Advanced Titanium-Catalyzed Synthesis of Cyclopentenones for Scalable Pharmaceutical Intermediate Production
Advanced Titanium-Catalyzed Synthesis of Cyclopentenones for Scalable Pharmaceutical Intermediate Production
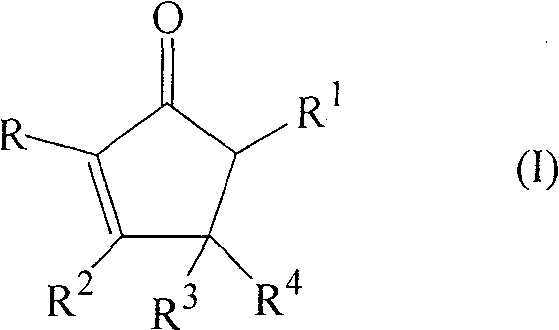
The landscape of organic synthesis for cyclic enones has been significantly advanced by the innovations detailed in patent CN100582072C, which introduces a robust single-step process for preparing substituted 2-cyclopenten-1-ones. This technology represents a pivotal shift from traditional high-energy methodologies to a more sustainable, titanium-catalyzed approach that operates under remarkably mild conditions. For R&D directors and process chemists seeking reliable pathways for complex ring systems, this patent offers a compelling alternative to legacy zirconium-based protocols. The core innovation lies in the specific combination of a metal complex, typically a titanium alkoxide halide, paired with a crucial co-ingredient such as a carboxylic acid anhydride or an anhydrous salt. This synergistic catalytic system enables the direct cyclization of substituted enones with aldehydes, bypassing the severe thermal degradation often associated with earlier methods. By leveraging this chemistry, manufacturers can achieve higher purity profiles and more consistent batch-to-batch reproducibility, which are critical metrics for any reliable pharmaceutical intermediate supplier aiming to serve the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the developments encapsulated in this patent, the synthesis of cyclopentenone derivatives was predominantly dominated by methods reported by researchers such as Ishii, which relied heavily on zirconium chloride derivatives like ZrOCl2 or ZrCl4. While chemically feasible, these conventional routes imposed severe operational constraints that rendered them economically and environmentally suboptimal for large-scale manufacturing. The most critical drawback was the requirement for extremely high reaction temperatures, typically ranging from 130°C to 200°C, with optimal results often only achieved at the upper end of this thermal spectrum. Such stringent thermal conditions inevitably led to significant side reactions, polymerization, and decomposition of sensitive functional groups, resulting in disappointingly low isolated yields, sometimes as poor as 17% for fully substituted rings. Furthermore, the high energy input required to sustain these temperatures, coupled with the generation of substantial waste streams from degraded byproducts, made these processes unattractive for modern green chemistry initiatives. The harsh environment also limited the scope of substrates, preventing the incorporation of thermally labile groups that are increasingly common in modern drug design.
The Novel Approach
In stark contrast, the novel approach disclosed in CN100582072C utilizes a sophisticated titanium-based catalytic system that dramatically lowers the activation energy barrier for the cyclization reaction. By employing a metal complex of the formula M(OR5)4-nXn, where M is preferably Ti(IV), alongside specific co-ingredients, the reaction can proceed efficiently at temperatures between 60°C and 140°C, with a preferred window of 70°C to 110°C. This reduction in thermal stress not only preserves the integrity of the starting materials but also significantly enhances the selectivity towards the desired cyclopentenone core. The method demonstrates remarkable versatility, accommodating a wide range of substituents including alkyl, alkenyl, and aryl groups without compromising yield. For instance, experimental data within the patent shows that even with sterically hindered substrates, the process maintains viable conversion rates. This technological leap transforms the synthesis from a high-risk, low-yield operation into a predictable and controllable unit operation, thereby addressing the primary pain points of cost reduction in fine chemical manufacturing and enabling the production of high-purity intermediates essential for downstream API synthesis.
Mechanistic Insights into Titanium-Catalyzed Cyclization
The efficacy of this synthetic route is rooted in the unique Lewis acidic properties of the titanium center, which acts as a powerful activator for the carbonyl functionalities involved in the reaction. The catalytic cycle likely initiates with the coordination of the titanium species to the oxygen atoms of the enone and the aldehyde, increasing their electrophilicity and facilitating the initial carbon-carbon bond formation. The presence of the co-ingredient, such as anhydrous magnesium chloride or acetic anhydride, plays a dual role: it acts as a water scavenger to maintain strictly anhydrous conditions (critical for catalyst stability) and may also participate in generating a more active cationic titanium species in situ. This cooperative catalysis ensures that the reaction proceeds through a concerted mechanism that favors the formation of the five-membered ring over competing oligomerization pathways. The specific choice of ligands on the titanium atom, such as isopropoxy groups in TiCl3(OiPr), provides a balance between steric bulk and electronic density, optimizing the turnover number of the catalyst. Understanding these mechanistic nuances allows process chemists to fine-tune the reaction parameters, such as the molar ratio of enone to aldehyde, which is typically maintained between 1:1.1 and 1:5 to drive the equilibrium towards completion.
