Scalable Synthesis of 2-Amino-4,5-Diarylpyrimidines for Neuroprotective Drug Development
The pharmaceutical industry is constantly seeking efficient pathways to access novel heterocyclic scaffolds with potent biological activity, particularly for treating complex neurological disorders. Patent CN101343253B introduces a groundbreaking methodology for the preparation of 2-amino-4,5-diarylpyrimidine compounds, a class of molecules demonstrating significant neuroprotective properties. This technology represents a paradigm shift from traditional multi-step syntheses to a streamlined, one-step condensation reaction utilizing natural isoflavone derivatives and guanidine. For R&D directors and procurement specialists, this patent offers a compelling value proposition: a robust, scalable, and cost-effective route to high-purity intermediates that serve as critical building blocks for next-generation therapeutics targeting conditions such as stroke, Alzheimer's disease, and Parkinson's disease. The ability to derive these complex pyrimidines directly from abundant natural precursors not only simplifies the synthetic challenge but also aligns with modern green chemistry principles, reducing the environmental footprint of pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted pyrimidine rings has relied heavily on the condensation of amidines or guanidines with various carbonyl-containing precursors such as alpha,beta-unsaturated ketones, beta-diketones, or N-aryl acetylene imines. While chemically feasible, these conventional routes often suffer from significant drawbacks that hinder their commercial viability. A primary concern is the lack of regioselectivity, which frequently leads to complex mixtures of isomers that are difficult and expensive to separate, drastically reducing the overall yield of the desired target molecule. Furthermore, many traditional methods require harsh reaction conditions, including extreme temperatures or the use of hazardous reagents, which complicate process safety and increase operational costs. The reliance on precious metal catalysts in some variations introduces additional supply chain risks and necessitates rigorous downstream purification to meet strict residual metal specifications required for pharmaceutical ingredients, adding layers of complexity and expense to the manufacturing process.
The Novel Approach
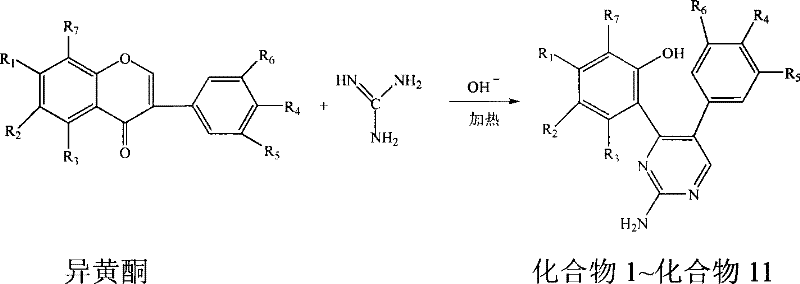
In stark contrast, the methodology disclosed in CN101343253B utilizes naturally occurring isoflavone derivatives as the foundational scaffold, reacting them directly with guanidine under mild basic conditions. This innovative approach eliminates the need for complex precursor synthesis, effectively shortening the production timeline from multiple steps to a single convergent reaction. By leveraging the inherent structural features of isoflavones, the reaction achieves high regioselectivity, ensuring that the pyrimidine ring forms precisely at the desired positions without generating significant isomeric impurities. The process operates at moderate temperatures ranging from 50°C to 100°C and utilizes common, recyclable solvents such as ethanol and methanol, which significantly lowers energy consumption and solvent waste disposal costs. This direct transformation not only enhances the overall atom economy but also simplifies the purification workflow, making it an exceptionally attractive option for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Base-Catalyzed Cyclization
The core of this synthesis lies in a base-catalyzed cyclization mechanism where the guanidine moiety attacks the electrophilic centers of the isoflavone backbone. Under controlled pH conditions of 8 to 10, maintained by the dropwise addition of aqueous sodium or potassium hydroxide, the reaction proceeds through a nucleophilic addition-elimination sequence. The basic environment facilitates the deprotonation of the guanidine, enhancing its nucleophilicity, which allows it to effectively engage with the carbonyl and olefinic functionalities of the isoflavone ring system. This interaction triggers a ring-closing event that constructs the six-membered pyrimidine heterocycle fused with the existing aromatic systems. The elegance of this mechanism is its tolerance for diverse functional groups; substituents such as methoxy, hydroxy, isopropoxy, and even halogen atoms like bromine can remain intact during the transformation, allowing for the generation of a wide library of analogues without the need for extensive protecting group strategies.
Impurity control is meticulously managed through the reaction parameters and subsequent workup. The specific pH range prevents the degradation of sensitive functional groups while ensuring complete conversion of the starting materials. Following the reaction, the mixture is distilled to dryness, and the residue is treated with dilute hydrochloric acid. This acidification step dissolves the basic pyrimidine product while leaving neutral organic impurities behind, which are removed via filtration. Subsequent neutralization precipitates the pure product, and a final recrystallization from aqueous ethanol removes any trace unreacted guanidine or isoflavone. This rigorous purification protocol ensures that the final high-purity pharmaceutical intermediates meet the stringent quality standards required for clinical development, minimizing the risk of toxicological issues associated with process-related impurities.
