Advanced Metal-Free Synthesis of 3-Sulfonylspiro[4,5]trienones for Pharmaceutical Applications
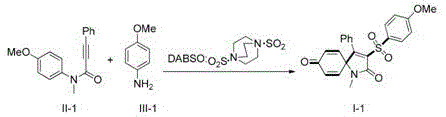
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. A recent technological breakthrough disclosed in patent CN110551059B introduces a highly efficient, transition-metal-free strategy for the synthesis of 3-sulfonylspiro[4,5]trienone compounds. These spirocyclic structures are increasingly recognized for their presence in numerous natural products and drug candidates, yet their traditional synthesis often involves harsh conditions or expensive reagents. This novel approach leverages visible-light photocatalysis to drive a radical sulfonylation/ipso-cyclization cascade, utilizing DABSO (DABCO·(SO2)2) as a convenient solid source of sulfur dioxide and readily available anilines as the aryl precursors. By shifting away from gaseous SO2 and unstable diazonium salts, this method offers a safer, more operationally simple pathway that aligns perfectly with modern green chemistry principles and the stringent purity requirements of global pharmaceutical supply chains.
![General reaction scheme for the synthesis of 3-sulfonylspiro[4,5]trienones using DABSO and anilines](/insights/img/3-sulfonylspiro-trienone-synthesis-pharma-supplier-20260306051110-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of sulfone-containing spirocycles has relied heavily on the use of sulfonyl chlorides or the oxidation of thioethers, both of which present significant logistical and chemical challenges for large-scale manufacturing. Sulfonyl chlorides, while reactive, are often moisture-sensitive, corrosive, and generate stoichiometric amounts of acidic waste that require careful neutralization and disposal, increasing the environmental footprint of the process. Furthermore, alternative strategies involving the insertion of sulfur dioxide often necessitate the use of gaseous SO2, which poses severe safety hazards regarding toxicity and requires specialized pressurized equipment that is not universally available in standard pilot plants. Additionally, many existing protocols depend on transition metal catalysts such as palladium or copper, which introduce the risk of heavy metal contamination in the final product, necessitating costly and time-consuming purification steps to meet regulatory limits for pharmaceutical intermediates. These factors collectively contribute to higher production costs, longer lead times, and reduced overall process reliability for manufacturers relying on legacy synthetic routes.
The Novel Approach
In stark contrast, the methodology described in CN110551059B revolutionizes this transformation by employing a visible-light-driven radical mechanism that operates under mild thermal conditions. As illustrated in the optimization studies, the reaction proceeds efficiently using inexpensive organic photocatalysts like Eosin Y or even under catalyst-free conditions with sufficient light intensity, completely eliminating the need for precious transition metals. The use of DABSO as a solid SO2 surrogate allows for precise stoichiometric control without the handling risks associated with toxic gases, while the use of anilines as the aryl source bypasses the need for pre-functionalized boronic acids or unstable diazonium salts. This one-pot protocol combines sulfonylation and cyclization into a single operational step, significantly reducing solvent consumption and processing time. The reaction tolerates a wide range of functional groups, including halogens and alkoxy substituents, demonstrating exceptional versatility for generating diverse libraries of spiro-compounds for drug discovery programs.
Mechanistic Insights into Visible-Light Driven Radical Cyclization
The core of this innovative synthesis lies in its elegant radical cascade mechanism, which is initiated by the interaction of light energy with the reaction components to generate highly reactive intermediates under mild conditions. Upon irradiation with blue LEDs, the photocatalyst (or the substrate itself in catalyst-free variants) facilitates the single-electron transfer processes necessary to convert the aniline starting material into an aryl radical species via an in situ generated diazonium intermediate. This aryl radical subsequently captures sulfur dioxide released from the DABSO adduct, forming a transient arylsulfonyl radical. This key sulfonyl radical then undergoes an intramolecular addition to the triple bond of the N-methylalkynamide, triggering a cascade of cyclization events that construct the rigid spiro[4,5]trienone skeleton. Crucially, isotopic labeling experiments confirmed that the oxygen atom in the newly formed carbonyl group originates from water added to the reaction mixture, highlighting the unique role of simple additives in driving the oxidative termination of the radical cycle. This mechanistic pathway avoids high-energy intermediates and harsh oxidants, ensuring high chemoselectivity and minimizing the formation of unwanted byproducts that complicate downstream purification.
From an impurity control perspective, this radical mechanism offers distinct advantages over ionic pathways that might suffer from competing nucleophilic attacks or rearrangement side reactions. The specificity of the radical addition to the alkyne moiety ensures that the spiro-center is formed with high fidelity, while the mild reaction temperature of 70°C prevents thermal degradation of sensitive functional groups often found in complex drug intermediates. The absence of transition metals further simplifies the impurity profile, as there are no metal-ligand complexes or metal-induced decomposition products to monitor or remove. This clean reaction profile translates directly into higher crude purity, reducing the burden on chromatographic separation steps and improving the overall mass balance of the manufacturing process. For R&D teams, understanding this mechanism allows for rational optimization of light intensity and photon flux to maximize quantum yield, ensuring that the process remains robust even when scaling from milligram to kilogram quantities.