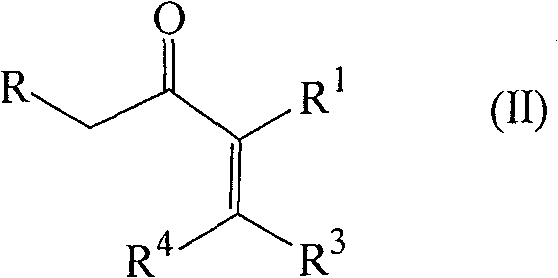
Furthermore, the control of impurities is intrinsically linked to the mildness of the catalytic system. In traditional high-temperature processes, thermal degradation often leads to a complex soup of polymeric tars and rearranged isomers that are difficult to separate. However, the titanium-catalyzed method minimizes these thermal stressors, resulting in a cleaner crude reaction profile. The selectivity is further enhanced by the specific interaction between the catalyst and the substrate geometry, which can influence the cis/trans ratio of the final product. For example, in the synthesis of 3,4-diphenyl-2,5-dimethyl-2-cyclopenten-1-one, the process yielded a trans/cis ratio of 85:15, demonstrating a strong stereochemical preference that simplifies downstream purification. This level of control is vital for pharmaceutical applications where isomeric purity can dictate biological activity. By avoiding the use of transition metals that are difficult to remove or toxic heavy metals, the process also aligns with strict regulatory guidelines regarding residual metals in drug substances, thereby reducing the burden on quality control laboratories.
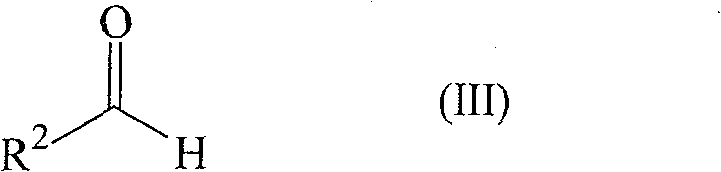
How to Synthesize Substituted 2-Cyclopenten-1-ones Efficiently
Implementing this synthesis on a pilot or commercial scale requires careful attention to the preparation of the catalytic species and the maintenance of anhydrous conditions throughout the process. The general procedure involves charging a reactor equipped with efficient agitation and temperature control with the starting ketone or enone, often dissolved in a solvent like butyl acetate which serves both as a reaction medium and azeotropic agent. The catalyst solution, prepared separately or in situ from precursors like TiCl4 and titanium tetraisopropoxide, is added along with the co-ingredient salt. Once the mixture reaches the target temperature of approximately 90°C to 100°C, the aldehyde is introduced slowly to manage the exotherm and ensure uniform reaction progression. Detailed standardized operating procedures for this specific transformation are critical for ensuring safety and consistency.
- Prepare a catalytic solution containing a titanium complex such as TiCl3(OiPr) and a co-ingredient like anhydrous magnesium chloride.
- Charge a reactor with the starting ketone or enone and solvent, then heat the mixture to a temperature range between 60°C and 140°C.
- Add the aldehyde reactant dropwise over several hours, maintain reaction temperature, and subsequently isolate the product via fractional distillation.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this titanium-catalyzed technology offers profound advantages that extend beyond simple chemical yield. The shift to milder reaction conditions directly translates into substantial operational expenditure savings, as the energy required to heat reactors to 200°C is eliminated in favor of much lower temperature ranges. This reduction in thermal demand not only lowers utility costs but also extends the lifespan of reactor vessels and heating jackets, reducing capital depreciation rates. Moreover, the simplified workup procedure, which often involves straightforward hydrolysis and fractional distillation without the need for complex chromatographic separations, significantly reduces the consumption of silica gel and elution solvents. These factors collectively contribute to a more lean and efficient manufacturing process, allowing suppliers to offer more competitive pricing structures without compromising on margin. The robustness of the catalyst system also means that raw material specifications can be slightly more relaxed compared to ultra-sensitive organometallic reactions, providing greater flexibility in sourcing bulk chemicals.
- Cost Reduction in Manufacturing: The elimination of extreme high-temperature requirements drastically reduces energy consumption per kilogram of product, while the use of inexpensive and abundant titanium precursors instead of precious metals lowers the direct material cost of the catalyst system. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove byproducts, thereby reducing the volume of solvents and adsorbents needed for purification, which is a major cost driver in fine chemical production.
- Enhanced Supply Chain Reliability: Because the reagents involved, such as titanium tetrachloride, magnesium chloride, and common aldehydes, are commodity chemicals available from multiple global vendors, the supply chain is not vulnerable to single-source bottlenecks. The process tolerance to various solvents, including the ability to use the reactants themselves as solvents, further mitigates risks associated with solvent shortages or price volatility, ensuring continuous production capability even during market disruptions.
- Scalability and Environmental Compliance: The absence of toxic heavy metals and the generation of less hazardous waste streams simplify the environmental permitting process and reduce waste disposal costs. The reaction's compatibility with standard stainless steel equipment facilitates easy scale-up from laboratory grams to multi-tonne commercial batches, allowing manufacturers to respond rapidly to increased market demand without requiring specialized exotic alloy reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cyclopentenone synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of the titanium-catalyzed method over traditional zirconium catalysts?
A: The titanium-catalyzed process described in CN100582072C operates at significantly milder temperatures (60°C-140°C) compared to the harsh 130°C-200°C required by zirconium methods, resulting in reduced energy consumption and improved safety profiles.
Q: Which solvents are compatible with this cyclopentenone synthesis route?
A: The process is highly flexible regarding solvents, functioning effectively in esters like butyl acetate, ethers, aromatic solvents, or even without solvent if the starting materials act as the medium, provided water content is kept below 1wt%.
Q: Can this method be scaled for commercial production of API intermediates?
A: Yes, the use of standard industrial equipment such as mechanical stirrers and fractionation columns, combined with mild reaction conditions and commercially available catalysts like TiCl4 derivatives, makes this route highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a robust and scalable supply chain for key pharmaceutical building blocks like substituted cyclopentenones. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global API manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the identity and purity of every batch against the highest industry standards. Our facility is equipped to handle the specific anhydrous conditions and distillation requirements outlined in this patent, guaranteeing a consistent supply of high-quality intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this titanium-catalyzed method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your drug development journey.
