Advanced Synthesis of 1,3-Bis(2-hydroxyethyl)imidazolium Chloride for High-Performance Conductive Materials
Advanced Synthesis of 1,3-Bis(2-hydroxyethyl)imidazolium Chloride for High-Performance Conductive Materials
The rapid evolution of the electronic materials sector demands precursors that offer superior electrochemical stability and tunable functionality. Patent CN101250160A introduces a groundbreaking methodology for the synthesis of 1,3-bis(2-hydroxyethyl)imidazolium chloride ([hhim]Cl), a symmetric functional ionic liquid designed specifically for next-generation conductive and polyelectrolyte materials. Unlike traditional ionic liquids that often suffer from limited functionalization sites, this novel compound features symmetrical hydroxyethyl groups that significantly enhance its utility in polymer modification. The patent outlines a robust sodium-catalyzed alkylation process that operates under controlled low-temperature conditions to maximize yield while minimizing impurity formation. This technical breakthrough addresses the critical industry need for high-purity ionic intermediates that can withstand the rigorous demands of energy storage and electronic device manufacturing.
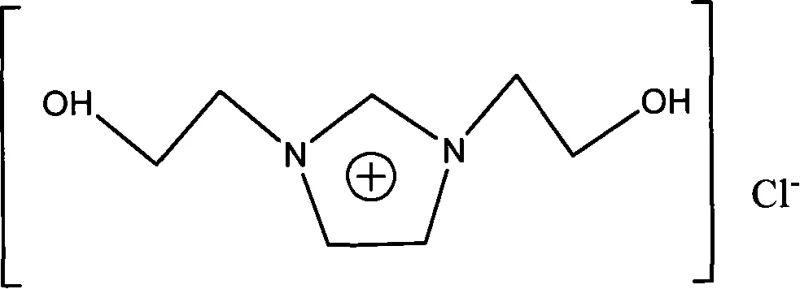
The structural integrity of the [hhim]Cl cation is paramount for its performance in solid electrolytes, where ion transport mechanisms rely heavily on molecular symmetry and functional group availability. The presence of two hydroxyl groups allows for extensive hydrogen bonding networks, which can be leveraged to improve the mechanical stability of polymer matrices without sacrificing ionic conductivity. Furthermore, the chloride counter-ion provides a balance of solubility and electrochemical stability that is essential for wide-window applications. By adopting this patented synthesis route, manufacturers can secure a reliable ionic liquid supplier source that delivers materials with verified electrochemical windows ranging from 2.0V to 2.4V. This level of performance validation is crucial for R&D directors seeking to integrate new materials into battery systems or conductive coatings where failure is not an option.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of functional ionic liquids has been dominated by monofunctional derivatives, such as 1-methyl-3-substituted imidazolium salts, which inherently limit the scope of chemical modification. These conventional materials typically possess only one reactive site, restricting their ability to cross-link or integrate effectively into complex polymer chains required for advanced solid electrolytes. The synthesis of these older generations often involves harsh conditions or expensive catalysts that introduce heavy metal contaminants, necessitating costly purification steps that erode profit margins. Additionally, the lack of symmetry in monofunctional ions can lead to inconsistent packing in the solid state, resulting in variable conductivity and thermal stability issues that plague mass production efforts. For procurement managers, relying on these legacy chemistries often means dealing with supply chain volatility and inconsistent batch-to-batch quality that complicates regulatory compliance in sensitive electronic applications.
The Novel Approach
The patented process described in CN101250160A revolutionizes this landscape by introducing a symmetric bis-functionalization strategy that doubles the reactive potential of the imidazolium core. By utilizing a sodium-catalyzed pathway in absolute ethanol, the method achieves high selectivity for the 1,3-disubstituted product without requiring exotic reagents or extreme pressures. The reaction proceeds through a controlled two-stage temperature profile, starting at cryogenic conditions (-1 to 2°C) to manage the exotherm of alkoxide formation, followed by a sustained reaction phase at 10-60°C to drive the double alkylation to completion. This approach not only simplifies the operational complexity but also ensures that the resulting ionic liquid possesses the precise structural symmetry needed for optimal electrochemical performance.
