Advanced Photoredox Synthesis of 1,1-Diarylalkane Derivatives for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures with high efficiency and minimal environmental impact. Patent CN110256211B introduces a groundbreaking preparation method for 1,1-diarylalkane derivatives, a structural motif prevalent in numerous bioactive molecules and therapeutic agents. This technology leverages a sophisticated photoredox catalytic system to achieve the 1,2-difunctionalization of olefins, constructing both C(sp3)-C(sp3) and C(sp3)-C(sp2) bonds in a single operational step. By utilizing readily available olefin compounds, (hetero)arenes, and N-hydroxyphthalimide (NHP) esters, this invention represents a significant leap forward in synthetic organic chemistry. The ability to generate alkyl radicals from stable carboxylic acid derivatives under mild visible light irradiation eliminates the need for hazardous reagents and extreme conditions traditionally associated with such transformations. For R&D directors and process chemists, this patent offers a viable pathway to access high-purity intermediates with superior impurity profiles, addressing the critical need for scalable and sustainable manufacturing processes in the modern chemical supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,1-diarylalkane derivatives has relied heavily on classical Friedel-Crafts alkylation or transition-metal-catalyzed cross-coupling reactions, which present substantial drawbacks for commercial manufacturing. Traditional Friedel-Crafts reactions often require stoichiometric amounts of strong Lewis acids or Brønsted acids, leading to significant waste generation and difficult downstream purification processes due to the formation of polyalkylated byproducts. Furthermore, transition-metal-catalyzed approaches frequently necessitate the use of expensive and toxic heavy metals, such as palladium or nickel, along with sensitive organometallic reagents that demand strict anhydrous and anaerobic conditions. These conventional strategies often suffer from poor functional group tolerance, limiting the scope of substrates that can be utilized without extensive protecting group manipulation. The harsh reaction conditions, including high temperatures and pressures, not only increase energy consumption but also pose safety risks during scale-up. Consequently, procurement managers and supply chain heads face challenges in sourcing cost-effective raw materials and ensuring consistent quality when relying on these outdated synthetic routes.
The Novel Approach
In stark contrast, the novel approach disclosed in CN110256211B utilizes a dual catalytic system comprising a photoredox catalyst and a Lewis acid to drive the reaction under exceptionally mild conditions. This method employs N-hydroxyphthalimide esters as versatile alkylating agents, which can be easily prepared from abundant and inexpensive carboxylic acids, thereby drastically simplifying the raw material supply chain. The reaction proceeds at temperatures ranging from 20°C to 40°C under blue LED illumination, eliminating the thermal stress on sensitive functional groups and reducing the energy footprint of the manufacturing process. The synergy between the photocatalyst, which generates alkyl radicals via single-electron transfer, and the Lewis acid, which activates the electrophilic species, ensures high regioselectivity and yield. This innovative strategy allows for the direct coupling of unactivated olefins with heteroarenes, bypassing the need for pre-functionalized substrates. 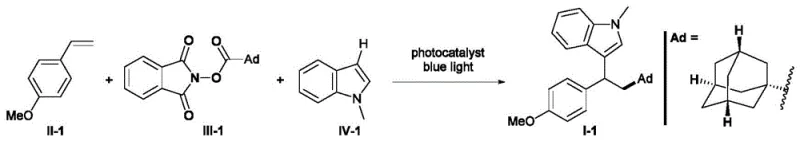 By streamlining the synthetic sequence into a single pot, this approach significantly reduces processing time and solvent usage, offering a compelling value proposition for cost reduction in fine chemical manufacturing.
