Advanced Green Synthesis of Chiral 2,2'-Dialkoxy-1,1'-Binaphthyl for Industrial Scale-Up
Advanced Green Synthesis of Chiral 2,2'-Dialkoxy-1,1'-Binaphthyl for Industrial Scale-Up
The pharmaceutical and advanced materials industries are constantly seeking robust, scalable, and environmentally sustainable pathways for producing high-value chiral intermediates. Patent CN102381947B introduces a groundbreaking three-step synthetic methodology for the preparation of chiral 2,2'-dialkoxy-1,1'-binaphthyl derivatives, which serve as critical electron donors in Ziegler-Natta catalyst systems and essential ligands in asymmetric synthesis. This technology represents a paradigm shift from traditional stoichiometric oxidation methods to a catalytic, aqueous-based process that maximizes atom economy and minimizes hazardous waste generation. By leveraging iron-catalyzed oxidative coupling followed by a highly efficient molecular recognition resolution strategy, this route delivers exceptional enantiomeric purity while drastically simplifying downstream processing. For R&D directors and procurement specialists, understanding the mechanistic elegance and operational simplicity of this patent is key to securing a reliable supply chain for next-generation polymerization catalysts and chiral pharmaceutical building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of binaphthyl derivatives has been plagued by significant inefficiencies, particularly regarding the oxidative coupling of 2-naphthol. Traditional protocols often rely on stoichiometric amounts of heavy metal oxidants or harsh reaction conditions that generate substantial quantities of toxic sludge, creating severe environmental compliance burdens and escalating disposal costs. Furthermore, conventional resolution techniques for separating enantiomers frequently suffer from low yields, requiring multiple recrystallization steps that erode overall process economics and extend production lead times. The inability to effectively recycle expensive chiral resolving agents or catalysts in these legacy processes results in a linear consumption of resources that is unsustainable for modern large-scale manufacturing. Additionally, many existing etherification methods operate at elevated temperatures that risk thermal racemization, compromising the optical purity of the final product and necessitating costly re-processing or rejection of batches. These cumulative inefficiencies create a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny.
The Novel Approach
In stark contrast, the methodology disclosed in CN102381947B employs a catalytic oxidative coupling system using Iron(III) chloride and hydrogen peroxide in an aqueous medium, transforming the reaction into a heterogeneous process that allows for facile product isolation via simple filtration. This innovation eliminates the need for organic solvents in the initial coupling step and enables the direct recycling of the iron catalyst from the filtrate, creating a closed-loop system that dramatically reduces chemical consumption. The subsequent resolution step utilizes N-benzyl cinchoninium chloride, a novel resolving agent that forms a precipitable molecular complex with one enantiomer, allowing for the simultaneous recovery of both R-BINOL and S-BINOL with high efficiency. Crucially, the final etherification is conducted under mild thermal conditions, specifically below 127°C, which kinetically suppresses racemization and ensures the integrity of the chiral center. This holistic approach not only enhances yield and purity but also aligns perfectly with green chemistry principles, offering a compelling value proposition for cost-conscious and sustainability-driven organizations.
Mechanistic Insights into FeCl3-Catalyzed Oxidative Coupling and Resolution
The core of this synthetic breakthrough lies in the catalytic cycle of Iron(III) chloride, which acts as a single-electron transfer mediator to facilitate the radical coupling of 2-naphthol. In this aqueous heterogeneous system, hydrogen peroxide serves as the terminal oxidant, regenerating the active iron species while producing water as the sole byproduct, thereby avoiding the accumulation of salt waste typical of stoichiometric oxidants. The reaction proceeds through a phenoxy radical intermediate, which undergoes selective C-C coupling at the 1,1' positions to form the binaphthyl backbone with high regioselectivity. The heterogeneity of the reaction allows the product to precipitate out of the aqueous phase as it forms, driving the equilibrium forward and simplifying the workup to a mere filtration step. This mechanistic design ensures that the catalyst remains dissolved in the mother liquor, ready for immediate reuse in the next batch without extensive purification, as evidenced by the consistent conversion rates observed over multiple cycles.
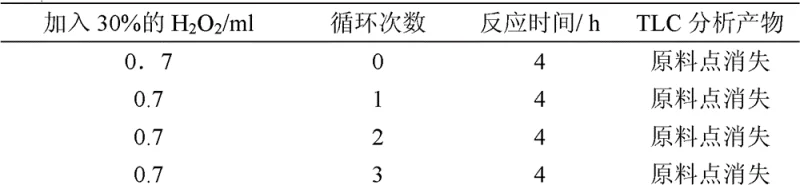
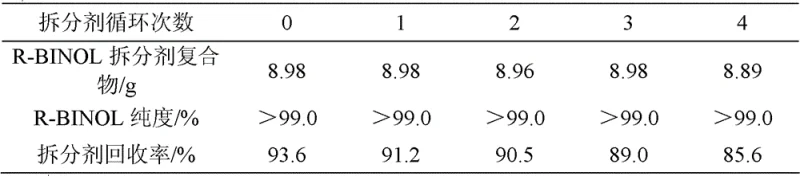
Following the formation of racemic BINOL, the process employs a sophisticated molecular recognition mechanism for chiral resolution. N-benzyl cinchoninium chloride interacts stereoselectively with the hydroxyl groups of the binaphthyl structure, forming a diastereomeric salt complex that exhibits distinct solubility properties in aprotic solvents. The R-enantiomer complex precipitates preferentially, allowing for its physical separation from the S-enantiomer which remains in solution. This dynamic equilibrium can be manipulated by adjusting solvent polarity and temperature, enabling the recovery of the resolving agent from the acid layer after decomposition of the complex. The ability to recover the resolving agent with high efficiency, maintaining its structural integrity over repeated cycles, is a testament to the robustness of this supramolecular interaction. Furthermore, the final etherification step relies on a nucleophilic substitution mechanism where the phenoxide anion attacks the alkylating agent; by strictly controlling the temperature and solvent dielectric constant, the transition state is stabilized in a way that prevents the bond rotation necessary for racemization, thus locking in the optical purity.
