Optimizing Tapentadol Intermediate Production: A Technical Analysis of Patent CN102206164A
Optimizing Tapentadol Intermediate Production: A Technical Analysis of Patent CN102206164A
The pharmaceutical industry continuously seeks robust, scalable, and environmentally sustainable pathways for the production of high-value analgesic intermediates. Patent CN102206164A presents a significant technological advancement in the synthesis of the key Tapentadol intermediate, specifically (2R,3R)-3-(3-methoxyphenyl)-N,N,2-trimethylpentylamine hydrochloride. This patent addresses critical bottlenecks found in prior art methodologies by replacing hazardous chlorinating agents and stoichiometric reducing metals with safer, catalytic alternatives. For R&D directors and process chemists, this represents a shift towards greener chemistry that maintains high stereochemical integrity while drastically simplifying downstream processing. The core innovation lies in the substitution of thionyl chloride with phosphorus pentachloride and the replacement of metal hydride reductions with catalytic hydrogenolysis, a change that fundamentally alters the safety and economic profile of the manufacturing process.
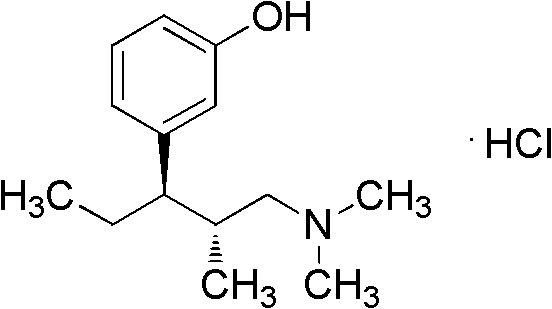
Tapentadol is a centrally acting synthetic analgesic with a dual mechanism of action, functioning as a mu-opioid receptor agonist and a norepinephrine reuptake inhibitor. Its clinical efficacy relies heavily on the specific stereochemistry of the molecule, necessitating highly enantioselective synthetic routes. The structure shown above highlights the critical chiral centers that must be preserved throughout the synthesis. The patent CN102206164A focuses on optimizing the conversion of the alcohol precursor to the final amine intermediate, a step that historically posed significant challenges regarding waste generation and reaction control. By refining this specific transformation, the patent offers a pathway that is not only chemically superior but also commercially more viable for large-scale API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in EP0693475, rely heavily on thionyl chloride for the chlorination step and metal hydrides like zinc borohydride or tin cyanoborohydride for the subsequent dechlorination. This conventional approach suffers from severe drawbacks that impact both operational safety and environmental compliance. The use of thionyl chloride generates substantial quantities of acidic off-gases, including hydrogen chloride and sulfur dioxide, requiring complex and costly scrubbing systems to prevent atmospheric release. Furthermore, the reliance on stoichiometric metal hydrides introduces significant hazards; these reagents are often pyrophoric or moisture-sensitive, creating fire risks in an industrial setting. Perhaps most critically from a supply chain perspective, the reduction step generates large volumes of heavy metal waste sludge, the disposal of which is increasingly regulated and expensive. These factors combine to create a process that is difficult to scale safely and economically.
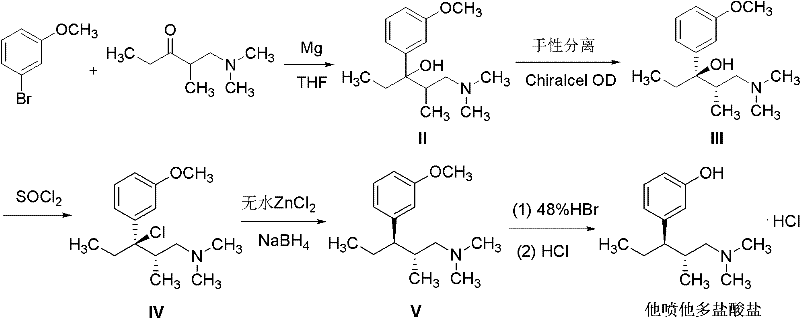
The Novel Approach
The methodology outlined in CN102206164A offers a transformative solution by re-engineering the reaction sequence to eliminate these specific pain points. Instead of gaseous or liquid chlorinating agents that release acid gases, the patent utilizes phosphorus pentachloride, a solid reagent that reacts cleanly without evolving corrosive vapors. This simple switch dramatically reduces the burden on废气 treatment infrastructure. More importantly, the reduction step is reinvented through catalytic hydrogenolysis. By employing a palladium on carbon (Pd/C) or Raney nickel catalyst under a hydrogen atmosphere, the process avoids the use of hazardous metal hydrides entirely. This catalytic approach not only eliminates heavy metal waste but also accelerates the reaction kinetics significantly. The patent explicitly notes a reduction in reaction time by nearly 80 hours compared to literature methods, a staggering improvement that directly translates to increased reactor throughput and reduced utility costs.
