Advanced Synthesis of 2-Difluoroethoxy-6-Trifluoromethylbenzenesulfonyl Chloride for Commercial Scale-Up
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of critical agrochemical intermediates. A recent breakthrough detailed in patent CN114539103A introduces a robust and environmentally superior method for producing 2-difluoroethoxy-6-trifluoromethylbenzenesulfonyl chloride, a key precursor for the widely used herbicide Penoxsulam. This technical advancement addresses long-standing challenges in the industry, specifically focusing on safety hazards and waste management complexities inherent in traditional manufacturing protocols. By shifting away from hazardous reagents like sodium hydride and problematic solvents such as DMF, this new pathway offers a compelling value proposition for R&D directors and supply chain managers alike. The process leverages a strategic three-step sequence involving controlled oxidation, nucleophilic aromatic substitution, and final chlorination, achieving a total yield exceeding 96% with product purity surpassing 98%. For stakeholders seeking a reliable agrochemical intermediate supplier, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a competitive edge in the herbicide market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 2-difluoroethoxy-6-trifluoromethylbenzenesulfonyl chloride has been fraught with significant operational risks and environmental burdens that complicate large-scale manufacturing. The conventional route typically relies on the use of sodium hydride to facilitate the introduction of the difluoroethoxy group, a reagent that demands extremely stringent control over moisture and oxygen levels to prevent violent exothermic reactions or explosions. Furthermore, these legacy processes often utilize dimethylformamide (DMF) as the reaction solvent, which presents severe stability issues under the strong alkaline and high-temperature conditions required for the synthesis. When DMF decomposes in the presence of strong bases at temperatures above 85°C, it generates difficult-to-treat wastewater and poses potential explosion hazards during the quenching phase. Additionally, the final chlorination step in older methods frequently employs a formic acid system, leading to the generation of substantial volumes of formic acid-containing wastewater that is notoriously difficult and costly to remediate. These factors collectively result in elevated production costs, extended lead times due to safety protocols, and a heavy regulatory burden regarding three-waste treatment.
The Novel Approach
In stark contrast to the hazardous legacy methods, the innovative synthesis route disclosed in the patent data fundamentally reengineers the chemical pathway to prioritize safety and efficiency without compromising yield. The new method initiates with the oxidation of 2-fluoro-6-trifluoromethylpropylphenyl sulfide using hydrogen peroxide in the presence of an organic acid, a much safer oxidant system that operates effectively at moderate temperatures between 60°C and 70°C. 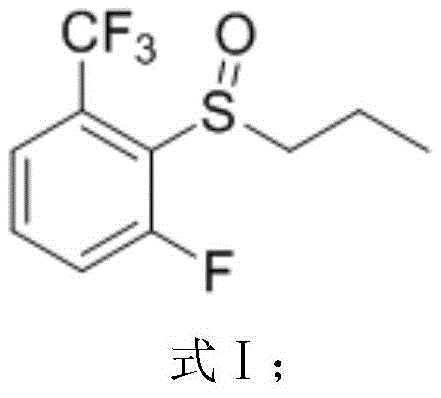 This first step generates the sulfoxide intermediate with exceptional conversion rates, setting a high-purity foundation for subsequent transformations. The critical substitution step then proceeds in 2-methyltetrahydrofuran (2-MeTHF), a greener and more stable solvent alternative to DMF, utilizing potassium hydroxide and difluoroethanol to install the ether functionality.
This first step generates the sulfoxide intermediate with exceptional conversion rates, setting a high-purity foundation for subsequent transformations. The critical substitution step then proceeds in 2-methyltetrahydrofuran (2-MeTHF), a greener and more stable solvent alternative to DMF, utilizing potassium hydroxide and difluoroethanol to install the ether functionality. 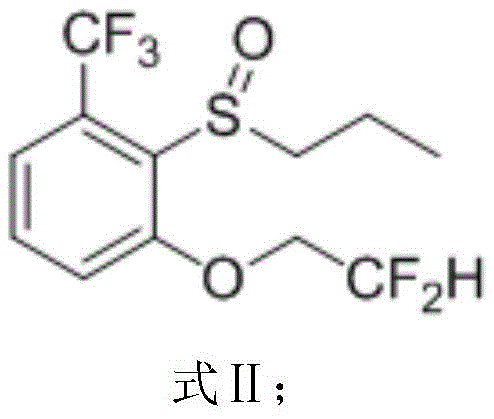 Finally, the chlorination is conducted in a dichloroethane-water biphasic system at mild temperatures of 20-30°C, completely eliminating the need for formic acid and its associated waste streams.
