Advanced One-Pot Synthesis of Relebactam Intermediates for Commercial Scale-Up
Introduction to Advanced Relebactam Manufacturing
The pharmaceutical landscape for beta-lactamase inhibitors is rapidly evolving, driven by the urgent need to combat multidrug-resistant bacterial infections. Central to this advancement is Relebactam (CAS 1174018-99-5), a potent diazabicyclooctanone compound developed to restore the efficacy of carbapenems against resistant strains such as Klebsiella pneumoniae and Pseudomonas aeruginosa. A pivotal breakthrough in the scalable production of this critical active pharmaceutical ingredient (API) is detailed in patent CN111943950A, which outlines a novel preparation method that fundamentally reimagines the synthetic pathway. Unlike traditional approaches that rely heavily on hazardous high-pressure hydrogenation and expensive starting materials, this new methodology leverages a sophisticated one-pot transfer hydrogenation and sulfonation strategy. For global procurement and R&D teams, this patent represents a significant shift towards safer, more cost-effective, and environmentally sustainable manufacturing protocols. By addressing the inherent instability of key intermediates and streamlining the purification workflow, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships aiming to secure long-term supply chains for next-generation antibiotics.
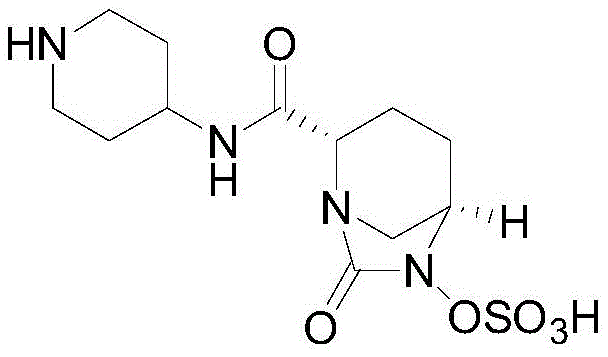
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Relebactam has been plagued by significant operational hurdles and safety concerns that hinder efficient commercial scale-up of complex intermediates. As documented in prior art such as WO2009091856 and CN105143224A, conventional routes typically commence with (2S,5R)-6-(benzyloxy)-7-oxo-1,6-diazabicyclo[3,2,1]octane-2-carboxylic acid, a starting material that is not only prohibitively expensive but also difficult to source in bulk quantities. Furthermore, these legacy processes invariably depend on catalytic hydrogenation using palladium on carbon under high-pressure hydrogen gas conditions to remove benzyl protecting groups. This requirement introduces severe safety risks, including the potential for explosive atmospheres and the need for specialized, high-cost pressure reactors. Additionally, the multi-step nature of these older pathways often involves the isolation of unstable N-hydroxy intermediates, which are prone to oxidative degradation, leading to lower yields, complex impurity profiles, and extensive waste generation that complicates environmental compliance.
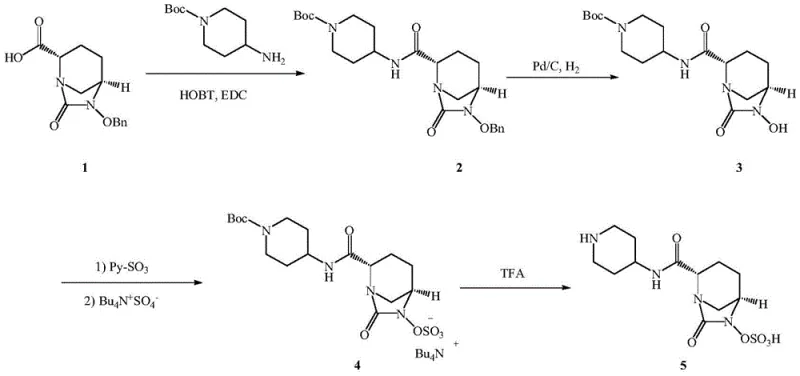
The Novel Approach
In stark contrast to these cumbersome legacy methods, the process disclosed in CN111943950A introduces a streamlined, five-step synthetic sequence that effectively bypasses the need for high-pressure infrastructure. The innovation begins with the utilization of a more accessible ethyl ester oxalate precursor, which undergoes mild deprotection and cyclization to form the core bicyclic structure. The true brilliance of this approach lies in the fourth step, where a telescoped one-pot reaction achieves debenzylation, sulfonation, and ammonium salt formation simultaneously. By replacing gaseous hydrogen with a silane-based hydrogen donor reagent, the process operates under atmospheric pressure, drastically simplifying the engineering controls required for production. This methodological shift not only enhances operator safety but also significantly improves the reaction selectivity, ensuring that the sensitive N-hydroxy species is immediately converted to the stable sulfate ester without prolonged exposure to oxidative conditions. The result is a robust, high-yielding pathway that delivers superior product quality while minimizing the generation of hazardous three wastes.
