Revolutionizing Chiral Amine Production for Repotrectinib Intermediates via Ruthenium Catalysis
Revolutionizing Chiral Amine Production for Repotrectinib Intermediates via Ruthenium Catalysis
The pharmaceutical industry is constantly seeking more efficient pathways to synthesize complex chiral intermediates, particularly for next-generation kinase inhibitors like Repotrectinib. Patent CN114349648A introduces a groundbreaking preparation method for chiral amine compounds that addresses critical bottlenecks in current manufacturing processes. This technology utilizes a highly selective ruthenium-based metal catalyst system to achieve direct asymmetric reductive amination in a single step. By leveraging specific chiral ligands and ammonium salts under hydrogen pressure, this method transforms readily available ketone substrates into optically pure amines with exceptional efficiency. The significance of this innovation lies in its ability to bypass multi-step sequences involving expensive chiral auxiliaries, offering a streamlined route to key intermediates such as (R)-I, which is essential for the production of Repotrectinib and related TRK inhibitors.
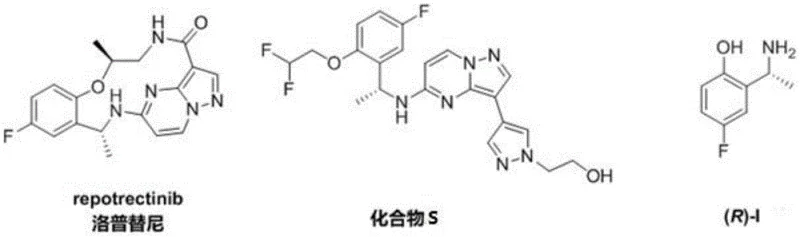
Repotrectinib represents a broad-spectrum third-generation TRK inhibitor capable of overcoming resistance mutations in ROS1, TRK, and ALK positive tumors. The demand for high-purity intermediates for such advanced therapeutics is escalating rapidly. The disclosed method not only simplifies the synthetic route but also aligns with green chemistry principles by minimizing waste generation. For R&D directors and process chemists, this patent offers a viable alternative to legacy methods, promising higher throughput and reduced environmental impact. The versatility of the catalyst system allows for the synthesis of various analogues, making it a robust platform technology for diverse pharmaceutical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically pure chiral amines like intermediate (R)-I has relied on cumbersome multi-step protocols that are both cost-prohibitive and environmentally burdensome. As detailed in the background of the patent, traditional Route 1 involves the use of stoichiometric amounts of expensive chiral prosthetic groups, specifically (R)-tert-butylsulfinamide. This approach requires the formation of a sulfinylimine followed by Grignard addition, a process that suffers from poor stereochemical control and necessitates purification via column chromatography, ultimately yielding the desired amine in only 58% yield. Furthermore, Route 2 employs lithium triethylborohydride and corrosive boron tribromide at low temperatures, introducing significant safety hazards and operational complexity. These conventional pathways are characterized by long reaction sequences, low overall yields, and the generation of substantial chemical waste, which poses challenges for large-scale manufacturing and regulatory compliance.