Finally, the chlorination is conducted in a dichloroethane-water biphasic system at mild temperatures of 20-30°C, completely eliminating the need for formic acid and its associated waste streams. 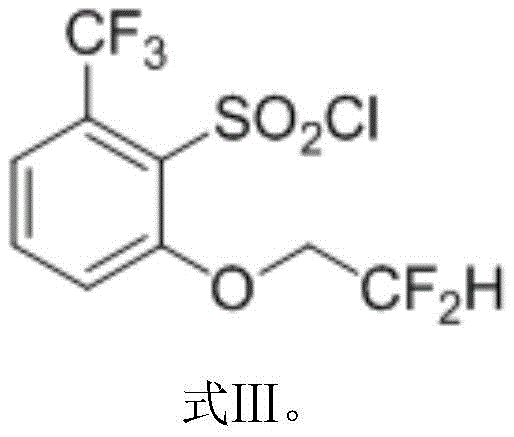 This holistic redesign not only mitigates safety risks but also streamlines the downstream purification process, offering a clear pathway for cost reduction in agrochemical intermediate manufacturing.
This holistic redesign not only mitigates safety risks but also streamlines the downstream purification process, offering a clear pathway for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into the Three-Step Synthetic Route
The success of this novel synthesis lies in the precise control of reaction kinetics and the selection of compatible reagents that minimize side reactions. In the initial oxidation step, the sulfur atom of the thioether is selectively oxidized to the sulfoxide state using hydrogen peroxide activated by a carboxylic acid catalyst. This mechanism avoids over-oxidation to the sulfone, which is a common impurity in less controlled systems, thereby ensuring the high purity of the intermediate required for the subsequent nucleophilic attack. The choice of organic acid, such as glacial acetic acid or formic acid, plays a crucial role in modulating the electrophilicity of the peroxide species, allowing the reaction to proceed smoothly at reflux temperatures without degrading the sensitive trifluoromethyl group on the aromatic ring. This careful balance is critical for maintaining the structural integrity of the molecule while introducing the necessary oxygen functionality.
Following the oxidation, the nucleophilic aromatic substitution represents the core transformation of the sequence, where the fluorine atom ortho to the sulfoxide group is displaced by the difluoroethoxide anion. The presence of the electron-withdrawing sulfoxide and trifluoromethyl groups activates the aromatic ring towards nucleophilic attack, facilitating the displacement of the fluoride leaving group under relatively mild basic conditions provided by potassium hydroxide. The use of 2-methyltetrahydrofuran as the solvent is particularly advantageous here, as it stabilizes the anionic intermediates and allows for easy separation of the product from the aqueous salt byproducts during the work-up phase. The final chlorination step involves the conversion of the sulfide/sulfoxide moiety into the sulfonyl chloride using chlorine gas. This reaction is highly exothermic and requires precise temperature control between 20°C and 30°C to prevent thermal runaway and ensure the selective formation of the sulfonyl chloride without chlorinating the aromatic ring or the alkyl side chains, thus preserving the high purity specifications demanded by pharmaceutical and agrochemical applications.
How to Synthesize 2-Difluoroethoxy-6-Trifluoromethylbenzenesulfonyl Chloride Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters to maximize yield and safety. The process begins with the preparation of the sulfoxide intermediate, followed by the etherification in a recyclable solvent system, and concludes with a controlled gas-phase chlorination. Each step has been engineered to allow for straightforward isolation techniques, such as phase separation and distillation, which are scalable and equipment-friendly. For detailed operational protocols, including specific molar ratios, addition rates, and quality control checkpoints, please refer to the standardized synthesis guide below.
- Oxidize 2-fluoro-6-trifluoromethylpropylphenyl sulfide using hydrogen peroxide and organic acid at 60-70°C to form the sulfoxide intermediate.