By streamlining the synthetic sequence into a single pot, this approach significantly reduces processing time and solvent usage, offering a compelling value proposition for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Photoredox-Catalyzed Olefin Difunctionalization
The core of this technological advancement lies in the intricate mechanistic pathway that facilitates the simultaneous formation of carbon-carbon bonds through a radical-polar crossover mechanism. Upon irradiation with visible light, the excited state of the iridium-based photocatalyst, such as fac-Ir(ppy)3, undergoes a single-electron transfer with the N-hydroxyphthalimide ester. This redox event triggers the fragmentation of the N-O bond, resulting in the release of carbon dioxide and the generation of a highly reactive alkyl radical species. This alkyl radical subsequently adds across the double bond of the olefin substrate in a Markovnikov or anti-Markovnikov fashion, depending on the electronic nature of the substituents, to form a new stabilized carbon-centered radical intermediate. The presence of the Lewis acid, preferably B(C6F5)3, plays a crucial role in activating the heteroarene nucleophile, enhancing its reactivity towards the radical intermediate. 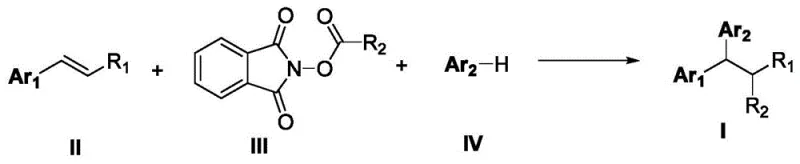 This cascade process culminates in the formation of the final 1,1-diarylalkane product after proton transfer and rearomatization, effectively achieving a 1,2-difunctionalization of the original olefin. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters and expand the substrate scope to include more complex molecular scaffolds required for drug discovery programs.
This cascade process culminates in the formation of the final 1,1-diarylalkane product after proton transfer and rearomatization, effectively achieving a 1,2-difunctionalization of the original olefin. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters and expand the substrate scope to include more complex molecular scaffolds required for drug discovery programs.
From an impurity control perspective, this mechanism offers distinct advantages over ionic pathways that are prone to rearrangement and elimination side reactions. The radical nature of the transformation ensures that the reaction proceeds through discrete intermediates that are less likely to undergo skeletal rearrangements common in carbocation-mediated processes. The mild conditions prevent the degradation of sensitive functional groups such as esters, nitriles, and halides, which are often incompatible with strong acidic or basic environments. Moreover, the high chemoselectivity of the photoredox system minimizes the formation of homocoupling byproducts or oligomers, leading to a cleaner crude reaction profile. This inherent purity reduces the burden on downstream purification units, such as crystallization or chromatography, thereby improving the overall mass balance and yield of the active pharmaceutical ingredient. For quality assurance teams, the predictability of this radical mechanism translates to more robust process validation and consistent batch-to-batch reproducibility.
How to Synthesize 1,1-Diarylalkane Derivatives Efficiently
To implement this synthesis route effectively, operators must adhere to precise protocols regarding reagent stoichiometry and environmental controls to maximize yield and safety. The process begins with the careful weighing of the olefin, N-hydroxyphthalimide ester, and heteroarene substrates according to the optimized molar ratios identified in the patent examples. It is critical to maintain an inert atmosphere throughout the reaction to prevent the quenching of the excited photocatalyst by molecular oxygen, which would otherwise inhibit the radical generation cycle. The choice of solvent, specifically polar aprotic solvents like DMA or DMF, is essential to solubilize the ionic intermediates and facilitate the electron transfer processes. While the general procedure is straightforward, attention to detail in the setup of the photoreactor and the monitoring of reaction progress via TLC or GC-MS is necessary to ensure complete conversion. Detailed standardized synthesis steps are provided below to guide your technical team in replicating this high-efficiency transformation.
- Charge a Schlenk tube with olefin compound, N-hydroxyphthalimide ester, heteroarene substrate, fac-Ir(ppy)3 photocatalyst, and B(C6F5)3 Lewis acid in DMA solvent.
- Replace the atmosphere with argon gas to ensure inert conditions and seal the reactor tightly to prevent oxygen quenching of the radical species.