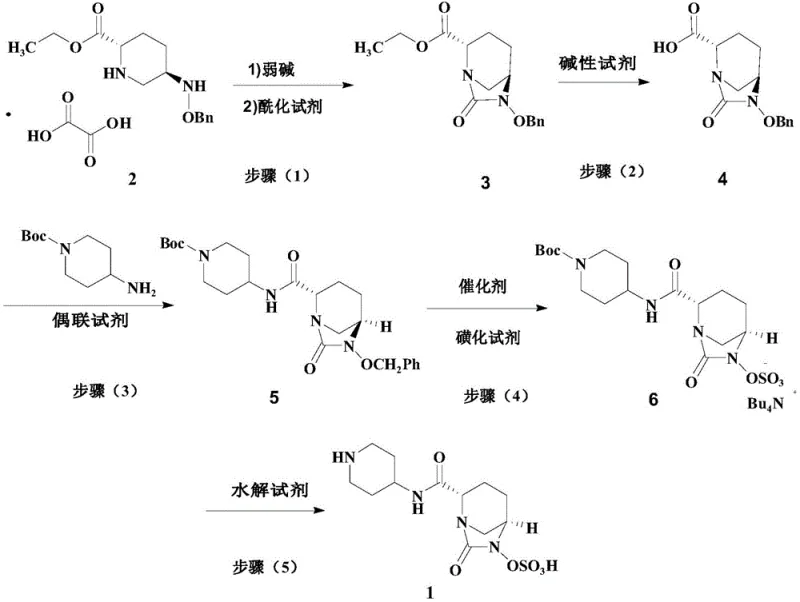
Mechanistic Insights into Transfer Hydrogenation and Sulfonation
The core chemical innovation driving the efficiency of this new process is the mechanistic elegance of the one-pot debenzylation and sulfonation sequence. In traditional catalytic hydrogenation, the removal of the benzyl group generates a reactive N-hydroxy intermediate that must be carefully managed to prevent decomposition. In the patented method, the addition of a silane hydrogen donor, such as methyldiethoxysilane, in the presence of a palladium catalyst facilitates a transfer hydrogenation mechanism. This allows for the cleavage of the benzyl ether bond under mild thermal conditions (10-20°C) without the kinetic energy spikes associated with high-pressure gas reactions. Crucially, the reaction mixture is not worked up; instead, a sulfonating reagent, specifically sulfur trioxide pyridine complex, is introduced directly into the same vessel. This immediate subsequent reaction captures the nascent N-hydroxy species, converting it into the thermodynamically stable sulfate ester before it can undergo any significant degradation or side reactions. This kinetic trapping strategy is essential for maintaining high purity levels, as it prevents the accumulation of colored impurities and oxidation byproducts that typically necessitate rigorous and yield-reducing chromatographic purifications in other synthetic routes.
Furthermore, the integration of ammonium salt formation within this same pot adds another layer of process intensification. By adding tetrabutylammonium hydrogen sulfate directly to the reaction filtrate, the product precipitates or partitions efficiently, allowing for a straightforward extraction and concentration workflow. This design minimizes the number of unit operations, reducing the total processing time and the physical footprint required for manufacturing. From an impurity control perspective, the use of specific coupling reagents like NDSC (N,N'-disuccinimidyl carbonate) in the preceding amide bond formation step ensures high regioselectivity, preventing the formation of N-acyl urea byproducts that are common with carbodiimide reagents. The cumulative effect of these mechanistic optimizations is a process that consistently delivers Relebactam intermediates with purity exceeding 99.5%, meeting the stringent specifications required for final API synthesis without the need for resource-intensive recrystallization cycles.
How to Synthesize Relebactam Efficiently
The execution of this advanced synthetic route requires precise control over reaction parameters, particularly temperature and stoichiometry, to maximize the benefits of the one-pot design. The process initiates with the deprotection of the oxalate salt using a weak inorganic base like sodium carbonate, followed by cyclization with triphosgene to establish the diazabicyclooctanone core. Subsequent hydrolysis with lithium hydroxide yields the free acid, which is then coupled with N-Boc-4-aminopiperidine. The critical operational phase involves dissolving the coupled intermediate in a solvent such as acetonitrile or acetone, adding the palladium catalyst and silane donor, and maintaining the temperature between 10-20°C. Upon completion of the reduction, the sulfonation reagents are added sequentially without isolation. Detailed standardized operating procedures, including specific solvent volumes, stirring rates, and quenching protocols, are essential for replicating the high yields reported in the patent examples.
- Deprotect the ethyl ester oxalate starting material using a weak base, followed by acylation with triphosgene to form the bicyclic intermediate.
- Hydrolyze the ester intermediate using lithium hydroxide to generate the free carboxylic acid species required for coupling.
- Couple the acid with N-Boc-4-aminopiperidine using NDSC to form the amide bond, establishing the piperidine side chain.
