Revolutionizing Apalutamide Production: A Deep Dive into Novel Copper-Catalyzed One-Pot Synthesis
Revolutionizing Apalutamide Production: A Deep Dive into Novel Copper-Catalyzed One-Pot Synthesis
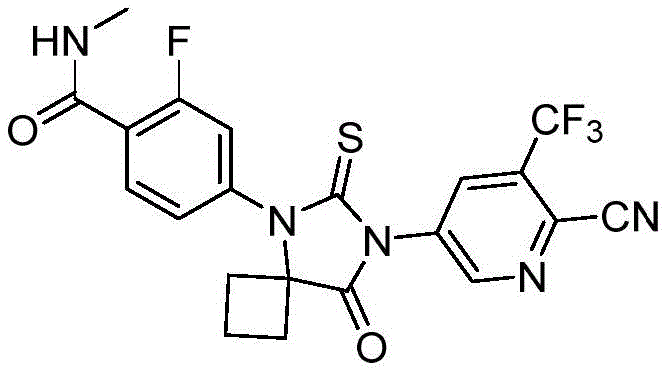
The pharmaceutical industry is constantly seeking more efficient, safer, and cost-effective pathways for the production of critical active pharmaceutical ingredients (APIs) and their intermediates. A recent breakthrough detailed in Chinese Patent CN112194633B introduces a transformative approach to synthesizing Apalutamide, a second-generation non-steroidal androgen receptor inhibitor marketed as Erleada®. This patent discloses a novel copper-catalyzed one-pot method that utilizes 6-thio-5,7-diazaspiro[3.4]octan-8-one as a key substrate to directly obtain Boc-Apalutamide. This technological advancement addresses long-standing challenges in the manufacturing of complex heterocyclic compounds, offering a streamlined alternative to traditional multi-step processes that often rely on hazardous reagents and energy-intensive conditions. For global procurement and R&D teams, understanding the nuances of this synthesis route is essential for securing a reliable API intermediate supplier capable of meeting stringent quality and volume demands.
The significance of this patent lies in its ability to simplify the synthetic landscape of Apalutamide. Traditionally, the construction of the spiro-hydantoin core and its subsequent coupling with the pyridine and benzamide fragments have been fraught with difficulties, including low yields, harsh reaction conditions, and the use of toxic materials. The disclosed method overcomes these hurdles by employing a robust copper catalytic system that facilitates the formation of critical carbon-nitrogen bonds under relatively controlled thermal conditions. By integrating the synthesis of the Boc-protected intermediate into a cohesive one-pot sequence, the process not only enhances overall yield but also minimizes the generation of waste, aligning with modern green chemistry principles. This makes it an attractive candidate for the commercial scale-up of complex pharmaceutical intermediates, ensuring a stable supply chain for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, several synthetic routes for Apalutamide were documented, each carrying significant drawbacks that hindered efficient large-scale production. For instance, early methods reported by the University of California involved the use of highly toxic sodium cyanide and irritating thiophosgene, coupled with microwave heating, which poses severe safety risks and scalability issues for industrial reactors. Other approaches, such as those by Aragon Pharmaceuticals, utilized complex sulfur sources and required lengthy reaction sequences involving multiple isolation steps, leading to increased production cycles and higher operational costs. Furthermore, some routes relied on unstable isothiocyanate intermediates that are difficult to store and handle, creating bottlenecks in the supply chain and compromising batch-to-batch consistency. These conventional methods often resulted in lower overall yields and required extensive purification efforts to remove heavy metal residues or toxic byproducts, thereby inflating the cost of goods sold.
The Novel Approach
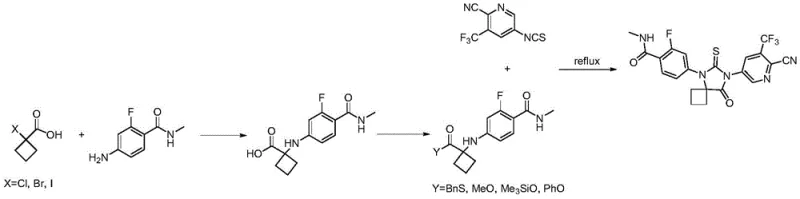
In stark contrast, the novel approach described in CN112194633B leverages a copper-catalyzed one-pot strategy that dramatically simplifies the manufacturing workflow. By starting with the readily accessible 6-thio-5,7-diazaspiro[3.4]octan-8-one, the process enables the direct construction of the target Boc-Apalutamide molecule through sequential coupling reactions within a single vessel. This methodology eliminates the need for isolating unstable intermediates, thereby reducing material loss and exposure to potentially hazardous substances. The use of standard heating methods instead of microwave irradiation ensures that the process is fully compatible with existing industrial infrastructure, facilitating easier technology transfer and scale-up. Moreover, the selection of catalysts such as cuprous iodide or active copper powder, combined with common bases like potassium carbonate and sodium hydride, provides a cost-effective and chemically robust environment for the reaction to proceed with high selectivity and efficiency.
Mechanistic Insights into Copper-Catalyzed Cyclization and Coupling
The core of this innovative synthesis lies in the precise orchestration of copper-catalyzed coupling reactions that forge the molecular architecture of Apalutamide. The mechanism begins with the activation of the spiro-hydantoin nitrogen by the copper catalyst, which facilitates a nucleophilic attack on the aryl bromide moiety of the N-(4-bromo-2-fluorobenzoyl) tert-butyl carbamate. This step is critical for establishing the connection between the spiro core and the fluorobenzamide fragment, a linkage that is essential for the biological activity of the final drug. The reaction conditions, typically maintained between 165-170°C in a polar aprotic solvent like N-methylpyrrolidone (NMP), ensure sufficient energy for the catalytic cycle to turnover effectively while minimizing side reactions. The presence of ligands or additives such as potassium iodide and 4-dimethylaminopyridine further stabilizes the catalytic species, enhancing the rate of the coupling and improving the overall conversion.
