Advanced Asymmetric Hydrogenation for Scalable Production of S-1,1-Bis(4-Fluorophenyl)propan-2-ol Intermediates
Introduction to Next-Generation Agrochemical Intermediate Synthesis
The global demand for high-performance fungicides continues to drive innovation in the synthesis of critical chiral intermediates. Patent CN112358379A introduces a groundbreaking preparation method for optically pure S-configuration 1,1-bis-(4-fluorophenyl)-2-propanol, a pivotal building block for the novel pyridine amide fungicide florypicoxamid. This technology addresses the longstanding challenges associated with producing high-purity chiral alcohols by leveraging homogeneous asymmetric hydrogenation. Unlike traditional multi-step sequences that rely on stoichiometric chiral auxiliaries or difficult separations, this approach utilizes a highly efficient transition metal catalyst system to directly reduce the corresponding ketone precursor. The result is a process that achieves exceptional optical purity of up to 97% ee and yields ranging from 95% to 98%, setting a new benchmark for efficiency in agrochemical intermediate manufacturing. For R&D and procurement teams alike, this represents a significant opportunity to optimize supply chains for next-generation crop protection agents.
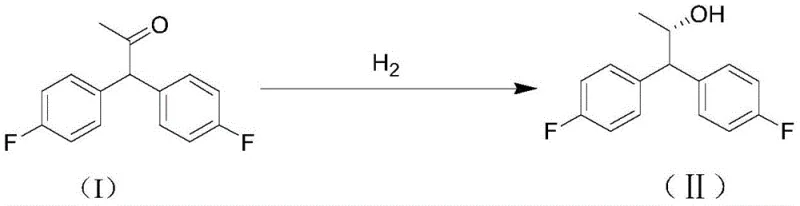
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of S-1,1-bis-(4-fluorophenyl)-2-propanol has relied on cumbersome pathways involving Grignard reagents and protecting group strategies. As illustrated in prior art, these conventional routes typically commence with the addition of a Grignard reagent to a protected chiral ester, followed by hydrosilylation and subsequent deprotection. These methods suffer from severe operational drawbacks, including the requirement for strictly anhydrous and low-temperature conditions to manage the high reactivity and instability of Grignard reagents. Furthermore, the use of hydrosilylation agents introduces additional safety hazards and waste streams, often necessitating complex workup procedures. Perhaps most critically for commercial production, these legacy processes frequently require column chromatography to isolate the desired enantiomer, a step that is notoriously difficult to scale and economically prohibitive for tonnage production. The cumulative effect of these inefficiencies is a process with poor atom economy, high operational costs, and significant environmental burden.
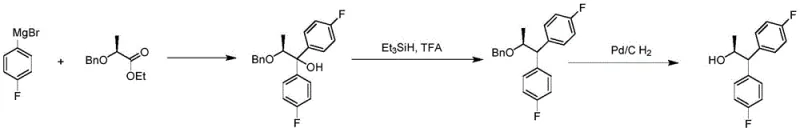
The Novel Approach
In stark contrast, the methodology disclosed in CN112358379A revolutionizes the production landscape by employing a direct asymmetric hydrogenation strategy. This novel approach bypasses the need for protecting groups and stoichiometric chiral reagents entirely. By utilizing a catalytic amount of a chiral transition metal complex, the process converts 1,1-bis-(4-fluorophenyl)acetone directly into the target chiral alcohol with high stereoselectivity. The reaction operates under relatively mild conditions, with temperatures ranging from 20°C to 80°C and hydrogen pressures between 1 MPa and 10 MPa, making it compatible with standard industrial hydrogenation reactors. The elimination of chromatographic purification is a game-changer, as the product can be isolated simply by filtration and solvent removal. This streamlined workflow not only drastically reduces the physical footprint required for manufacturing but also minimizes solvent consumption and waste generation, aligning perfectly with modern green chemistry principles and the needs of a reliable agrochemical intermediate supplier.
Mechanistic Insights into Homogeneous Asymmetric Hydrogenation
The core of this technological breakthrough lies in the sophisticated design of the catalytic system. The reaction proceeds via a homogeneous asymmetric hydrogenation mechanism where a chiral ligand coordinates with a transition metal precursor, such as Iridium, Ruthenium, or Rhodium, to form an active catalytic species. Preferred ligands include tridentate structures like f-amphox, f-amphol, and O-spiroPNN, as well as axially chiral diphosphines like BINAP and SegPhos. These ligands create a highly defined chiral environment around the metal center, which dictates the facial selectivity of the hydrogen addition to the prochiral ketone substrate. The interaction between the catalyst and the substrate ensures that hydrogen is delivered specifically to one face of the carbonyl group, thereby generating the S-configuration with high fidelity. The patent reports turnover numbers (TON) as high as 100,000, indicating that a single catalyst molecule can convert a vast number of substrate molecules before deactivation, which is crucial for minimizing residual metal contamination in the final product.
Impurity control is inherently superior in this catalytic regime compared to stoichiometric methods. In Grignard-based routes, side reactions such as over-addition or elimination can lead to complex impurity profiles that are difficult to separate. In the hydrogenation pathway, the primary side reaction is typically the formation of the undesired R-enantiomer or the achiral alcohol if racemization occurs, both of which are tightly controlled by the ligand architecture and reaction parameters. The use of specific bases, such as potassium tert-butoxide or cesium carbonate, further modulates the activity and selectivity of the catalyst, suppressing unwanted side pathways. The result is a crude product of such high quality that it meets pharmaceutical and agrochemical standards without the need for extensive downstream purification. This mechanistic robustness ensures consistent batch-to-batch quality, a critical factor for maintaining the integrity of the final fungicide active ingredient.
