Scalable Synthesis of High-Performance Phosphorus-Nitrogen Flame Retardants for Engineering Plastics
Scalable Synthesis of High-Performance Phosphorus-Nitrogen Flame Retardants for Engineering Plastics
The global shift towards sustainable and high-performance materials has intensified the demand for advanced halogen-free flame retardants that do not compromise the mechanical integrity of engineering polymers. Patent CN110643066B introduces a breakthrough in this domain by disclosing a novel class of phosphorus-nitrogen synergistic flame retardants derived from 9,10-dihydro-9-oxa-10-phosphaphenanthrene-10-oxide (DOPO) scaffolds. Unlike traditional additive flame retardants that often suffer from migration issues or require excessive loading levels that weaken the host material, this invention utilizes a unique molecular architecture designed to maximize char formation and gas phase inhibition. The core innovation lies in the strategic combination of high-phosphorus content DOPO derivatives with nitrogen-rich triallylamine structures, creating a robust additive capable of achieving UL-94 V0 ratings at remarkably low concentrations. 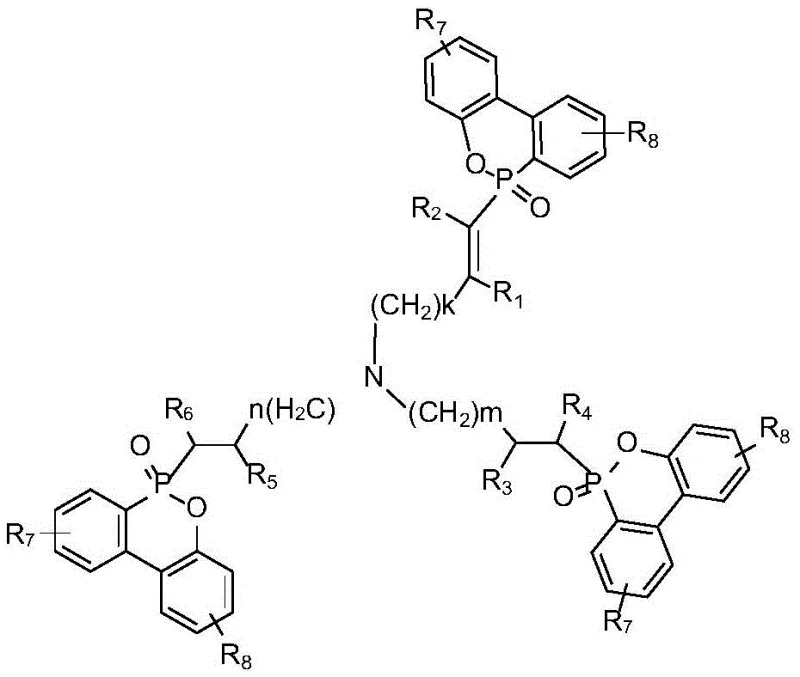
For procurement specialists and supply chain managers seeking a reliable polymer additive supplier, this technology represents a significant opportunity for cost reduction in polymer additive manufacturing. The synthesis pathway described in the patent is exceptionally streamlined, operating via a one-step Michael addition reaction that requires only thermal energy and absolutely no catalyst. This simplicity translates directly into operational efficiency, as it removes the complex purification stages typically associated with transition-metal catalyzed processes. By leveraging the high reactivity of the P-H bond within the DOPO structure, the process ensures high conversion rates and minimal byproduct formation, making it an ideal candidate for large-scale industrial production where consistency and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to flame retardancy have long been plagued by a trade-off between fire safety and material performance. Conventional halogenated flame retardants, while effective, are increasingly restricted due to the release of corrosive and toxic gases such as hydrogen halides and dioxins during combustion, posing severe environmental and health risks. On the other hand, many existing halogen-free alternatives, particularly simple phosphorus-based additives, often require very high loading levels—sometimes exceeding 20 to 30 weight percent—to achieve acceptable fire resistance. Such high loading acts as a plasticizer or a stress concentrator within the polymer matrix, drastically reducing tensile strength, impact resistance, and thermal stability. Furthermore, additive-type flame retardants that lack chemical bonding capability with the polymer chain are prone to migration and leaching over time, leading to a gradual loss of flame retardant efficacy and potential surface blooming issues that ruin the aesthetic and functional quality of the final product.
