Scalable Chemical Synthesis of Laetispicine: Technical Breakthroughs for Commercial Production
The pharmaceutical industry constantly seeks robust methods to access bioactive natural products that exhibit potent therapeutic potential but suffer from scarce natural availability. Patent CN101798297B presents a groundbreaking chemical synthesis method for Laetispicine, a natural product derived from Piper laetispicum known for its significant antidepressant, sedative, and analgesic properties. Historically, the reliance on plant extraction has been a major bottleneck, with natural content levels reported as low as 0.03%, rendering large-scale clinical development economically unfeasible. This patent discloses a comprehensive fourteen-step convergent synthesis that effectively bypasses these agricultural limitations. By utilizing piperonal and pentanediol as primary feedstocks, the methodology establishes a reproducible pathway to generate high-purity Laetispicine. For R&D directors and procurement specialists, this represents a critical shift from variable botanical sourcing to controlled chemical manufacturing, ensuring consistent quality and supply continuity for antidepressant drug development programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional acquisition of Laetispicine relies exclusively on the extraction from the roots and stems of Piper laetispicum, a process fraught with inherent inefficiencies and supply chain vulnerabilities. The natural abundance of the target molecule is exceptionally low, necessitating the processing of massive quantities of biomass to isolate minute amounts of the active ingredient. This not only drives up the cost of goods significantly but also introduces batch-to-batch variability due to seasonal, geographical, and climatic factors affecting plant growth. Furthermore, the extraction process often co-elutes structurally similar impurities that are difficult to separate, complicating downstream purification and potentially impacting the safety profile of the final drug substance. From a sustainability perspective, the heavy reliance on wild or cultivated plant resources poses ecological risks and cannot support the volumes required for global pharmaceutical distribution. Consequently, the conventional extraction method fails to meet the rigorous demands of modern Good Manufacturing Practice (GMP) standards for commercial API production.
The Novel Approach
The synthetic route detailed in the patent offers a transformative solution by employing a convergent strategy that assembles the complex molecular architecture from simple, commercially available building blocks. Instead of harvesting the molecule from nature, the process constructs the carbon skeleton through precise organic transformations, specifically leveraging a sulfone tetrazole-mediated coupling and a Wittig-Horner olefination. This approach allows for the independent optimization of two key fragments: the aromatic core containing the methylenedioxy motif and the aliphatic side chain bearing the polyene system. By separating the synthesis into these distinct modules, chemists can maximize yields at each stage and troubleshoot issues without compromising the entire batch. The final assembly involves a highly stereoselective condensation that establishes the crucial double bond geometry, ensuring the synthetic product matches the spectroscopic data of the natural isolate. This level of control translates directly into a reliable pharmaceutical intermediates supplier capability, decoupling production from agricultural cycles.