How to Synthesize 2-Amino-4,5-Diarylpyrimidines Efficiently
The practical implementation of this synthesis is designed for ease of operation in standard chemical reactors, requiring no specialized high-pressure equipment. The process begins by charging the specific isoflavone derivative into a reaction vessel followed by the addition of a solvent, typically ethanol, in an amount 60 to 180 times the weight of the substrate. Guanidine is then introduced at a molar ratio ranging from 1:1 to 1:10 relative to the isoflavone. The reaction mixture is heated to an optimal temperature of 80°C, although the effective range spans from 50°C to 100°C depending on the specific substituents. Throughout the reaction, which lasts between 5 to 50 hours, a 5-12% aqueous alkali solution is continuously added to maintain the system pH between 8 and 10. Detailed standardized synthesis steps follow below.
- Charge isoflavone derivative and solvent (ethanol/methanol) into a reactor, then add guanidine at a molar ratio of 1: 1 to 1:10.
- Heat the mixture to 50-100°C while maintaining pH 8-10 via dropwise addition of aqueous alkali (NaOH/KOH) for 5-50 hours.
- Distill to dryness, dissolve residue in dilute HCl, filter, neutralize to precipitate crude product, and purify via ethanol recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isoflavone-based synthesis route offers substantial strategic benefits beyond mere technical feasibility. The most significant advantage is the drastic simplification of the supply chain for raw materials. Isoflavones are natural products that can be sourced from abundant botanical origins, reducing dependency on scarce or volatile petrochemical-derived precursors. This stability in raw material availability translates directly into enhanced supply chain reliability, ensuring consistent production schedules and mitigating the risk of shortages that often plague complex synthetic routes. Furthermore, the elimination of expensive transition metal catalysts removes a major cost driver and a potential bottleneck related to catalyst sourcing and recovery.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the reduction in unit operations. By consolidating the synthesis into a single step, manufacturers save significantly on labor, energy, and equipment usage time. The absence of heavy metal catalysts means there is no need for costly scavenging resins or complex filtration systems to meet residual metal limits, which further drives down processing costs. Additionally, the use of common solvents like ethanol allows for efficient recovery and recycling systems, minimizing solvent purchase and waste disposal expenses, leading to substantial cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more resilient supply chain. Since the process does not rely on moisture-sensitive reagents or cryogenic temperatures, it is less susceptible to minor fluctuations in plant utilities or environmental conditions. The starting materials are stable and commercially available, reducing lead time for high-purity intermediates. This reliability allows for better inventory planning and reduces the need for excessive safety stock, optimizing working capital for the organization.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method is superior. The atom economy is high, meaning less waste is generated per kilogram of product. The solvents used are generally recognized as safe and are easily biodegradable, simplifying wastewater treatment compliance. The scalability is proven by the straightforward nature of the workup—distillation, acid-base extraction, and crystallization are unit operations that scale linearly from the laboratory to multi-ton commercial production without the engineering challenges associated with exotic chemistries.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 2-amino-4,5-diarylpyrimidine compounds. These insights are derived directly from the experimental data and embodiments detailed in the patent documentation, providing clarity on yield optimization, purity profiles, and therapeutic potential. Understanding these aspects is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing drug development pipelines.
Q: What are the primary advantages of using isoflavones as starting materials for pyrimidine synthesis?
A: Using natural isoflavone derivatives allows for a significantly shorter synthetic route compared to traditional methods involving alpha,beta-unsaturated ketones. This approach offers higher atom economy, eliminates the need for expensive transition metal catalysts, and leverages readily available natural precursors, thereby simplifying supply chain logistics and reducing raw material costs.
Q: How is high purity achieved in the final 2-amino-4,5-diarylpyrimidine products?
A: High purity is ensured through a robust workup procedure involving acid-base extraction followed by recrystallization. The crude product is dissolved in dilute hydrochloric acid to remove insoluble impurities, then neutralized to precipitate the target compound. Final purification is achieved using 50-80% ethanol solutions, which effectively removes unreacted starting materials and side products, yielding crystals suitable for pharmaceutical applications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is highly scalable due to its mild reaction conditions (50-100°C) and the use of common, recyclable solvents like ethanol and methanol. The absence of sensitive catalysts and the simplicity of the isolation procedure (filtration and recrystallization) make it ideal for scaling from kilogram to multi-ton production without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4,5-Diarylpyrimidines Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN101343253B for developing advanced neuroprotective therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 2-amino-4,5-diarylpyrimidines meets the highest international standards for pharmaceutical intermediates. We are committed to delivering quality and consistency, leveraging our deep technical expertise to optimize this specific isoflavone condensation process for your unique requirements.
We invite you to collaborate with us to unlock the full commercial potential of this technology. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how this efficient synthesis can improve your project's economics. Please contact our technical procurement team today to request specific COA data for our available analogues and to discuss comprehensive route feasibility assessments. Let us be your partner in bringing these promising neuroprotective agents from the laboratory to the patients who need them most.