The Novel Approach
The methodology outlined in patent CN110643066B fundamentally disrupts this status quo by introducing a reactive-style additive that mimics the permanence of reactive flame retardants while retaining the processing ease of additive types. By synthesizing a molecule that integrates both phosphorus and nitrogen functionalities into a single, sterically bulky structure, the invention leverages a powerful synergistic effect. The phosphorus component promotes the formation of a stable, insulating polyphosphoric acid char layer on the polymer surface, while the nitrogen component releases inert gases like ammonia and nitrogen upon heating, which dilutes combustible volatiles and suffocates the flame. Crucially, this high-efficiency mechanism allows for effective flame retardancy at loading levels as low as 5 wt%, thereby preserving the intrinsic mechanical properties of the base resin. The one-step synthesis further distinguishes this approach, eliminating the need for multi-stage reactions and harsh reagents, which significantly lowers the barrier to entry for commercial adoption.
Mechanistic Insights into Catalyst-Free Michael Addition
The chemical elegance of this synthesis lies in the exploitation of the nucleophilic character of the P-H bond found in DOPO derivatives. Under thermal conditions, typically ranging from 130°C to 140°C, the P-H bond becomes sufficiently activated to undergo a Michael addition reaction with the electron-deficient carbon-carbon double bonds present in triallylamine. This reaction proceeds through a concerted mechanism where the phosphorus atom attacks the beta-carbon of the allyl group, resulting in the formation of a stable P-C bond without the generation of any small molecule byproducts. The absence of a catalyst is a critical feature, as it prevents the introduction of metal impurities that could catalyze unwanted degradation of the polymer matrix during subsequent processing or end-use. The reaction can be conducted either in a solvent system, such as xylene or toluene, to manage viscosity and heat transfer, or in a solvent-free molten state, which aligns perfectly with green chemistry principles by minimizing volatile organic compound (VOC) emissions. 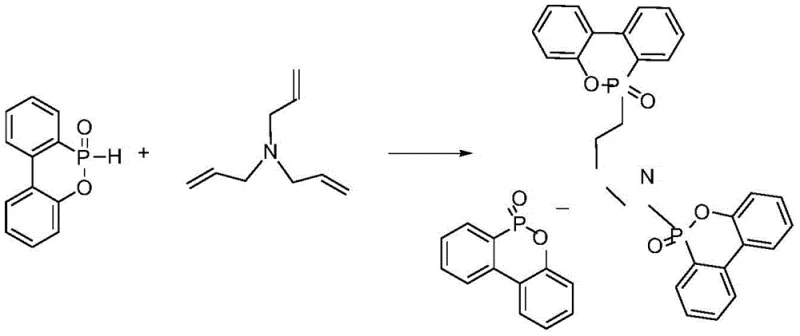
From an impurity control perspective, the specificity of the P-H addition to the alkene functionality ensures a clean reaction profile. Since the substituent groups on the aromatic rings of the DOPO derivative and the alkyl chains of the amine do not participate in the reaction, there is minimal risk of side reactions such as polymerization of the allyl groups or oxidation of the phosphorus center, provided an inert atmosphere is maintained. The resulting product is a well-defined oligomeric or monomeric species depending on the stoichiometry, which can be easily purified by simple filtration and washing. This high level of chemical control is essential for R&D directors who require consistent batch-to-batch reproducibility. The structural integrity of the P-O-C and P=O bonds in the final product ensures thermal stability up to nearly 300°C, allowing the additive to withstand the high processing temperatures of engineering thermoplastics like polycarbonate and polyphenyl ether without decomposing prematurely.
How to Synthesize Phosphorus-Nitrogen Flame Retardant Efficiently
The synthesis protocol described in the patent offers a robust framework for producing this high-value additive with minimal equipment requirements. The process begins by charging the DOPO derivative and a portion of the triallylamine into a reactor under nitrogen protection, followed by heating to initiate the melting or dissolution phase. Once the system reaches the target temperature, the remaining amine is added gradually to control the exotherm and ensure complete conversion.
- Charge DOPO derivative and triallylamine into an inert gas-protected reactor, optionally using a solvent like xylene or operating in a molten state.
- Heat the mixture to reflux or maintain temperature (e.g., 130-140°C) and drip the remaining amine reactant while stirring constantly.