Mechanistic Insights into Convergent Olefination and Functionalization
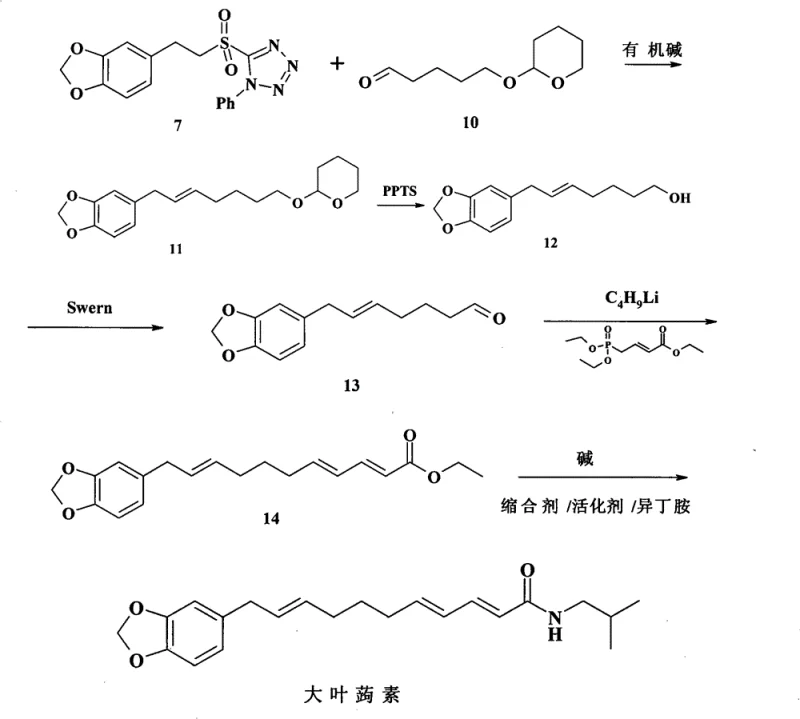
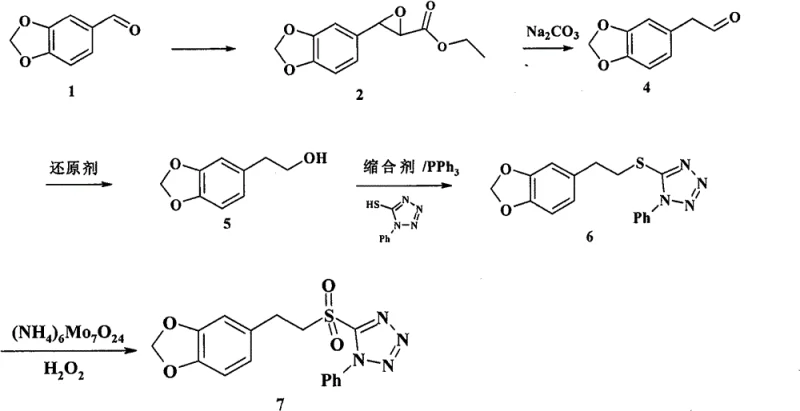
The heart of this synthesis lies in the strategic construction of the polyene chain and the installation of the amide functionality. The process initiates with the preparation of a specialized sulfone tetrazole intermediate (Compound 7), which serves as a potent nucleophile for carbon-carbon bond formation. This fragment is synthesized from piperonal through a Darzens condensation to form an epoxy ester, followed by hydrolysis and decarboxylation to yield the corresponding acetaldehyde. Subsequent reduction and Mitsunobu reaction with 1-phenyl-5-mercaptotetrazole install the sulfur-containing heterocycle, which is finally oxidized to the sulfone. This sulfone group is critical as it activates the adjacent protons for deprotonation by strong bases like LDA or LHMDS, facilitating the nucleophilic attack on the side-chain aldehyde. The resulting elimination of the tetrazole sulfinate group drives the formation of the trans-alkene with high fidelity, a key feature for maintaining the biological activity of the final molecule.
Following the initial coupling, the synthetic sequence proceeds through a series of meticulous functional group manipulations designed to extend the carbon chain and introduce the final amide moiety. The coupled intermediate undergoes acidic deprotection to reveal a primary alcohol, which is then subjected to Swern oxidation conditions to generate the corresponding aldehyde without over-oxidation to the carboxylic acid. This aldehyde serves as the substrate for a Wittig-Horner reaction with a phosphonate ester, extending the conjugated system to form the trienoic acid ethyl ester. The use of phosphonate chemistry here is advantageous for controlling the E/Z geometry of the newly formed double bonds. Finally, hydrolysis of the ester followed by amidation with isobutylamine using carbodiimide coupling reagents yields the target Laetispicine. Throughout this sequence, impurity control is managed through careful selection of reagents and purification steps like column chromatography, ensuring that the final product meets stringent purity specifications required for clinical applications.
How to Synthesize Laetispicine Efficiently
Executing this synthesis requires strict adherence to anhydrous conditions and temperature controls, particularly during the organometallic and oxidation steps. The process is divided into the preparation of the two main fragments followed by their convergence. The core fragment synthesis involves handling reactive intermediates like epoxy esters and sulfones, while the side chain requires precise protection group chemistry using tetrahydropyranyl (THP) ethers. The coupling step is sensitive to base strength and stoichiometry, requiring optimization to minimize side reactions. For a detailed breakdown of the specific reaction conditions, molar ratios, and workup procedures necessary to replicate this high-yielding pathway, please refer to the standardized technical guide below.