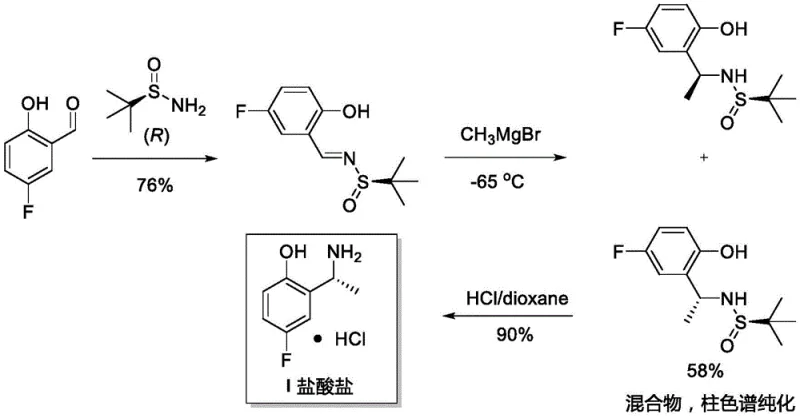
The Novel Approach
In stark contrast to these legacy methods, the novel approach described in CN114349648A achieves the synthesis of chiral amines through a direct, one-step reductive amination. This transformative process utilizes a ruthenium-based catalyst in the presence of an ammonium salt and hydrogen gas to convert ketone substrates directly into the corresponding chiral amines. By eliminating the need for chiral auxiliaries and protecting group manipulations, this method drastically shortens the process flow. The reaction conditions are relatively mild, typically operating between 60°C and 100°C under hydrogen pressures of 30 to 60 atm. This catalytic strategy not only improves atom economy but also simplifies the downstream processing, as the crude products can often be purified through standard extraction and crystallization techniques rather than labor-intensive chromatography. The result is a significantly more economical and sustainable manufacturing process suitable for industrial application.
Mechanistic Insights into Ru-Catalyzed Asymmetric Reductive Amination
The core of this technological advancement lies in the sophisticated design of the ruthenium-based metal catalyst system. The catalyst typically comprises a ruthenium precursor, such as Ru(OAc)2 or [RuCl(p-cymene)]Cl, coordinated with a chiral diphosphine ligand. The patent specifies a range of high-performance ligands including Segphos, BINAP, and TunePhos derivatives, each possessing distinct steric and electronic properties that dictate the stereochemical outcome of the reaction. During the catalytic cycle, the ruthenium center activates molecular hydrogen and facilitates the transfer of hydride to the imine intermediate formed in situ from the ketone and ammonium salt. The chiral environment created by the ligand ensures that hydride delivery occurs selectively to one face of the planar imine, thereby establishing the desired stereocenter with high fidelity. This mechanism allows for precise control over the configuration of the resulting amine, whether (R) or (S), simply by selecting the appropriate enantiomer of the chiral ligand.
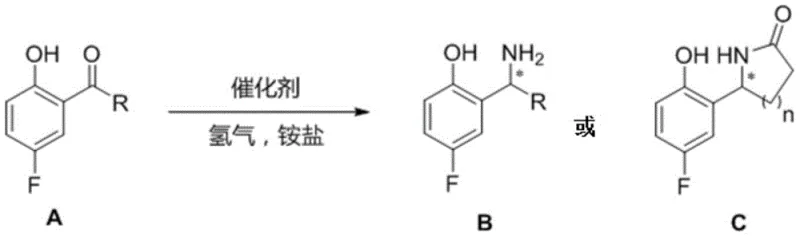
Impurity control is another critical aspect where this catalytic method excels. In traditional stoichiometric routes, side reactions such as over-reduction or incomplete deprotection often lead to complex impurity profiles that are difficult to separate. The catalytic reductive amination described here minimizes these risks by operating under thermodynamic control with high selectivity. The use of ammonium salts like ammonium acetate or ammonium benzoate serves as a mild nitrogen source, preventing the formation of harsh byproducts associated with ammonia gas or unstable imines. Furthermore, the robustness of the ruthenium catalyst ensures consistent performance across different substrate scopes, from simple alkyl ketones to more complex aryl and heteroaryl derivatives. This reliability is paramount for pharmaceutical manufacturing, where batch-to-batch consistency and strict impurity limits are non-negotiable requirements for regulatory approval.
How to Synthesize Chiral Amine Compounds Efficiently
The practical implementation of this synthesis route is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically argon, to prevent catalyst deactivation. The substrate, catalyst, and ammonium salt are dissolved in a suitable alcohol solvent such as methanol or ethanol. The reaction is then conducted in a high-pressure autoclave, where hydrogen gas is introduced to the specified pressure. Following the reaction period, the mixture is carefully quenched and worked up to isolate the pure chiral amine. Detailed standardized synthesis steps for replicating this high-efficiency protocol are provided in the guide below.