- Cool the reaction mixture, filter the precipitate, wash thoroughly with solvents like tetrahydrofuran or ethanol, and dry to obtain the final powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this technology are profound, primarily driven by the drastic simplification of the manufacturing workflow. The elimination of catalysts not only reduces raw material costs but also removes the necessity for expensive downstream purification steps such as column chromatography or complex aqueous workups designed to strip metal residues. This streamlined process significantly shortens the production cycle time, allowing for faster turnover and improved responsiveness to market demand. Furthermore, the ability to operate in a molten state without solvents presents a compelling argument for cost reduction in polymer additive manufacturing, as it negates the capital and operational expenditures associated with solvent recovery systems, distillation columns, and VOC abatement technologies. The high yield reported in the examples, reaching up to 87 percent, further underscores the material efficiency of the process, ensuring that valuable raw materials are converted into saleable product rather than waste.
- Cost Reduction in Manufacturing: The catalyst-free nature of this Michael addition reaction fundamentally alters the cost structure of flame retardant production. By removing the dependency on precious metal catalysts or complex organocatalysts, the direct material cost is significantly lowered. Additionally, the simplified purification process—often requiring only filtration and drying—reduces labor hours, energy consumption, and waste disposal fees. This lean manufacturing approach allows suppliers to offer competitive pricing while maintaining healthy margins, providing a distinct advantage in price-sensitive markets like consumer electronics and automotive components where every cent counts in the bill of materials.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically DOPO and triallylamine, are commodity chemicals with well-established global supply chains. Unlike specialized intermediates that may be sourced from single suppliers and subject to geopolitical risks or capacity constraints, these feedstocks are produced by multiple manufacturers worldwide. This diversity in sourcing options mitigates the risk of supply disruptions and provides procurement teams with the leverage to negotiate better terms. Moreover, the robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility quality or operator error, ensuring a steady and reliable flow of finished goods to meet Just-In-Time delivery schedules.
- Scalability and Environmental Compliance: The scalability of this process is exceptional, transitioning seamlessly from laboratory benchtop scales to multi-ton industrial reactors. The option to run the reaction solvent-free is particularly advantageous for meeting increasingly stringent environmental regulations regarding VOC emissions and hazardous waste generation. Facilities can expand production capacity without needing to invest heavily in additional environmental control infrastructure. This 'green' credential is not merely a regulatory checkbox but a valuable marketing asset for downstream customers who are under pressure to reduce the carbon footprint of their supply chains and achieve sustainability goals such as LEED certification or compliance with eco-label standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this flame retardant technology. These insights are derived directly from the experimental data and process descriptions within the patent documentation, providing a factual basis for decision-making.
Q: What is the primary advantage of this phosphorus-nitrogen flame retardant over traditional halogenated options?
A: This novel additive offers a halogen-free solution that eliminates toxic smoke generation during combustion. Furthermore, the synergistic effect between phosphorus and nitrogen allows for significantly lower loading rates (as low as 5 wt%) to achieve UL-94 V0 ratings, thereby preserving the mechanical strength of the base polymer which is often compromised by high filler loads.
Q: Does the synthesis process require expensive transition metal catalysts?
A: No, the patented process relies on a thermal Michael addition reaction that proceeds efficiently without any catalyst. This eliminates the need for costly catalyst removal steps and prevents potential metal contamination in the final polymer matrix, simplifying the purification workflow and reducing overall production costs.
Q: Which polymer matrices are compatible with this new flame retardant technology?
A: The technology demonstrates excellent compatibility and efficacy in a wide range of engineering thermoplastics and thermosets, including polylactic acid (PLA), polycarbonate (PC), polyphenyl ether (PPE), epoxy resins, and polyurethane. It achieves high flame retardancy levels without significantly degrading the tensile strength of these materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphorus-Nitrogen Flame Retardant Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced additives play in the next generation of high-performance polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that consistency is key; therefore, our rigorous QC labs and stringent purity specifications guarantee that every batch of flame retardant meets the exacting standards required for aerospace, automotive, and electronic applications. Our commitment to technical excellence means we can support your R&D efforts with custom modifications to the core structure to optimize compatibility with specific polymer matrices.
We invite you to collaborate with us to unlock the full potential of this catalyst-free flame retardant technology for your product lines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current formulation needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your supply chain resilience and drive down your total cost of ownership.