- Prepare the core sulfone tetrazole intermediate (Compound 7) from piperonal via Darzens condensation, decarboxylation, and Mitsunobu reaction.
- Synthesize the side chain aldehyde (Compound 10) from pentanediol using THP protection and Swern oxidation.
- Couple the core and side chain via base-mediated condensation, followed by deprotection, oxidation, Wittig-Horner reaction, and final amidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from extraction to total synthesis offers profound strategic benefits that extend beyond mere technical feasibility. The primary advantage is the stabilization of raw material costs and availability. By shifting the source of Laetispicine from a scarce botanical resource to commodity chemicals like piperonal and pentanediol, manufacturers eliminate the volatility associated with crop yields and harvest seasons. This ensures a consistent flow of materials, reducing the risk of production stoppages due to raw material shortages. Furthermore, the synthetic route allows for production scaling in standard chemical reactors, enabling the manufacture of kilogram to ton quantities that are impossible to achieve through extraction. This scalability is essential for meeting the demands of clinical trials and potential commercial launch, providing a secure foundation for long-term supply agreements.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the expensive and labor-intensive processes associated with plant harvesting, drying, and solvent extraction. While the chemical synthesis involves multiple steps, the use of inexpensive starting materials and the ability to optimize yields at each stage leads to a significantly lower cost of goods sold compared to natural extraction. Additionally, the convergent nature of the synthesis allows for parallel processing of intermediates, improving overall throughput and equipment utilization rates. The removal of complex purification steps needed to separate natural analogs further contributes to operational efficiency and cost savings.
- Enhanced Supply Chain Reliability: Relying on a chemical synthesis route diversifies the supply chain, reducing dependency on single-source agricultural suppliers who may be affected by geopolitical or environmental disruptions. The starting materials are widely produced fine chemicals with established global supply networks, ensuring that procurement teams can source inputs from multiple vendors to mitigate risk. This reliability is crucial for maintaining continuous manufacturing schedules and meeting delivery commitments to downstream pharmaceutical partners. It also simplifies inventory management, as synthetic intermediates typically have longer shelf lives and more stable storage requirements than botanical extracts.
- Scalability and Environmental Compliance: Chemical synthesis offers superior control over waste streams and environmental impact compared to large-scale biomass processing. The process can be engineered to minimize solvent usage and incorporate recycling loops for reagents, aligning with green chemistry principles. Scaling up from laboratory to pilot and commercial plants is straightforward using standard unit operations such as reactors, distillation columns, and crystallizers. This ease of scale-up reduces the time and capital investment required to bring the product to market, allowing companies to respond quickly to increasing demand without the lead times associated with expanding agricultural cultivation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Laetispicine via this patented method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of the synthetic route. Understanding these details is essential for stakeholders evaluating the potential for integrating this intermediate into their development pipelines.
Q: What is the primary advantage of this synthetic route over natural extraction?
A: Natural extraction yields are extremely low (approximately 0.03%), making it unsustainable for commercial demand. This chemical synthesis provides a reliable, scalable alternative independent of agricultural variables.
Q: How is stereochemistry controlled in the polyene chain?
A: The route utilizes specific olefination strategies, including a sulfone-mediated coupling and a Wittig-Horner reaction, which favor the formation of the thermodynamically stable E-isomers required for biological activity.
Q: Are the starting materials commercially available?
A: Yes, the synthesis begins with commodity chemicals such as piperonal and 1,5-pentanediol, ensuring robust supply chain continuity and cost stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Laetispicine Supplier
The successful implementation of this complex fourteen-step synthesis requires a partner with deep expertise in multi-step organic synthesis and process optimization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Laetispicine meets the highest standards for pharmaceutical research. We understand the critical nature of antidepressant development and are committed to providing a stable, high-quality supply of this valuable intermediate to support your clinical and commercial needs.
We invite you to collaborate with us to optimize your supply chain and reduce costs associated with natural product sourcing. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing capabilities can accelerate your project timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