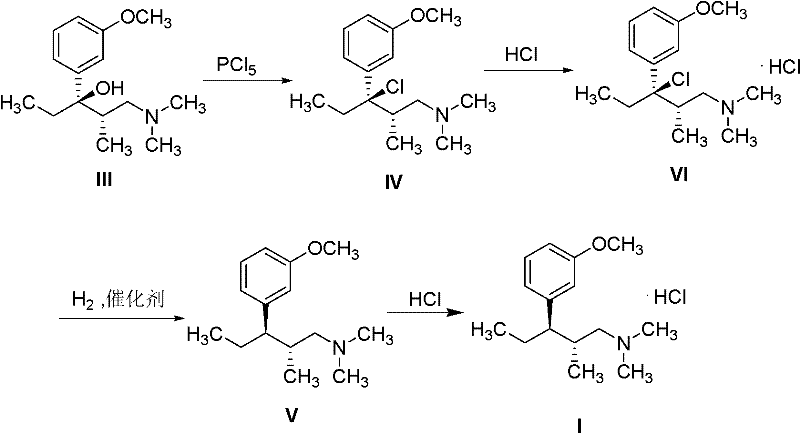
Mechanistic Insights into Stereoselective Hydrogenolysis
The core chemical transformation in this patent is the stereoselective hydrogenolysis of the benzylic chloride intermediate. Mechanistically, this involves the adsorption of the chloro-amine substrate onto the surface of the palladium catalyst. Under a hydrogen atmosphere, the carbon-chlorine bond is cleaved, and a hydrogen atom is delivered to the benzylic position. The preservation of stereochemistry at the adjacent chiral center is paramount, as any epimerization would lead to the formation of diastereomeric impurities that are difficult to separate. The patent specifies the use of mild conditions, typically in solvents like methanol or tetrahydrofuran at moderate temperatures (40-45°C), which helps maintain the integrity of the chiral centers. The choice of solvent plays a crucial role here; polar protic solvents like methanol can stabilize the transition state and facilitate the removal of the chloride ion as hydrochloric acid, which is then neutralized or removed during workup.
Impurity control is another critical aspect addressed by the mechanistic design of this route. In traditional metal hydride reductions, over-reduction or side reactions with the amine functionality can occur, leading to complex impurity profiles. The catalytic hydrogenation method described is highly chemoselective for the carbon-chlorine bond, leaving the dimethylamine group and the aromatic ring intact. Furthermore, the intermediate step of converting the crude chloro-compound into its hydrochloride salt (Intermediate VI) serves as a powerful purification checkpoint. Salt formation allows for recrystallization, which effectively removes non-basic impurities and unreacted starting materials before the final reduction step. This ensures that the catalyst is not poisoned by impurities and that the final product meets stringent purity specifications required for pharmaceutical intermediates.
How to Synthesize (2R,3R)-3-(3-methoxyphenyl)-N,N,2-trimethylpentylamine Efficiently
The synthesis protocol derived from this patent provides a clear, step-by-step guide for producing the target intermediate with high efficiency. The process begins with the chlorination of the optically active alcohol precursor using phosphorus pentachloride in a suitable solvent like dichloromethane. Following the reaction, the mixture is worked up to isolate the chloro-intermediate, which is then immediately converted to its stable hydrochloride salt form. This salt form is advantageous for storage and handling, preventing degradation and facilitating accurate dosing in the subsequent step. The final stage involves dissolving the salt in a solvent such as methanol and subjecting it to hydrogen gas in the presence of a palladium catalyst.
