Advanced Enzymatic Production of High-Purity Pyroglutamic Acid Esters for Global Personal Care Markets
The landscape of fine chemical manufacturing for personal care actives is undergoing a significant paradigm shift, driven by the urgent need for greener, more efficient synthetic routes that align with stringent global safety regulations. Patent CN1754963A introduces a groundbreaking enzymatic synthesis method for preparing pyroglutamic acid esters, a class of compounds renowned for their exceptional moisturizing properties and ability to enhance skin barrier function. Unlike traditional chemical esterification which relies on harsh mineral acids and toxic solvents, this innovation leverages the specificity of hydrolases, particularly lipases, to catalyze the formation of ester bonds under mild, solvent-free conditions. For R&D directors and procurement strategists in the cosmetics industry, this technology represents a pivotal opportunity to secure a reliable functional active ingredients supplier capable of delivering high-purity products with a drastically reduced environmental footprint. The patent details a robust process where pyroglutamic acid or its short-chain esters react with long-chain alcohols in the presence of immobilized enzymes, achieving near-quantitative conversion without the generation of hazardous waste streams.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of pyroglutamic acid esters has been plagued by significant technical and economic inefficiencies inherent to classical Fischer esterification protocols. Traditional methods typically necessitate the use of aggressive catalysts such as sulfuric acid, thionyl chloride, or hydrogen chloride, which require extremely fierce reaction conditions and the presence of large volumes of organic solvents to manage heat and viscosity. These harsh environments not only pose severe safety risks regarding corrosion and toxicity but also lead to the formation of undesirable byproducts and colored impurities that are difficult to remove. Furthermore, the downstream processing involves complex neutralization steps, extensive washing, and energy-intensive distillation to recover solvents, all of which inflate the operational expenditure and extend the production lead time. The reliance on stoichiometric amounts of corrosive reagents also generates substantial amounts of inorganic salt waste, creating a heavy burden on wastewater treatment facilities and contradicting modern sustainability goals.
The Novel Approach
In stark contrast, the novel enzymatic approach described in the patent circumvents these drawbacks by utilizing biocatalysts that operate with high selectivity under mild thermal conditions, typically between 35°C and 70°C. This method eliminates the need for any organic solvent, conducting the reaction in a neat mixture of substrates which simplifies the reactor setup and removes the risk of solvent residues in the final cosmetic formulation. The use of immobilized lipase B from Candida antarctica allows for the catalyst to be easily separated via filtration and potentially reused, significantly lowering the catalyst cost per kilogram of product. By applying reduced pressure during the reaction, the process continuously removes the low-boiling byproducts (such as ethanol or water), driving the equilibrium towards completion without the need for a large excess of alcohol. This streamlined workflow not only enhances the overall yield but also ensures that the final product meets the rigorous purity specifications required for direct application in sensitive skin care products.
Mechanistic Insights into Lipase-Catalyzed Transesterification
The core of this technological advancement lies in the precise mechanistic action of the hydrolase enzyme, which facilitates the nucleophilic attack of the alcohol hydroxyl group on the carbonyl carbon of the pyroglutamic acid derivative. In the absence of water, the lipase functions primarily as a transesterification catalyst, exchanging the short-chain alkoxy group of the starting ester (e.g., ethyl or methyl) with the long-chain alcohol substrate. The enzyme's active site stabilizes the tetrahedral intermediate, lowering the activation energy required for the bond rearrangement while maintaining stereochemical integrity, which is crucial for the biological activity of the resulting moisturizer. The reaction kinetics are further optimized by the physical state of the enzyme; immobilization on a solid support prevents enzyme aggregation and denaturation at elevated temperatures, ensuring consistent catalytic activity over extended reaction periods of up to 24 hours.
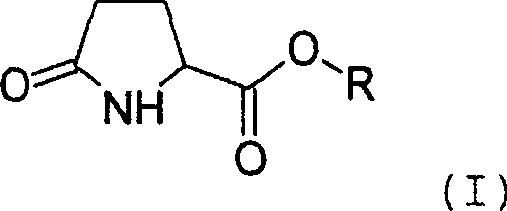
Impurity control in this biocatalytic system is inherently superior to chemical catalysis due to the enzyme's substrate specificity, which minimizes side reactions such as polymerization or degradation of the pyrrolidone ring. The patent highlights that even when using technical grade mixtures of pyroglutamic acid and its esters, the enzymatic process selectively converts the available carboxylic acid functionalities without affecting other sensitive functional groups. The removal of byproducts via vacuum distillation serves a dual purpose: it shifts the thermodynamic equilibrium according to Le Chatelier's principle to favor product formation, and it simultaneously purifies the reaction mixture in situ. This results in a crude product that is often pure enough for direct use after simple filtration, eliminating the need for column chromatography or recrystallization steps that typically result in material loss. Consequently, the impurity profile is dominated only by unreacted starting materials, which can be easily managed or are benign in the final application.