How to Synthesize 3-Sulfonylspiro[4,5]trienone Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot setting, operators must carefully control the photon flux and reagent stoichiometry to ensure consistent reproducibility and high yields. The process begins with the sequential addition of the N-methylalkynamide, aniline derivative, DABSO, tert-butyl nitrite, and water into a sealed Schlenk tube under an inert argon atmosphere to prevent oxygen quenching of the radical species. Acetonitrile is selected as the optimal solvent due to its ability to dissolve all reactants while remaining transparent to the activating blue light wavelength. Once the mixture is prepared, the reactor is placed in an oil bath heated to 70°C and irradiated with a 5W blue LED source for approximately 24 hours, allowing the radical cascade to reach completion as monitored by TLC or GC-MS analysis.
- Charge a Schlenk tube with N-methylalkynamide, aniline derivative, DABSO, tBuONO, water, and acetonitrile solvent under inert atmosphere.
- Add photocatalyst (e.g., Eosin Y) and seal the reactor, then place it in an oil bath heated to 70°C under 5W blue LED irradiation.
- Stir for 24 hours, monitor by TLC/GC-MS, then perform aqueous workup and silica gel column chromatography to isolate the pure spiro compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DABSO-mediated photocatalytic route presents a compelling value proposition centered on cost efficiency, raw material security, and operational simplicity. By replacing expensive and hazardous sulfonyl chlorides or gaseous sulfur dioxide with stable, solid DABSO and commodity anilines, the raw material costs are significantly reduced while simultaneously mitigating safety risks associated with storage and handling. The elimination of transition metal catalysts removes the necessity for specialized scavenging resins or complex extraction protocols designed to lower heavy metal residues, thereby streamlining the purification workflow and reducing the consumption of silica gel and solvents. Furthermore, the reliance on visible light as the primary energy driver, rather than high-temperature heating or high-pressure reactors, lowers the energy intensity of the process and allows for the use of standard glass-lined reactors equipped with LED arrays, facilitating easier technology transfer from lab to plant.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with widely available commodity chemicals. Anilines are produced on a massive industrial scale for the dye and polymer industries, ensuring a stable and competitive pricing structure compared to specialized organoboron reagents or sulfonyl chlorides. Additionally, the use of organic photocatalysts like Eosin Y, which are orders of magnitude cheaper than iridium or ruthenium complexes, drastically lowers the catalyst cost per kilogram of product. The simplified workup procedure, which avoids metal scavenging and extensive washing steps, further reduces labor costs and waste disposal fees, contributing to a leaner overall cost of goods sold (COGS) structure for the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of robust, shelf-stable reagents that do not require cold chain logistics or specialized pressure vessels for transport. DABSO is a solid powder that can be stored indefinitely under ambient conditions, eliminating the supply risks associated with compressed gas cylinders or moisture-sensitive liquids that may degrade during shipping delays. The broad substrate scope demonstrated in the patent data indicates that the process is tolerant to variations in raw material quality, allowing procurement teams to source anilines and alkynamides from multiple qualified vendors without compromising reaction performance. This flexibility reduces the risk of single-source bottlenecks and ensures continuous production capability even during market fluctuations or geopolitical disruptions affecting specific chemical supply lines.
- Scalability and Environmental Compliance: Scaling this photochemical process is facilitated by the modular nature of LED lighting systems, which can be easily integrated into existing reactor setups or flow chemistry platforms to increase throughput without proportional increases in energy consumption. The reaction generates minimal hazardous waste, as the primary byproducts are benign nitrogen species and water, aligning with increasingly strict environmental regulations regarding solvent emissions and heavy metal discharge. The absence of corrosive acids or toxic gases simplifies the permitting process for new manufacturing lines and reduces the capital expenditure required for scrubber systems and corrosion-resistant equipment. This environmentally friendly profile not only lowers compliance costs but also enhances the sustainability credentials of the supply chain, a factor that is becoming increasingly critical for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light mediated synthesis for 3-sulfonylspiro[4,5]trienones. These insights are derived directly from the experimental data and optimization studies detailed in the patent literature, providing a factual basis for evaluating the technology's fit within your specific manufacturing context. Understanding these nuances is essential for R&D and operations teams planning to integrate this methodology into their process development pipelines.
Q: What are the advantages of using DABSO over traditional sulfonyl chlorides?
A: DABSO serves as a stable, solid surrogate for gaseous sulfur dioxide, eliminating the need for hazardous gas handling equipment and allowing for milder reaction conditions compared to the corrosive and moisture-sensitive nature of sulfonyl chlorides.
Q: Is transition metal removal required for this synthesis?
A: No, the process utilizes an organic photocatalyst like Eosin Y or operates under metal-free conditions with just light, significantly simplifying downstream purification and ensuring low heavy metal residues in the final API intermediate.
Q: What is the substrate scope for the aniline component?
A: The method demonstrates broad tolerance for various substituents on the aniline ring, including electron-donating groups like methoxy and electron-withdrawing groups like halogens, enabling the synthesis of diverse structural analogues.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Sulfonylspiro[4,5]trienone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free photocatalytic technology for accelerating the development of next-generation therapeutic agents. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-sulfonylspiro[4,5]trienone intermediate delivered meets the highest global quality standards. We are committed to leveraging our technical expertise to optimize this novel route for your specific project needs, ensuring maximum yield and minimal environmental impact.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this DABSO-based protocol compared to your current legacy methods. We encourage you to contact us today to obtain specific COA data for our reference standards and to schedule a consultation for detailed route feasibility assessments, ensuring your project moves forward with confidence and speed.