- React the sulfoxide with difluoroethanol and potassium hydroxide in 2-methyltetrahydrofuran at 75-85°C to introduce the difluoroethoxy group.
- Chlorinate the resulting ether sulfoxide using chlorine gas in a dichloroethane-water system at 20-30°C to yield the final sulfonyl chloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend beyond simple chemical efficiency. By eliminating high-risk reagents like sodium hydride, facilities can significantly reduce their insurance premiums and safety compliance overheads, leading to substantial cost savings in overall operational expenditure. The removal of DMF from the process not only mitigates the risk of solvent decomposition and explosion but also drastically simplifies wastewater treatment protocols, as the new solvent system allows for efficient recycling and recovery of 2-methyltetrahydrofuran and dichloroethane. This reduction in hazardous waste volume directly translates to lower disposal costs and a smaller environmental footprint, aligning with increasingly stringent global sustainability mandates. Furthermore, the high total yield of over 96% ensures that raw material utilization is maximized, reducing the cost of goods sold and enhancing the economic viability of producing this critical herbicide intermediate on a commercial scale.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium hydride removes the need for specialized handling equipment and inert atmosphere protocols, which are capital-intensive to maintain. Additionally, the ability to recycle solvents such as 2-methyltetrahydrofuran and dichloroethane creates a closed-loop system that minimizes raw material consumption and waste generation. This streamlined approach reduces the overall complexity of the manufacturing process, allowing for faster batch turnover and lower utility costs associated with heating and cooling cycles. Consequently, manufacturers can achieve a more competitive pricing structure for the final intermediate while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents, such as hydrogen peroxide and potassium hydroxide, ensures a robust supply chain that is less susceptible to disruptions compared to processes dependent on specialty chemicals like sodium hydride. The simplified work-up procedures, which utilize standard phase separations and distillations rather than complex chromatographic purifications, enable faster production cycles and shorter lead times for order fulfillment. This operational agility allows suppliers to respond more rapidly to fluctuations in market demand, ensuring a continuous and reliable flow of high-purity intermediates to downstream herbicide producers.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction conditions that are easily manageable in large-scale reactors without the extreme safety risks associated with legacy methods. The reduction in hazardous waste generation, particularly the avoidance of formic acid and DMF-containing effluents, simplifies regulatory compliance and reduces the burden on environmental treatment facilities. This eco-friendly profile not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the brand reputation of the supplier as a responsible partner in the global agrochemical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 2-difluoroethoxy-6-trifluoromethylbenzenesulfonyl chloride, based on the detailed disclosures in the patent literature. These insights are intended to clarify the feasibility and benefits of adopting this advanced manufacturing route for industrial applications. Understanding these details is crucial for technical teams evaluating process transfers or capacity expansions.
Q: How does this new synthesis method improve safety compared to conventional routes?
A: The novel process eliminates the use of sodium hydride, a highly reactive and moisture-sensitive reagent that poses significant explosion risks. Additionally, it replaces DMF with 2-methyltetrahydrofuran, avoiding the decomposition and wastewater treatment issues associated with amide solvents under alkaline conditions.
Q: What is the expected purity and yield of the final product using this method?
A: According to the patent data, the total yield of the three-step process exceeds 96%, with the final product achieving a purity of over 98%. Individual step yields are consistently high, ranging from 98% to 99%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. It utilizes recyclable solvents like 2-methyltetrahydrofuran and dichloroethane, simplifies work-up procedures through phase separation, and avoids hazardous reagents, making it ideal for industrial manufacturing of agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Difluoroethoxy-6-Trifluoromethylbenzenesulfonyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and safe synthesis routes in the production of high-value agrochemical intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and secure. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of 2-difluoroethoxy-6-trifluoromethylbenzenesulfonyl chloride meets the exacting standards required for Penoxsulam synthesis. We leverage our deep technical expertise to optimize reaction parameters, ensuring maximum yield and minimal impurity profiles that align with the latest patent advancements.
We invite global partners to collaborate with us to leverage these technological breakthroughs for their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your herbicide production needs.