Following the initial coupling, the reaction mixture undergoes a second transformation where the remaining nitrogen of the hydantoin ring is alkylated with 2-cyano-3-trifluoromethyl-5-bromopyridine. This step requires a stronger base, such as sodium hydride, to deprotonate the nitrogen and generate a reactive nucleophile capable of displacing the bromide on the pyridine ring. The temperature is subsequently raised to 175-180°C to drive this second coupling to completion. The beauty of this one-pot design is that the solvent and catalyst system from the first step can often be carried over or slightly adjusted, avoiding the need for intermediate workups that would otherwise introduce impurities or reduce yield. This seamless transition between reaction stages is key to achieving the reported high purity of over 95% and yields exceeding 85% for the Boc-protected intermediate, demonstrating superior control over the impurity profile compared to stepwise methods.
How to Synthesize Boc-Apalutamide Efficiently
The practical execution of this synthesis involves a carefully controlled sequence of reagent additions and temperature ramps to maximize efficiency and safety. The process begins with the preparation of the spiro-hydantoin precursor, followed by its introduction into the coupling reactor along with the aryl bromide and catalyst system. Detailed operational parameters, including specific molar ratios, stirring rates, and quenching procedures, are critical for reproducing the high yields observed in the patent examples. For R&D teams looking to implement this technology, adhering to the specified thermal profiles and base addition protocols is essential to prevent exothermic runaways and ensure consistent product quality. The following guide outlines the standardized synthesis steps derived from the patent data, providing a roadmap for laboratory and pilot-scale production.
- Prepare the spiro-hydantoin core by reacting 1-aminocyclobutanecarboxylic acid with thiourea at 185-195°C.
- Perform a copper-catalyzed coupling of the spiro-hydantoin with N-(4-bromo-2-fluorobenzoyl) tert-butyl carbamate in NMP at 165-170°C.
- Without isolation, add sodium hydride and 2-cyano-3-trifluoromethyl-5-bromopyridine, heating to 175-180°C to complete the one-pot synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost reduction in pharmaceutical manufacturing achieved by streamlining the process. By consolidating multiple reaction steps into a single one-pot operation, the method significantly reduces the consumption of solvents, reagents, and energy, all of which are major cost drivers in API production. Furthermore, the elimination of hazardous reagents like sodium cyanide and thiophosgene lowers the costs associated with specialized waste disposal and safety compliance, contributing to a leaner and more sustainable manufacturing model. The high yield and purity reported in the patent also imply less material loss and reduced need for extensive chromatographic purification, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis drastically cuts down on unit operations, meaning fewer reactors are tied up for shorter periods, which increases asset utilization and lowers overhead costs. The avoidance of expensive and unstable reagents, such as specific isothiocyanates, further stabilizes the raw material budget, protecting against price volatility in the chemical market. Additionally, the simplified downstream processing reduces the load on purification teams and equipment, allowing for faster batch turnover and improved cash flow dynamics for the manufacturing facility.
- Enhanced Supply Chain Reliability: By utilizing robust and commercially available starting materials like 1-aminocyclobutanecarboxylic acid and thiourea, the supply chain becomes less vulnerable to disruptions caused by niche reagent shortages. The process's tolerance for standard industrial heating methods, as opposed to specialized microwave equipment, ensures that production can be easily scaled across different manufacturing sites without requiring capital-intensive retrofitting. This flexibility enhances the resilience of the supply network, ensuring continuous availability of high-purity API intermediates even during periods of high market demand or logistical constraints.
- Scalability and Environmental Compliance: The method's design inherently supports green chemistry initiatives by minimizing waste generation and avoiding the use of persistent toxic substances. This alignment with environmental regulations reduces the regulatory burden on the manufacturer and mitigates the risk of production halts due to compliance issues. The scalability of the copper-catalyzed system allows for a smooth transition from kilogram-scale development to multi-ton commercial production, ensuring that the supply can grow in tandem with the market uptake of the final drug product without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial implications of this patent, we have compiled a set of answers based on the detailed experimental data and process descriptions provided in the documentation. These questions cover critical aspects such as safety improvements, cost drivers, and scalability potential, offering clarity for stakeholders evaluating this technology for their supply chains. Understanding these details is vital for making informed decisions about sourcing strategies and partnership opportunities in the competitive landscape of oncology drug manufacturing.
Q: How does this new copper-catalyzed route improve safety compared to traditional methods?
A: This method eliminates the need for highly toxic sodium cyanide and unstable isothiocyanates used in prior art, significantly enhancing operational safety and environmental compliance.
Q: What represents the key cost-saving advantage of this one-pot strategy?
A: By combining multiple coupling steps into a single reactor vessel without intermediate isolation, the process drastically reduces solvent consumption, labor hours, and purification costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for industrial mass production due to its simple operation, high yield (>85%), and avoidance of difficult-to-scale microwave heating techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Apalutamide Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to meet the evolving needs of the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the innovations presented in CN112194633B and possesses the technical capability to implement this copper-catalyzed one-pot strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of high-quality intermediates. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Apalutamide intermediate meets the highest industry standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this more efficient manufacturing process. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials in your downstream processes. Together, we can drive innovation and efficiency in the production of life-saving medications, ensuring that patients have timely access to essential therapies.