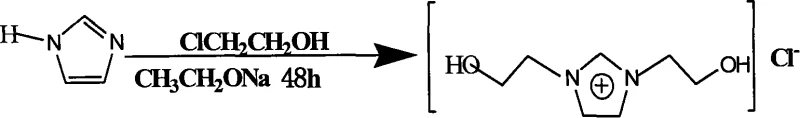
Furthermore, the novel approach incorporates a sophisticated purification regimen involving activated carbon decolorization and silica gel chromatography, which effectively removes trace organic impurities and unreacted starting materials. This attention to detail in the downstream processing ensures that the final product meets the stringent purity specifications required for high-end electronic chemical manufacturing. The ability to crystallize the product at low temperatures (0 to -2°C) further enhances the physical form of the material, making it easier to handle, transport, and dose in subsequent polymerization reactions. For supply chain heads, this translates to a more robust and predictable manufacturing process that reduces the risk of production delays caused by off-spec materials or complex purification bottlenecks.
Mechanistic Insights into Sodium-Catalyzed N-Alkylation
The core of this synthesis lies in the in situ generation of sodium ethoxide, which acts as a potent base to deprotonate the imidazole nitrogen atoms, creating a highly nucleophilic imidazolide species. This mechanistic pathway is superior to direct alkylation because it avoids the accumulation of acidic byproducts that can catalyze decomposition or polymerization of the sensitive 2-chloroethanol reagent. The use of metallic sodium in absolute ethanol ensures a dry, oxygen-free environment that is critical for preventing the oxidation of the hydroxyl groups or the formation of ether linkages as side products. By maintaining the reaction mixture at -1 to 2°C during the initial catalyst formation and reagent addition, the process kinetically favors the desired N-alkylation over competing O-alkylation or elimination reactions that could degrade the yield. This precise control over the reaction kinetics is what allows the process to achieve yields consistently above 70%, a significant improvement over unoptimized batch methods.
Impurity control is managed through a combination of stoichiometric precision and physical separation techniques. The patent specifies a molar ratio of imidazole to 2-chloroethanol between 1:2.1 and 1:2.2, providing a slight excess of the alkylating agent to drive the second alkylation step to completion without leaving significant amounts of the mono-alkylated intermediate. Following the reaction, the removal of ethanol and unreacted chloroethanol under reduced pressure concentrates the ionic liquid, while the subsequent washing with ethyl acetate removes non-polar organic impurities. The final purification via silica gel column chromatography using a methanol-chloroform eluent system is particularly effective at separating the target cation from any quaternary ammonium byproducts or colored degradation species. This multi-stage purification strategy ensures that the final [hhim]Cl product exhibits the high ionic conductivity (0.5-0.7 s/m) necessary for its intended application in conductive materials.
How to Synthesize 1,3-Bis(2-hydroxyethyl)imidazolium Chloride Efficiently
The synthesis of this high-value ionic liquid requires strict adherence to the patented temperature profiles and reagent addition rates to ensure both safety and product quality. The process begins with the careful preparation of the catalytic system, followed by the sequential addition of heterocyclic and alkyl halide components under inert atmosphere conditions. Detailed operational parameters, including specific stirring speeds and vacuum levels for solvent removal, are critical for reproducing the high yields reported in the patent examples. For technical teams looking to implement this route, it is essential to recognize that the crystallization step is not merely a purification tactic but a critical unit operation that defines the physical morphology of the final product. The detailed standardized synthesis steps below outline the precise execution required to achieve commercial-grade quality.
- Prepare sodium ethoxide catalyst by reacting sodium metal with absolute ethanol at low temperatures (-1 to 2°C) to ensure safety and reagent stability.