- Combine the ketone substrate (Compound A), ruthenium-based metal catalyst (e.g., Ru(OAc)2[(R)-Segphos]), and ammonium salt (e.g., ammonium acetate) in an alcohol solvent within a reaction vessel under inert atmosphere.
- Charge the reactor with hydrogen gas to a pressure of 30 to 60 atm and heat the mixture to a temperature between 60°C and 100°C for 12 to 48 hours with stirring.
- Quench the reaction with a weak alkaline solution such as saturated sodium bicarbonate, extract the organic phase, and purify the resulting chiral amine product via standard workup procedures.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology presents a compelling value proposition centered on cost efficiency and supply security. The shift from stoichiometric chiral auxiliaries to a catalytic system fundamentally alters the cost structure of producing these critical intermediates. By removing the requirement for expensive reagents like tert-butylsulfinamide and avoiding the use of hazardous chemicals such as boron tribromide, the raw material costs are significantly reduced. Additionally, the consolidation of multiple synthetic steps into a single operation reduces labor costs, energy consumption, and equipment occupancy time. This streamlining of the manufacturing process translates directly into lower unit costs, allowing for more competitive pricing in the global market for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries represents a major financial saving, as these reagents are often among the most expensive components in asymmetric synthesis. Furthermore, the simplified workup procedure reduces the consumption of solvents and silica gel required for chromatographic purification. The high yields reported in the patent examples, often exceeding 90%, mean that less starting material is wasted, further enhancing the overall economic efficiency of the process. These factors combined contribute to a substantial reduction in the cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on complex multi-step syntheses increases the risk of supply disruptions due to the failure of any single step or the shortage of a specific reagent. This one-step catalytic method mitigates such risks by simplifying the supply chain to readily available ketones and robust catalysts. The use of common solvents like methanol and ethanol ensures that there are no bottlenecks related to specialized solvent availability. Moreover, the scalability of the reaction, as demonstrated by the successful gram-scale experiments, assures buyers that the supply can be ramped up quickly to meet clinical or commercial demand without the need for extensive process re-engineering.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern sustainability goals. The reduction in chemical waste and the avoidance of toxic reagents simplify waste treatment processes and lower disposal costs. The high atom economy of the reductive amination reaction means that a greater proportion of the reactants end up in the final product, minimizing the environmental footprint. For companies aiming to reduce their carbon footprint and adhere to stricter environmental regulations, adopting this greener synthesis route offers a strategic advantage. The ease of scale-up also means that transitioning from pilot batches to multi-ton commercial production can be achieved with minimal technical barriers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ruthenium-catalyzed synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on the feasibility and performance of the technology.
Q: What are the key advantages of this ruthenium-catalyzed method over traditional sulfinamide routes?
A: This method eliminates the need for stoichiometric chiral auxiliaries like tert-butylsulfinamide, reducing the synthesis from three or four steps to a single direct reductive amination step, thereby significantly lowering waste and production costs.
Q: Which ligands are most effective for achieving high enantiomeric excess in this process?
A: The patent highlights chiral diphosphine ligands such as Segphos, DM-Segphos, DTBM-Segphos, BINAP, and TunePhos derivatives, which consistently deliver enantiomeric excess values exceeding 90% and often reaching up to 99% ee.
Q: Is this process scalable for industrial manufacturing of API intermediates?
A: Yes, the process has been demonstrated on a gram scale (Example 19) with high yields (94%) and maintains high enantioselectivity, indicating strong potential for commercial scale-up due to the use of robust catalytic systems and simple workup procedures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this ruthenium-catalyzed technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical materials. Our state-of-the-art facilities are equipped to handle high-pressure hydrogenation reactions safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We are committed to leveraging advanced catalytic technologies to drive down costs and accelerate timelines for our global partners.
We invite pharmaceutical companies and research institutions to collaborate with us to explore the full potential of this synthesis route for their specific projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production needs, demonstrating exactly how this method can optimize your supply chain. Please contact our technical procurement team today to request specific COA data and route feasibility assessments for your target chiral amine compounds.