- Chlorinate optically active (2S,3R)-1-dimethylamino-3-(3-methoxyphenyl)-2-methylpentan-3-ol using phosphorus pentachloride in dichloromethane to form the chloro-intermediate.
- Convert the crude chloro-intermediate into its stable hydrochloride salt (Intermediate VI) to facilitate purification and handling.
- Perform stereoselective hydrogenolysis on Intermediate VI using a palladium on carbon (Pd/C) catalyst under hydrogen pressure to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN102206164A offers compelling strategic advantages beyond mere chemical elegance. The shift away from hazardous reagents directly correlates to a reduction in operational risk and insurance costs. By eliminating the need for specialized handling of pyrophoric metal hydrides and corrosive thionyl chloride, facilities can operate with greater safety margins and reduced regulatory burden. This inherently safer design also simplifies the qualification of manufacturing sites, as the environmental, health, and safety (EHS) profile of the process is significantly improved. Consequently, this leads to a more resilient supply chain that is less susceptible to disruptions caused by safety incidents or regulatory crackdowns on waste disposal.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive and hazardous reagents. Traditional methods require stoichiometric amounts of metal hydrides, which are costly to purchase and even costlier to dispose of as hazardous waste. By switching to catalytic hydrogenation, the consumption of reducing agents is minimized to catalytic levels, and the primary consumable becomes hydrogen gas, which is inexpensive. Additionally, the drastic reduction in reaction time—shortened by nearly 80 hours according to the patent data—means that reactor assets are utilized much more efficiently. This increased throughput allows for the production of larger volumes without the need for capital expenditure on new equipment, effectively lowering the fixed cost per kilogram of the intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of specialized reagents and the capacity of waste treatment facilities. The new method relies on commodity chemicals like phosphorus pentachloride and hydrogen, which are widely available globally, reducing the risk of raw material shortages. Furthermore, the stability of the hydrochloride salt intermediate allows manufacturers to produce and stockpile bulk quantities without fear of rapid degradation. This ability to decouple production stages provides a buffer against demand fluctuations, ensuring that downstream API synthesis can proceed uninterrupted. The simplified workup procedures also mean that batch cycles are faster, allowing suppliers to respond more agilely to urgent orders from pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat and mass transfer issues, particularly with exothermic reactions involving hazardous reagents. The catalytic hydrogenation described is inherently easier to scale because the heat of reaction is manageable and the reagents are less aggressive. From an environmental standpoint, the absence of heavy metal sludge and acidic off-gases aligns perfectly with modern green chemistry principles and strict environmental regulations. This compliance future-proofs the supply chain against tightening environmental laws, ensuring long-term viability. Manufacturers adopting this route can market their products as 'green' or 'sustainably produced,' adding value for pharmaceutical customers who are increasingly focused on the carbon footprint and environmental impact of their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: Why is phosphorus pentachloride preferred over thionyl chloride in this synthesis?
A: Phosphorus pentachloride is a solid reagent that avoids the generation of large volumes of acidic waste gases like sulfur dioxide and hydrogen chloride, which are typical byproducts of thionyl chloride reactions, thereby simplifying废气 treatment and improving workplace safety.
Q: How does the new hydrogenolysis method improve upon traditional metal hydride reductions?
A: Traditional methods using zinc borohydride or tin cyanoborohydride generate significant amounts of hazardous metal waste sludge and pose fire risks. The catalytic hydrogenation method described in the patent is cleaner, produces no heavy metal waste, and significantly reduces reaction time.
Q: What are the purity advantages of isolating the hydrochloride salt intermediate?
A: Converting the intermediate into its hydrochloride salt form allows for effective recrystallization, resulting in higher optical and chemical purity. This stable solid form also simplifies storage and direct feeding into subsequent reaction steps without the need for free-basing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tapentadol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and compliant synthesis routes for high-value pharmaceutical intermediates like Tapentadol. Our technical team has extensively analyzed the methodology presented in CN102206164A and possesses the expertise to implement this advanced catalytic hydrogenation process at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practical manufacturing. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest quality standards required for API synthesis.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for analgesic intermediates. By leveraging our technical capabilities, you can achieve Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you secure a reliable, cost-effective, and sustainable source of Tapentadol intermediates that supports your long-term commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
