Advanced Continuous Flow Synthesis of DL-Aminopropionitrile for Commercial Scale-Up
Advanced Continuous Flow Synthesis of DL-Aminopropionitrile for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust, scalable, and safe methodologies for producing critical antihypertensive intermediates. A significant breakthrough in this domain is detailed in patent CN115286532A, which discloses a novel method for the continuous synthesis of DL-aminopropionitrile, a pivotal precursor for the drug Methyldopa. This technology represents a paradigm shift from traditional batch processing to advanced continuous flow chemistry, leveraging micro-channel reactors to achieve superior control over reaction parameters. By integrating precise mixing dynamics with tubular reactor systems, this approach effectively addresses longstanding challenges such as the hazardous handling of solid sodium cyanide, excessive side reactions, and low production efficiency. The result is a streamlined process capable of delivering high-purity intermediates essential for downstream pharmaceutical applications.
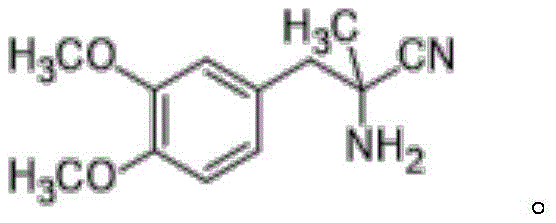
The structural integrity and purity of DL-aminopropionitrile are paramount for the efficacy of the final antihypertensive medication. As illustrated in the chemical structure, the molecule features a dimethoxyphenyl group attached to an aminonitrile backbone, requiring precise synthetic conditions to avoid isomerization or degradation. The patented continuous flow method ensures that the delicate balance of reagents—veratone, ammonium chloride, and sodium cyanide—is maintained throughout the reaction pathway. This level of control is difficult to achieve in conventional stirred tank reactors, where gradients in concentration and temperature often lead to inconsistent product quality. For R&D directors and process chemists, adopting this continuous methodology offers a clear pathway to optimizing the impurity profile and ensuring batch-to-batch consistency in commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of DL-3-(3,4-dimethoxyphenyl)-2-amino-2-methylpropanenitrile has relied heavily on batch-type intermittent production processes, typically involving large stirred kettles. In these traditional setups, solid sodium cyanide must be fed manually or via complex dosing systems into an aqueous ammonia solution containing ammonium chloride and veratone. This solid-liquid heterogeneous reaction environment presents significant operational hazards and inefficiencies. The mixing efficiency in large vessels is often insufficient to prevent local hotspots, leading to uncontrolled exothermic reactions and the formation of numerous byproducts. Furthermore, the reaction times in batch processes are notoriously long, often extending between 8 to 9 hours, which severely limits throughput. The subsequent workup requires cooling, filtration, and extensive water washing to remove impurities, generating substantial volumes of wastewater and complicating the isolation of the crude product.
The Novel Approach
In stark contrast, the novel approach outlined in the patent utilizes a sophisticated continuous flow system comprising micro-mixers and tubular reactors. This method transforms the synthesis into a seamless pipeline operation where feed liquids are precisely metered and mixed at the molecular level before entering the reaction zone. The first stage involves mixing an ammonia water solution of sodium cyanide and ammonium chloride with a solution of veratone in an organic solvent. This mixture immediately enters a first tubular reactor where the initial Strecker reaction occurs under tightly controlled thermal conditions. The second stage introduces additional organic solvent to create an oil-water two-phase system, facilitating in-situ extraction of the product into the organic phase. This innovative design not only accelerates the reaction kinetics, reducing residence times to mere minutes, but also effectively prevents the precipitation of solids within the reactor, thereby eliminating clogging risks and ensuring smooth continuous operation.
Mechanistic Insights into Micro-Channel Strecker Reaction
The core of this technological advancement lies in the exploitation of micro-fluidic dynamics to enhance mass and heat transfer during the Strecker reaction. In a conventional batch reactor, the diffusion of cyanide ions to the carbonyl carbon of veratone is limited by bulk mixing, often resulting in concentration gradients that favor side reactions. However, within the micro-channel mixers described in the patent, the reagents are subjected to intense shear forces and laminar flow regimes that reduce the diffusion path length to the micrometer scale. This ensures that the nucleophilic attack of the cyanide ion on the imine intermediate occurs uniformly and rapidly. The precise temperature control, maintained between 30°C and 60°C in the first reactor and 20°C to 40°C in the second, prevents thermal degradation of the sensitive aminonitrile functionality. Such thermal precision is critical for suppressing the hydrolysis of the nitrile group or the polymerization of reactive intermediates, which are common failure modes in less controlled environments.
Furthermore, the mechanism incorporates a strategic in-situ extraction step that fundamentally alters the reaction equilibrium and product stability. By introducing a water-immiscible organic solvent, such as dichloromethane or dichloroethane, into the reaction stream at the second micro-mixer, the synthesized DL-aminopropionitrile is continuously partitioned into the organic phase. This immediate removal of the product from the aqueous reaction medium serves a dual purpose: it drives the reaction forward according to Le Chatelier's principle and protects the product from prolonged exposure to the aqueous alkaline environment which could promote decomposition. The result is a reaction liquid that exits the second tubular reactor as a stable organic solution, ready for direct separation and purification. This mechanistic refinement effectively bypasses the need for solid-liquid separation steps like filtration or centrifugation, which are major bottlenecks in traditional processing, thereby significantly enhancing the overall atom economy and process efficiency.