How to Synthesize Pyroglutamic Acid Esters Efficiently
To implement this synthesis effectively, manufacturers must carefully control the molar ratios of the reactants and the physical parameters of the reactor to maximize the turnover number of the expensive biocatalyst. The patent specifies that a molar ratio of alcohol to pyroglutamic acid derivative of less than 2:1, and preferably close to 1:1, is sufficient to drive the reaction to completion, which is a significant improvement over conventional methods requiring large excesses. The detailed standardized synthesis steps involve charging the reactor with the substrate mixture, adding the immobilized enzyme, and applying heat and vacuum in a coordinated manner to distill off the leaving group. For a comprehensive understanding of the specific operational parameters, temperature gradients, and work-up procedures, please refer to the technical guide below.
- Charge the reactor with a mixture of pyroglutamic acid (or its short-chain ester) and a long-chain alcohol (C4-C22) in a molar ratio preferably less than 1.5: 1.
- Add immobilized lipase B from Candida antarctica (e.g., Novozym 435) at a loading of less than 5 weight percent relative to the total reactants.
- Heat the mixture to 35-70°C under reduced pressure (e.g., 20 mbar) to continuously distill off byproducts (water or short-chain alcohol) until conversion exceeds 98%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic advantages that extend beyond mere technical feasibility. The elimination of organic solvents removes a major variable cost associated with solvent purchase, storage, and recovery, while also mitigating the regulatory risks linked to volatile organic compound (VOC) emissions. The ability to use technical grade starting materials rather than highly purified reagents allows for significant raw material cost optimization, as the biocatalyst tolerates impurities that would poison traditional chemical catalysts. Furthermore, the mild reaction conditions reduce the energy consumption required for heating and cooling, contributing to a lower carbon footprint and aligning with corporate sustainability mandates. The simplified downstream processing, which often requires only filtration to remove the solid enzyme, drastically reduces the batch cycle time and increases the throughput capacity of existing manufacturing assets.
- Cost Reduction in Manufacturing: The transition to a solvent-free enzymatic process fundamentally alters the cost structure of pyroglutamic acid ester production by removing the need for expensive solvent recovery infrastructure and hazardous waste disposal. Since the enzyme loading is kept low, typically below 5 weight percent, and the catalyst can be immobilized for potential reuse, the operational expenditure per unit of product is significantly decreased. Additionally, the high conversion efficiency means that raw material utilization is maximized, reducing the cost of goods sold (COGS) associated with unreacted feedstocks. The avoidance of corrosive acids also extends the lifespan of reactor vessels and piping, leading to lower capital maintenance costs over the long term.
- Enhanced Supply Chain Reliability: By simplifying the synthesis route and reducing the number of unit operations, the supply chain becomes more resilient to disruptions and easier to scale. The use of commercially available, standard commodity alcohols (such as oleyl alcohol or octanol) ensures a stable supply of raw materials without dependence on specialized, hard-to-source reagents. The robustness of the enzymatic process against variations in feedstock quality means that production schedules are less likely to be delayed by quality control issues with incoming materials. This reliability is crucial for meeting the just-in-time delivery requirements of major personal care brands and maintaining continuous inventory levels.
- Scalability and Environmental Compliance: The solvent-free nature of this technology makes it inherently safer and easier to scale from pilot plant to commercial production, as there are no flammable solvent vapors to manage in large reactors. This facilitates the commercial scale-up of complex moisturizing agents without the need for explosion-proof facilities or extensive environmental permitting for VOC emissions. The process generates minimal waste, primarily consisting of the solid enzyme support and small amounts of distilled byproducts, which simplifies compliance with increasingly strict environmental regulations. This green chemistry profile enhances the marketability of the final ingredient to eco-conscious consumers and brands seeking clean-label certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis route, derived directly from the patent specifications and experimental data. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing manufacturing portfolio. The answers provided reflect the specific capabilities of the biocatalytic system in handling diverse substrates and operating conditions.
Q: Does this enzymatic process require organic solvents?
A: No, the patented method specifically operates under solvent-free conditions. This eliminates the need for costly solvent recovery systems and ensures the final product is free from residual organic solvents, which is critical for cosmetic safety standards.
Q: What is the typical conversion efficiency of this biocatalytic route?
A: The process achieves quantitative yields with transformation efficiencies exceeding 98% within 24 hours. By applying vacuum to remove reaction byproducts, the equilibrium is shifted effectively towards the desired ester product.
Q: Can technical grade pyroglutamic acid mixtures be used as starting materials?
A: Yes, the method is robust enough to utilize industrial grade mixtures of pyroglutamic acid and its short-chain esters (methyl or ethyl). This flexibility allows manufacturers to use cheaper, readily available feedstocks without compromising final product purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyroglutamic Acid Ester Supplier
As the demand for high-performance, naturally derived moisturizers continues to surge, partnering with a technically proficient manufacturer is critical for securing a competitive edge in the personal care market. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced biocatalysis reactors capable of executing the solvent-free enzymatic processes described in patent CN1754963A with stringent purity specifications. We understand the critical importance of batch-to-batch consistency for cosmetic actives and employ comprehensive analytical methods to guarantee that every shipment meets the highest international standards.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced enzymatic capabilities can support your next-generation product development initiatives.