- Execute a critical one-pot reaction involving catalytic debenzylation using a silane hydrogen donor, followed immediately by sulfonation and ammonium salt formation.
- Remove the Boc protecting group using trifluoroacetic acid to yield the final high-purity Relebactam product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. The elimination of high-pressure hydrogenation equipment represents a substantial capital expenditure saving, as facilities no longer require specialized autoclaves or extensive safety infrastructure dedicated to handling compressed hydrogen gas. This reduction in equipment complexity directly correlates to lower maintenance costs and reduced downtime, enhancing the overall reliability of the manufacturing asset. Moreover, the use of commercially available and relatively inexpensive starting materials, such as the ethyl ester oxalate derivative, mitigates the supply risk associated with sourcing proprietary or scarce intermediates. By simplifying the post-treatment workflow—replacing multiple isolation and purification steps with a streamlined one-pot operation—the process significantly reduces solvent consumption and waste disposal costs. This aligns perfectly with modern green chemistry initiatives, potentially lowering the environmental compliance burden and facilitating smoother regulatory approvals in jurisdictions with strict waste management laws.
- Cost Reduction in Manufacturing: The transition from high-pressure catalytic hydrogenation to atmospheric transfer hydrogenation eliminates the need for expensive pressure-rated reactors and the associated safety monitoring systems. This structural change in the process design leads to significant operational cost savings by reducing energy consumption and equipment depreciation. Additionally, the high selectivity of the one-pot sulfonation step minimizes the loss of valuable material to side reactions, thereby improving the overall mass balance and reducing the cost of goods sold (COGS) per kilogram of finished intermediate. The avoidance of specialized reagents like thioglycolic acid, which is noted in other literature for its poor economy and odor issues, further contributes to a more cost-efficient production model.
- Enhanced Supply Chain Reliability: Dependence on a single, complex starting material often creates a bottleneck in the supply chain; however, this new route utilizes precursors that are more readily accessible and easier to synthesize in bulk. The robustness of the one-pot procedure reduces the likelihood of batch failures due to intermediate instability, ensuring a consistent and predictable output of high-purity product. This reliability is critical for maintaining continuous supply to downstream API manufacturers, reducing the risk of stockouts that can disrupt the production of life-saving antibiotic combinations. The simplified logistics of handling non-hazardous silane reagents instead of high-pressure hydrogen cylinders also streamline the inbound logistics and storage requirements at the manufacturing site.
- Scalability and Environmental Compliance: The process is inherently designed for industrial mass production, with reaction conditions that are easily scalable from pilot plant to multi-ton commercial batches without significant re-engineering. The reduction in the number of isolation steps means less solvent is used and less waste is generated, directly addressing the 'three wastes' (waste water, waste gas, waste residue) challenge. This environmentally friendly profile not only lowers disposal costs but also positions the manufacturer favorably against increasingly stringent global environmental regulations. The ability to produce high-purity material with minimal purification steps ensures that the process remains economically viable even at large scales, supporting the growing global demand for effective beta-lactamase inhibitors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in CN111943950A, providing clarity on how this technology resolves historical pain points in Relebactam production. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer or licensing agreements.
Q: How does the new synthesis method improve safety compared to traditional routes?
A: The novel process described in CN111943950A eliminates the need for high-pressure catalytic hydrogenation (H2/Pd-C), which is a standard but hazardous step in conventional Relebactam synthesis. By utilizing transfer hydrogenation with silane reagents at atmospheric pressure, the method drastically reduces explosion risks and simplifies equipment requirements.
Q: What is the expected purity of the Relebactam produced via this route?
A: The patent data indicates that the final refined product achieves a purity of greater than 99.5%, with specific examples demonstrating purity levels up to 99.72%. This high purity is attributed to the simplified post-treatment processes and the reduced formation of oxidation byproducts during the one-pot sulfonation step.
Q: Why is the one-pot debenzylation and sulfonation step considered advantageous?
A: Combining debenzylation, sulfonation, and ammonium salt formation into a single pot minimizes the isolation of unstable N-hydroxy intermediates. This reduces the probability of oxidative degradation and impurity enrichment, leading to higher overall yields and a more robust manufacturing process suitable for industrial mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Relebactam Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antibiotics like Relebactam depends on a partnership grounded in technical excellence and supply chain resilience. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are equipped with state-of-the-art facilities capable of handling the specific requirements of this novel synthesis, including rigorous QC labs that enforce stringent purity specifications to guarantee product consistency. Our commitment to quality assurance means that every batch of Relebactam intermediate we produce adheres to the highest international standards, providing our partners with the confidence needed to navigate the complex regulatory landscape of pharmaceutical manufacturing.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the availability of this critical medicine, ensuring that cost reduction in API manufacturing does not come at the expense of quality or safety, but rather enhances the global capacity to fight resistant bacterial infections.
