the need for expensive cryogenic cooling and hazardous solvent handling. By operating at mild temperatures of 15-17°C, the energy demand for refrigeration is significantly lower compared to traditional sub-zero methods. Additionally, the high yield of over 90% means that less raw material is wasted, directly improving the material cost efficiency. The use of inexpensive acid scavengers like potassium carbonate further contributes to margin expansion, making the overall process economically superior to legacy routes that suffer from yields below 10% or require costly recrystallization cycles.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the method relies on widely available bulk chemicals rather than specialized or restricted reagents. The robustness of the reaction conditions, specifically the tolerance for slight variations within the 15-17°C window, ensures consistent batch-to-batch quality, reducing the risk of production failures or out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream API manufacturers who cannot afford interruptions in their chemotherapy drug production lines. The simplified process flow also reduces the dependency on highly skilled operators, mitigating labor-related supply risks.
- Scalability and Environmental Compliance: The method is inherently designed for commercial scale-up of complex pharmaceutical intermediates, with a straightforward transition from laboratory to pilot and full production scales. The reduction in three wastes, particularly the avoidance of benzene-contaminated waste streams, aligns with increasingly strict global environmental regulations. This compliance advantage future-proofs the supply chain against tightening environmental laws, avoiding potential shutdowns or fines. The ability to recycle solvents like cyclohexane and ethyl acetate further enhances the sustainability profile, appealing to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Thiotepa synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational nuances that drive the process success. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer or licensing. The responses cover critical aspects such as reagent timing, solvent selection, and purification parameters that define the quality of the final product.
Q: Why is the acid scavenger added only after 49% of the thiophosphoryl halide?
A: Adding the scavenger too early can lead to hydrolysis of the sensitive aziridine ring due to excessive basicity, while adding it too late allows HCl accumulation to degrade the product. The 49% threshold balances reaction rate with stability.
Q: What is the advantage of using a THF and Cyclohexane mixed solvent system?
A: While THF alone gives high yields, the resulting amine salts remain soluble, complicating purification. Adding cyclohexane reduces the solubility of these salts, facilitating easier filtration and significantly improving final product purity.
Q: How does this method compare to traditional benzene-based synthesis?
A: Traditional methods often utilize carcinogenic benzene and suffer from low yields and difficult purification. This novel method eliminates benzene, operates at mild temperatures (15-17°C), and achieves yields exceeding 90% with purity up to 99.3%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiotepa Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and manufacture of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial reality is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By leveraging advanced synthesis methods like the one described in CN108484669B, we can offer clients a supply of Thiotepa that meets the highest international standards for safety and efficacy, providing a solid foundation for your drug development pipeline.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your specific project requirements. Whether you need specific COA data to support your regulatory filings or route feasibility assessments to evaluate cost structures, we are prepared to provide comprehensive support. Please contact us today to request a Customized Cost-Saving Analysis tailored to your volume needs, and let us demonstrate how our technical expertise can drive value and reliability in your supply chain.
