Advanced Manufacturing of Atorvastatin Calcium Intermediates via Non-Toxic Acylation Routes
Introduction to Next-Generation Statin Intermediate Manufacturing
The global demand for lipid-lowering agents continues to drive innovation in the synthesis of key pharmaceutical building blocks, specifically within the statin class. A recent technological breakthrough detailed in patent CN113788766A introduces a robust and environmentally benign preparation method for Atorvastatin calcium intermediates, addressing critical pain points in traditional manufacturing. This novel approach fundamentally restructures the synthetic pathway by utilizing aniline and diketene as primary feedstocks, effectively bypassing the hazardous reagents that have long plagued this sector. For R&D directors and procurement strategists, this shift represents a significant opportunity to enhance supply chain resilience while adhering to increasingly strict environmental, health, and safety (EHS) regulations. By focusing on high-yield acylation and safe deprotection strategies, this technology offers a scalable solution for producing high-purity pharmaceutical intermediates without compromising on operational safety or economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-phenylisobutyrylacetamide and subsequent Atorvastatin intermediates has relied on chemically aggressive and operationally dangerous reagents. Prior art, such as the processes disclosed in U.S. Pat. No. 3,2004072893A and Chinese patent CN101337906B, frequently employs sodium cyanide (NaCN) for carbon-carbon bond formation or utilizes petroleum ether for purification. The use of NaCN introduces severe safety risks due to its acute toxicity, necessitating expensive containment systems and complex wastewater treatment protocols to neutralize cyanide residues before discharge. Furthermore, reliance on petroleum ether poses significant fire and explosion hazards due to its low flash point and high volatility, complicating storage and handling in large-scale commercial facilities. These legacy methods often suffer from moderate yields, typically hovering around 76% to 98%, but the hidden costs associated with hazard mitigation, waste disposal, and regulatory compliance often erode the apparent economic benefits of high conversion rates.
The Novel Approach
In stark contrast, the methodology outlined in CN113788766A pioneers a cleaner, more efficient route that prioritizes operator safety and environmental sustainability. The process initiates with the acylation of aniline using diketene in a benign organic solvent like ethanol, proceeding under mild thermal conditions to generate N-acetoacetanilide with exceptional efficiency. This intermediate is then subjected to a controlled reaction with isobutyryl chloride, followed by a crucial deacetylation step using an aqueous ammonium chloride solution, completely eliminating the need for toxic cyanide sources. The final coupling reaction to form the target intermediate M4 utilizes 2-halo-1-(4-fluorophenyl)-2-acetophenone under basic conditions, yielding a product with purity levels reaching 99.6%. This streamlined workflow not only simplifies the post-treatment procedure to basic liquid separation and recrystallization but also drastically reduces the generation of hazardous three-waste streams, making it an ideal candidate for modern green chemistry manufacturing standards.
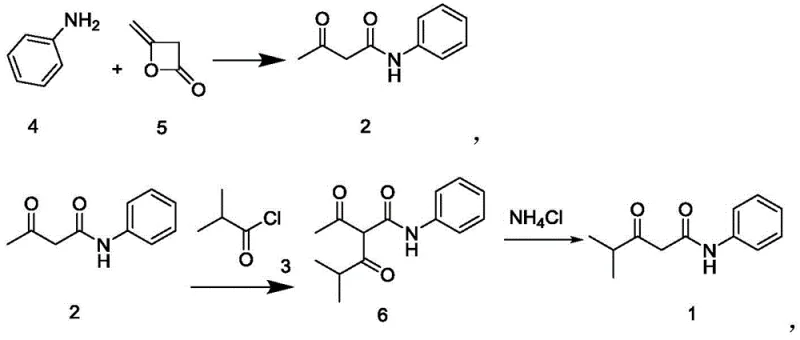
Mechanistic Insights into Diketene-Mediated Acylation and Coupling
The core of this technological advancement lies in the strategic utilization of diketene as an acylating agent, which offers superior atom economy compared to traditional ester or acid chloride approaches. In the initial step, the nucleophilic attack of the aniline nitrogen on the strained four-membered ring of diketene proceeds rapidly at temperatures between 10°C and 40°C, opening the ring to form the beta-keto amide structure of Compound 2. This reaction is highly exothermic but easily controllable, ensuring minimal formation of side products such as di-acylated species. Subsequent functionalization involves the introduction of the isobutyryl group, where the presence of a catalytic amount of 4-dimethylaminopyridine (DMAP) facilitates the acylation at the active methylene position. The use of calcium hydroxide and calcium oxide as bases in this stage provides a heterogeneous environment that aids in driving the equilibrium forward while minimizing hydrolysis of the sensitive amide bonds.
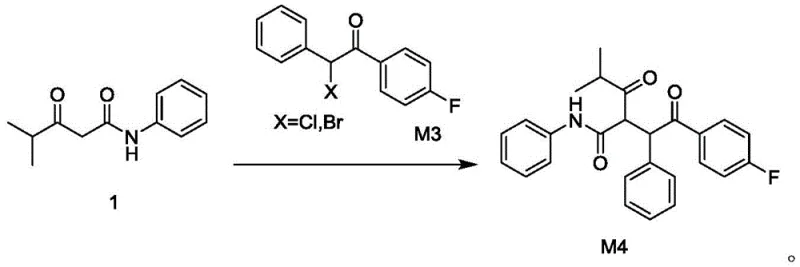
Impurity control is meticulously managed through the selection of specific crystallization solvents and the avoidance of harsh acidic or basic workups that could degrade the product. The final coupling step to generate Compound M4 involves the alkylation of the enolate derived from Compound 1 with the alpha-halo ketone M3. By optimizing the molar ratio of reactants and maintaining a reaction temperature of 45°C in isopropanol, the process ensures complete consumption of the starting materials while suppressing potential elimination side reactions. The resulting crude product is purified via a methanol-water recrystallization system, which effectively removes unreacted starting materials and inorganic salts, delivering a final product that meets the rigorous purity specifications required for downstream API synthesis. This mechanistic precision ensures that the impurity profile remains consistent and manageable, a critical factor for regulatory approval.