- Perform double N-alkylation by adding imidazole followed by 2-chloroethanol (molar ratio 1: 2.1), maintaining strict temperature control during the exothermic addition.
- Purify the crude ionic liquid through activated carbon decolorization, silica gel column chromatography, and low-temperature crystallization to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers substantial strategic advantages for organizations focused on cost reduction in electronic chemical manufacturing and supply chain resilience. The reliance on commodity chemicals such as imidazole, 2-chloroethanol, and sodium metal eliminates the dependency on scarce or geopolitically sensitive raw materials that often plague the specialty chemical sector. This foundational simplicity in the bill of materials allows for significant cost optimization, as the input costs are stable and predictable compared to complex organometallic catalysts. Moreover, the elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which drastically simplifies the downstream processing workflow. This streamlined approach reduces the overall cycle time from raw material intake to finished goods, enabling faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and widely available reagents, avoiding the high capital expenditure associated with specialized catalytic systems. By operating at atmospheric pressure and moderate temperatures (10-60°C), the energy consumption is kept to a minimum, directly lowering the utility costs per kilogram of product. The high selectivity of the reaction minimizes waste generation, reducing the burden on waste treatment facilities and lowering disposal costs. Additionally, the ability to recover and recycle the ethanol solvent further enhances the economic efficiency of the process, contributing to a lower total cost of ownership for the manufacturing asset.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthesis is straightforward, as imidazole and chloroethanol are produced at scale by multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, ensuring consistent output even when supply chains face disruptions. The crystallization step produces a stable solid or viscous liquid that is easier to store and transport than many hygroscopic or thermally unstable ionic liquids, simplifying logistics and warehousing requirements. This stability extends the shelf life of the product, allowing for larger batch production runs that optimize inventory management and reduce the frequency of changeovers.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, moving seamlessly from laboratory glassware to industrial reactors without requiring fundamental changes to the chemistry or equipment design. The use of ethanol as a primary solvent aligns with green chemistry principles, as it is biodegradable and less toxic than many chlorinated or aromatic solvents traditionally used in fine chemical synthesis. The process generates minimal hazardous waste, primarily consisting of aqueous salt solutions and organic washes that can be treated using standard effluent protocols. This environmental profile facilitates easier regulatory approval and permits, accelerating the time to market for new products derived from this ionic liquid platform.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3-bis(2-hydroxyethyl)imidazolium chloride. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this material into your existing product lines or R&D pipelines. We encourage technical stakeholders to review these points carefully to assess the alignment with their specific project requirements.
Q: What are the key advantages of symmetric ionic liquids over monofunctional variants?
A: Symmetric ionic liquids like [hhim]Cl possess dual functional groups that offer more modification sites for polymer chains, significantly enhancing the stability and conductivity of resulting solid electrolytes compared to traditional monofunctional analogues.
Q: How does the sodium-catalyzed method improve process safety and yield?
A: By generating the active alkoxide species in situ at low temperatures (-1 to 2°C), the process minimizes side reactions and thermal runaway risks, leading to consistent yields between 70% and 80% with simplified downstream processing.
Q: What represent the critical quality parameters for this ionic liquid in electronic applications?
A: For conductive material applications, the critical parameters include a wide electrochemical window (2.0-2.4V) and high ionic conductivity (0.5-0.7 s/m), both of which are rigorously validated in the patented synthesis protocol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3-Bis(2-hydroxyethyl)imidazolium Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance ionic liquids play in the advancement of electronic materials and energy storage technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the electrochemical and physical standards defined in the patent. Our commitment to excellence means that every gram of [hhim]Cl we deliver is backed by comprehensive data packages that validate its suitability for your specific conductive material applications.
We invite you to collaborate with us to explore how this innovative synthesis route can drive value in your operations. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current material usage. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals. Let us be your partner in transforming advanced chemical concepts into commercial reality.