How to Synthesize S-1,1-Bis(4-Fluorophenyl)propan-2-ol Efficiently
Implementing this synthesis requires careful attention to catalyst preparation and reaction conditions to maximize enantioselectivity and yield. The process begins with the in situ or pre-formation of the metal-ligand complex in a suitable organic solvent such as isopropanol or tetrahydrofuran. Once the active catalyst is generated, it is introduced to a mixture of the ketone substrate and a base under an inert atmosphere. The reaction vessel is then pressurized with hydrogen and heated to the optimal temperature range. While the general procedure is straightforward, fine-tuning the molar ratios of ligand to metal and catalyst to substrate is essential for achieving the reported high TON values. For detailed operational parameters and specific embodiment examples, please refer to the standardized synthesis guide below.
- Prepare the catalyst system by mixing a chiral ligand (such as f-amphox or BINAP) with a transition metal precursor (Ir, Ru, or Rh) in an organic solvent under inert atmosphere.
- Combine the ketone substrate (1,1-bis(4-fluorophenyl)acetone) with a base (e.g., potassium tert-butoxide) and the prepared catalyst solution in a high-pressure reactor.
- Charge the reactor with hydrogen gas (1-10 MPa) and maintain the reaction temperature between 20°C and 80°C until conversion is complete, followed by filtration and solvent removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from stoichiometric chiral synthesis to catalytic asymmetric hydrogenation offers profound economic and logistical benefits. The primary driver of value is the drastic simplification of the manufacturing process. By eliminating the need for column chromatography and reducing the number of unit operations, the overall production cycle time is significantly compressed. This efficiency translates directly into lower manufacturing costs, as less labor, energy, and solvent are consumed per kilogram of product. Furthermore, the high atom economy of the hydrogenation reaction means that raw material utilization is optimized, reducing the cost of goods sold (COGS) and minimizing the environmental impact associated with waste disposal. These factors combine to create a more resilient and cost-effective supply chain for high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The transition to a catalytic process fundamentally alters the cost structure of production. Traditional methods relying on stoichiometric chiral reagents or protecting groups incur high material costs and generate substantial waste, driving up disposal expenses. In contrast, the asymmetric hydrogenation method utilizes catalytic amounts of expensive ligands and metals, spreading their cost over a large volume of product due to the high turnover numbers. Additionally, the removal of chromatographic purification steps eliminates the need for large quantities of silica gel and elution solvents, which are significant cost centers in fine chemical manufacturing. This structural change allows for substantial cost savings that can be passed down the supply chain, enhancing competitiveness in the global fungicide market.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses and the reliance on specialized, hard-to-source reagents. The new hydrogenation route relies on commercially available starting materials and standard industrial gases (hydrogen), reducing dependency on niche suppliers. The robustness of the catalyst system, capable of operating effectively across a range of temperatures and pressures, ensures that production is less susceptible to minor fluctuations in process conditions. This reliability minimizes the risk of batch failures and production delays, ensuring a steady flow of intermediates to downstream formulation plants. For supply chain heads, this means a more predictable inventory profile and reduced need for safety stock buffers.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often exposes hidden bottlenecks, particularly regarding heat transfer and mass transfer in heterogeneous mixtures. The homogeneous nature of this hydrogenation reaction facilitates excellent mixing and heat distribution, making the scale-up from kilograms to tons technically straightforward. Moreover, the green chemistry attributes of the process—specifically the reduction in solvent usage and the absence of hazardous hydrosilylation byproducts—simplify regulatory compliance. Meeting increasingly stringent environmental regulations is easier with a cleaner process, reducing the administrative burden and potential fines associated with waste management. This scalability ensures that the technology can meet growing global demand for florypicoxamid without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of S-1,1-bis-(4-fluorophenyl)-2-propanol. These answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your existing supply chain or R&D pipeline.
Q: What represents the primary advantage of the asymmetric hydrogenation method over traditional Grignard routes?
A: The asymmetric hydrogenation method eliminates the need for moisture-sensitive Grignard reagents and hazardous hydrosilylation steps, significantly improving operational safety and atom economy while achieving high enantioselectivity (up to 97% ee) without column chromatography.
Q: Which catalyst systems are most effective for this transformation according to the patent data?
A: The patent highlights the efficacy of Iridium and Ruthenium complexes paired with chiral ligands such as f-amphox, O-spiroPNN, BINAP, and SegPhos, which demonstrate turnover numbers (TON) reaching up to 100,000.
Q: Is this process suitable for large-scale industrial manufacturing of fungicide intermediates?
A: Yes, the process is designed for industrialization, utilizing standard hydrogenation equipment, common organic solvents, and avoiding complex purification steps like column chromatography, which facilitates cost-effective scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-1,1-Bis(4-Fluorophenyl)propan-2-ol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of next-generation agrochemicals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of S-1,1-bis-(4-fluorophenyl)-2-propanol meets the exacting standards required for fungicide synthesis. Our commitment to process excellence allows us to deliver products with consistent optical purity and minimal impurity profiles, supporting our partners in maintaining their own quality certifications.
We invite you to collaborate with us to leverage this advanced hydrogenation technology for your specific applications. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a competitive advantage in the global agrochemical market.