- Illuminate the reaction mixture with a 10W blue LED light source while stirring at 30°C for 24 hours, then purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this photoredox methodology presents a strategic opportunity to optimize costs and enhance supply reliability. The shift from expensive organometallic reagents to stable carboxylic acid derivatives significantly lowers the raw material expenditure, as carboxylic acids are commodity chemicals available in bulk quantities from diverse global suppliers. The elimination of cryogenic conditions and the ability to run reactions at near-ambient temperatures result in substantial energy savings, reducing the operational overhead associated with heating and cooling infrastructure. Furthermore, the simplified workup procedure, which involves standard extraction and chromatography, minimizes the consumption of specialized consumables and reduces the volume of hazardous waste requiring disposal. These factors collectively contribute to a more lean and agile manufacturing process that can adapt quickly to fluctuating market demands without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The utilization of earth-abundant starting materials and the avoidance of precious metal catalysts in stoichiometric amounts lead to significant cost savings in the overall production budget. By replacing complex multi-step sequences with a concise one-pot reaction, labor costs and equipment occupancy time are drastically reduced, allowing for higher throughput in existing facilities. The high atom economy of the difunctionalization reaction ensures that a greater proportion of the input mass is converted into the desired product, minimizing waste disposal fees and maximizing resource efficiency. Additionally, the robustness of the catalytic system reduces the frequency of catalyst replacement and maintenance, further lowering the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing stability is greatly improved as the key reagents, such as styrenes and NHP esters, are derived from widely available feedstocks rather than specialized custom synthons. This diversification of the supply base mitigates the risk of shortages caused by geopolitical issues or single-source dependencies, ensuring continuous production schedules. The mild reaction conditions also reduce the wear and tear on reactor vessels and piping, extending the lifespan of capital equipment and minimizing unplanned downtime due to maintenance. Consequently, supply chain heads can forecast lead times with greater accuracy and maintain lower safety stock levels, freeing up working capital for other strategic investments within the organization.
- Scalability and Environmental Compliance: The protocol is inherently designed for scalability, with linear heat and mass transfer characteristics that facilitate the transition from laboratory benchtop to pilot plant and full commercial scale. The absence of toxic heavy metals and volatile organic solvents in excessive amounts aligns with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile enhances the company's reputation among eco-conscious partners and customers, potentially opening up new market segments that prioritize sustainable sourcing. The ease of waste treatment due to the benign nature of the byproducts simplifies regulatory compliance and reduces the administrative burden associated with environmental reporting and auditing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios. The responses highlight the versatility of the method and its alignment with modern industry standards for safety, efficiency, and quality.
Q: What are the primary limitations of conventional alkylation methods addressed by this patent?
A: Conventional methods often rely on harsh reaction conditions, expensive transition metal catalysts, or pre-functionalized organometallic reagents that limit functional group tolerance. This patent overcomes these issues by utilizing mild photoredox conditions and stable N-hydroxyphthalimide esters derived from cheap carboxylic acids.
Q: How does the photoredox system improve impurity control compared to thermal methods?
A: The use of visible light irradiation at low temperatures (20-40°C) minimizes thermal decomposition pathways and side reactions common in high-temperature thermal processes. The specific combination of Ir-based photocatalysts and Lewis acids ensures high selectivity for the desired C(sp3)-C(sp2) bond formation.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the protocol uses commercially available raw materials like styrenes and carboxylic acid derivatives, operates at near-ambient temperatures, and avoids cryogenic conditions. These factors significantly enhance the feasibility of scaling the process from laboratory grams to multi-kilogram commercial batches.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1-Diarylalkane Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced photoredox technologies in accelerating the development of next-generation pharmaceuticals and fine chemicals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial realities. Our state-of-the-art facilities are equipped with specialized photoreactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. We are committed to delivering high-purity 1,1-diarylalkane derivatives that empower your R&D teams to push the boundaries of drug discovery while maintaining the highest standards of quality and consistency.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive efficiency and innovation in your supply chain.