How to Synthesize DL-Aminopropionitrile Efficiently
Implementing this continuous synthesis route requires a systematic setup of pumping systems, micro-mixers, and temperature-controlled tubular reactors. The process begins with the preparation of two distinct feed streams: an aqueous phase containing the cyanide and ammonium sources, and an organic phase containing the ketone substrate. These streams are then converged in a high-efficiency micro-mixer to initiate the reaction before flowing through a heated coil reactor. The key to success lies in optimizing the residence time and the ratio of solvents to ensure complete conversion while maintaining a single-phase or stable emulsion flow that prevents reactor fouling. Detailed operational parameters, including specific flow rates and molar ratios derived from the patent examples, are essential for replicating the high yields and purity reported. For a comprehensive guide on the exact procedural steps and equipment configuration, please refer to the standardized synthesis protocol below.
- Prepare Feed Liquid A by dissolving ammonium chloride and sodium cyanide in ammonia water, and Feed Liquid B by dissolving veratone in an organic solvent like dichloromethane.
- Pump Feed A and Feed B into a first micro-mixer and tubular reactor maintained at 30-60°C to initiate the Strecker reaction, forming a mixture containing the intermediate.
- Introduce additional organic solvent into the reaction mixture via a second micro-mixer and react in a second tubular reactor at 20-40°C to facilitate in-situ extraction and complete the reaction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition from batch to continuous flow synthesis offers profound strategic benefits beyond mere technical elegance. The primary advantage is the drastic simplification of the manufacturing workflow, which directly translates to reduced operational expenditures and enhanced safety profiles. By eliminating the need for handling solid sodium cyanide in open vessels and removing energy-intensive filtration and drying steps, the process inherently lowers the cost of goods sold. The continuous nature of the reaction also means that production can be scaled linearly by running the system for longer durations or numbering up reactor units, rather than building massive new infrastructure. This flexibility allows manufacturers to respond more agilely to market demand fluctuations without the risk of over-investing in fixed assets, providing a more resilient supply chain for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The implementation of micro-reactor technology significantly reduces manufacturing costs by minimizing raw material waste and energy consumption. Traditional batch processes often suffer from yield losses due to side reactions and inefficient mixing, necessitating expensive purification steps to meet pharmaceutical grade specifications. In contrast, the continuous flow method achieves high conversion rates and selectivity, meaning less starting material is wasted and fewer resources are dedicated to downstream purification. Additionally, the reduction in wastewater generation due to the elimination of extensive washing steps lowers environmental compliance costs. The ability to operate with higher concentrations of reagents further enhances space-time yield, allowing more product to be generated per unit volume of reactor, which optimizes capital utilization.
- Enhanced Supply Chain Reliability: Supply chain reliability is bolstered by the inherent safety and controllability of the continuous process. Handling toxic cyanide salts in a closed, automated pipeline system drastically reduces the risk of operator exposure and accidental spills, which are common causes of production shutdowns in batch facilities. The robustness of the micro-channel system against thermal runaways ensures consistent production uptime, mitigating the risk of supply disruptions caused by safety incidents or equipment failures. Furthermore, the simplified process flow reduces the dependency on specialized labor for complex batch operations, making the supply chain less vulnerable to workforce shortages. This reliability is crucial for maintaining the continuity of supply for downstream API manufacturers who depend on steady deliveries of high-quality intermediates.
- Scalability and Environmental Compliance: Scaling up chemical processes is traditionally fraught with engineering challenges, but continuous flow chemistry offers a straightforward path from laboratory to commercial production. The parameters optimized in small-diameter tubular reactors can be directly translated to larger production scales by increasing the length of the reactor coils or operating multiple units in parallel, avoiding the non-linear heat and mass transfer issues associated with scaling up batch kettles. From an environmental perspective, the process aligns with green chemistry principles by reducing solvent usage and waste generation. The efficient in-situ extraction minimizes the volume of aqueous waste requiring treatment, and the closed-loop nature of the system prevents the release of volatile organic compounds. This strong environmental profile facilitates easier regulatory approval and supports corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the continuous synthesis of DL-aminopropionitrile. These insights are derived directly from the experimental data and technical disclosures found in the relevant patent literature. Understanding these details is vital for stakeholders evaluating the feasibility of adopting this technology for their own manufacturing lines or sourcing strategies. The answers provided reflect the specific advantages of micro-reactor systems over conventional methods, focusing on yield optimization, safety improvements, and process intensification.
Q: What are the primary advantages of using micro-reactors for DL-aminopropionitrile synthesis?
A: The use of micro-reactors allows for precise temperature control and rapid mixing, which significantly reduces side reactions and impurities compared to traditional batch kettle methods. It also solves the difficulty of feeding solid sodium cyanide safely and efficiently.
Q: How does the continuous process improve yield and purity?
A: By utilizing a two-stage tubular reactor system with in-situ solvent extraction, the process prevents the precipitation of solids during reaction, thereby improving atom utilization and achieving purities exceeding 97% with yields around 97%.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the continuous flow nature of the process facilitates automatic operation and pipeline transfer, making it highly scalable for industrial production while minimizing operator exposure to hazardous cyanide materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable DL-Aminopropionitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow technologies in the production of high-value pharmaceutical intermediates like DL-aminopropionitrile. Our team of expert process engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of micro-reactor synthesis are fully realized in an industrial setting. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify the absence of critical impurities. By leveraging our state-of-the-art manufacturing facilities, we can offer a reliable supply of this essential methyldopa intermediate, supporting your drug development and commercialization timelines with unwavering consistency.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through advanced process chemistry. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for DL-aminopropionitrile and other complex intermediates. Together, we can drive innovation in pharmaceutical manufacturing, ensuring safer, more efficient, and more sustainable production of life-saving medications.