How to Synthesize Chiral 2,2'-Dialkoxy-1,1'-Binaphthyl Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize the benefits of the catalytic and resolution steps. The process begins with the preparation of racemic BINOL, followed by the critical resolution step to isolate the desired enantiomers, and concludes with the etherification to yield the final dialkoxy product. Each stage is designed to be operationally simple, utilizing common industrial solvents and reagents to ensure ease of scale-up. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency pathway.
- Perform catalytic oxidative coupling of 2-naphthol using FeCl3 and H2O2 in water to produce racemic BINOL.
- Resolve racemic BINOL using N-benzyl cinchoninium chloride in aprotic solvents to separate R-BINOL and S-BINOL.
- Conduct etherification of chiral BINOL with alkylating agents in polar solvents below 127°C to obtain the final dialkoxy product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates directly into enhanced operational resilience and significant cost optimization. The elimination of stoichiometric heavy metal oxidants and the implementation of catalyst recycling loops drastically reduce the raw material intensity of the process, leading to lower variable costs per kilogram of product. Moreover, the use of water as the primary reaction medium for the coupling step removes the need for expensive, flammable organic solvents in the largest volume stage of the synthesis, thereby lowering safety infrastructure costs and insurance premiums. The high recovery rate of the chiral resolving agent ensures that this high-value reagent does not become a cost bottleneck, stabilizing the bill of materials against market fluctuations. These factors combine to create a manufacturing process that is not only economically superior but also less susceptible to supply disruptions associated with specialty chemical shortages.
- Cost Reduction in Manufacturing: The transition from stoichiometric to catalytic oxidation fundamentally alters the cost structure of BINOL production by removing the expense of disposable oxidants and their associated waste treatment. The ability to recycle the iron catalyst directly from the aqueous filtrate without regeneration means that the effective catalyst cost approaches zero over a long production campaign. Additionally, the high recovery efficiency of the N-benzyl cinchoninium chloride resolving agent ensures that the cost of chirality introduction is minimized, as the same mass of resolving agent can be utilized for multiple batches. This circular economy approach within the reactor significantly lowers the total cost of ownership for the manufacturing process, providing a competitive pricing advantage in the global market for chiral intermediates.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as 2-naphthol, hydrogen peroxide, and iron chloride, the process reduces dependency on exotic or geographically concentrated raw materials that are prone to supply chain volatility. The robustness of the aqueous coupling reaction tolerates variations in feedstock quality better than sensitive organometallic processes, ensuring consistent output even when raw material specifications fluctuate slightly. Furthermore, the simplified workup procedures, which rely on filtration and phase separation rather than complex chromatography or distillation, reduce the equipment footprint and maintenance requirements, minimizing unplanned downtime. This operational simplicity ensures a steady, predictable flow of high-purity product to downstream customers, reinforcing the reliability of the supply chain.
- Scalability and Environmental Compliance: The heterogeneous nature of the oxidative coupling reaction makes it inherently scalable, as heat and mass transfer limitations are easily managed in large-scale reactors compared to homogeneous systems. The absence of halogenated solvents in the primary step and the generation of water as the main byproduct simplify wastewater treatment and ensure compliance with increasingly stringent environmental regulations. The process design inherently minimizes the E-factor (mass of waste per mass of product), making it an attractive option for companies aiming to reduce their carbon footprint and meet sustainability goals. This alignment with green chemistry principles future-proofs the manufacturing asset against regulatory changes and enhances the brand value of the final product in eco-conscious markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios or for qualifying new suppliers.
Q: How is the catalyst recycled in this synthesis method?
A: The Iron(III) chloride catalyst remains in the aqueous filtrate after the oxidative coupling step and can be directly reused for subsequent batches without complex regeneration, significantly reducing waste.
Q: What is the purity level achievable for R-BINOL and S-BINOL?
A: Through the molecular recognition resolution technique using N-benzyl cinchoninium chloride, both R-BINOL and S-BINOL can achieve purity levels exceeding 99.0%, suitable for high-end catalytic applications.
Q: Does the etherification step cause racemization?
A: No, by strictly controlling the reaction temperature below 127°C, specifically optimizing between 20°C and 60°C, the process prevents racemization, ensuring the optical purity of the final dialkoxy-binaphthyl product is maintained.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Binaphthyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of advanced catalysts and pharmaceutical agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of R-BINOL, S-BINOL, or their derivatives meets the exacting standards required for Ziegler-Natta catalyst preparation and asymmetric synthesis. Our commitment to process excellence means we can deliver the cost and quality benefits of this patented green synthesis method to our global partners reliably.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can enhance your supply chain security and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to our catalytically produced intermediates impacts your bottom line. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique volume and purity requirements, ensuring a partnership built on transparency and technical superiority.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