How to Synthesize Atorvastatin Calcium Intermediate Efficiently
The synthesis of these critical statin precursors requires precise control over reaction parameters to maximize yield and minimize impurity formation. The patented process delineates a clear, three-stage protocol that begins with the formation of the acetoacetanilide backbone, followed by side-chain modification, and concludes with the assembly of the final pharmacophore. Operators must pay close attention to the dropwise addition rates of reactive chlorides and the maintenance of specific temperature windows during the exothermic phases to prevent thermal runaway. The following guide summarizes the critical operational parameters derived from the experimental examples, providing a roadmap for implementing this technology in a pilot or commercial setting. For a comprehensive breakdown of the standardized operating procedures, please refer to the detailed synthesis instructions below.
- Perform acylation of aniline with diketene in ethanol at 30°C to obtain N-acetoacetanilide (Compound 2).
- React Compound 2 with isobutyryl chloride using DMAP catalyst, followed by ammonium chloride treatment to synthesize N-phenylisobutyrylacetamide (Compound 1).
- Couple Compound 1 with 2-halo-1-(4-fluorophenyl)-2-acetophenone (M3) in isopropanol with potassium carbonate to yield the final intermediate M4.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthetic route offers substantial strategic advantages beyond mere technical feasibility. By transitioning away from highly regulated and dangerous reagents like sodium cyanide, manufacturers can significantly reduce the overhead costs associated with specialized hazardous waste disposal and safety monitoring infrastructure. The elimination of petroleum ether further mitigates the risks of fire and explosion, potentially lowering insurance premiums and allowing for storage in less restrictive facility zones. Moreover, the use of commodity chemicals such as aniline, diketene, and isobutyryl chloride ensures a stable and diversified supply base, reducing vulnerability to raw material shortages that often plague niche reagent markets. This stability translates directly into more predictable production schedules and enhanced reliability for downstream API customers.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates several costly unit operations found in legacy methods, particularly those related to the neutralization and detoxification of cyanide-containing waste streams. By utilizing simple liquid-liquid separations and standard recrystallization techniques, the requirement for expensive chromatography purification or complex distillation setups is removed, leading to a drastic simplification of the production workflow. Additionally, the high atom economy of the diketene acylation step ensures that raw material utilization is maximized, reducing the overall cost of goods sold (COGS) per kilogram of finished intermediate. These efficiencies collectively contribute to a more competitive pricing structure without sacrificing product quality.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is inherently more secure due to the widespread availability of aniline and diketene in the global chemical market. Unlike specialized reagents that may be subject to geopolitical trade restrictions or limited supplier pools, these foundational chemicals are produced at massive scales for various industries, ensuring consistent availability and price stability. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without significant yield loss, further enhances manufacturing reliability. This resilience allows suppliers to maintain continuous production runs even during minor utility fluctuations, ensuring on-time delivery for critical pharmaceutical projects.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like ethanol and toluene that are easily recovered and recycled in standard industrial distillation columns. The absence of heavy metals or persistent organic pollutants in the waste stream simplifies compliance with increasingly stringent environmental regulations, such as REACH in Europe or TSCA in the United States. This environmental compatibility facilitates faster regulatory approvals for new manufacturing sites and reduces the long-term liability associated with site remediation. Consequently, this technology supports the commercial scale-up of complex pharmaceutical intermediates with a significantly reduced environmental footprint.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this manufacturing route, we have compiled answers to common inquiries regarding the process specifics and quality outcomes. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these nuances is essential for integrating this technology into existing production lines or designing new facilities dedicated to statin intermediate synthesis.
Q: How does this new method improve safety compared to traditional Atorvastatin intermediate synthesis?
A: The patented method eliminates the use of highly toxic sodium cyanide (NaCN) and flammable petroleum ether, replacing them with safer reagents like ammonium chloride and standard organic solvents such as ethanol and toluene, significantly reducing operational hazards and wastewater toxicity.
Q: What represents the key cost-saving advantage of using diketene in this pathway?
A: Using diketene allows for a direct and high-yielding acylation of aniline under mild conditions (30°C), avoiding complex multi-step protections or expensive catalysts required in older methods, thereby streamlining the production process and lowering raw material costs.
Q: What purity levels can be achieved with this specific crystallization protocol?
A: Through optimized recrystallization steps using methanol and water mixtures, the process consistently achieves target product purities exceeding 99.6%, meeting the stringent quality specifications required for downstream API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atorvastatin Calcium Intermediate Supplier
As the pharmaceutical industry evolves towards greener and more efficient manufacturing paradigms, partnering with an experienced CDMO becomes crucial for navigating these technological transitions. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Atorvastatin intermediate meets the highest international standards. We understand the critical nature of supply continuity in the pharma sector and are committed to delivering consistent quality and volume to support your global drug development pipelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your operational context. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to optimize your supply chain and bring life-saving medications to market faster and more efficiently.
