Advanced Reductive Amination Strategy for High-Purity Chroman Derivatives and Commercial Scale-Up
Advanced Reductive Amination Strategy for High-Purity Chroman Derivatives and Commercial Scale-Up
The pharmaceutical industry constantly seeks robust synthetic pathways that balance safety, efficiency, and scalability, particularly for complex central nervous system (CNS) active agents. Patent CN1622944A introduces a transformative methodology for the production of 2-[5-(4-fluorophenyl)-3-pyridylmethylaminomethyl]chroman, a critical intermediate with potent dopamine D2 receptor antagonistic and 5-HT1A receptor agonistic properties. This innovation marks a significant departure from prior art, specifically addressing the limitations associated with hazardous alkylating agents. By shifting the synthetic paradigm to a direct reductive amination protocol, the patent outlines a process that not only enhances operator safety but also streamlines the purification landscape. For R&D directors and process chemists, this represents a viable route to access high-purity intermediates essential for treating conditions ranging from stroke and migraine to schizophrenia and Alzheimer's disease. The technical depth of this disclosure provides a solid foundation for scaling these complex molecules from laboratory benchtop to multi-ton commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific chroman derivative relied heavily on the nucleophilic substitution reactions described in earlier patents such as US 5,767,132. In these conventional pathways, 2-aminomethylchroman is reacted with 3-(chloromethyl)-5-(4-fluorophenyl)pyridine. While chemically feasible, this approach presents severe drawbacks for modern manufacturing environments. The chloromethyl pyridine reagent is classified as a potent skin irritant and a potential allergen, posing significant occupational health risks that require expensive containment engineering controls. Furthermore, molecules containing both alkylating and alkylatable functional groups within the same structure, often referred to as haloamino compounds, exhibit a dangerous tendency toward exothermic decomposition. This thermal instability complicates reactor management and increases the risk of runaway reactions during scale-up. Additionally, the nucleophilic nature of the amine reactant often leads to uncontrolled double alkylation, generating difficult-to-remove bis-arylpyridine by-products that compromise the purity profile of the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast to the hazardous alkylation routes, the methodology disclosed in CN1622944A utilizes a direct reductive amination strategy that fundamentally alters the reaction landscape. This novel approach involves the condensation of 5-(4-fluorophenyl)pyridine-3-carbaldehyde with 2-aminomethylchroman or its stable salt forms under reducing conditions. By replacing the unstable chloromethyl species with a robust aldehyde precursor, the process eliminates the risk of exothermic decomposition associated with haloamino compounds. The reaction proceeds smoothly in the presence of mild reducing agents such as sodium borohydride or via catalytic hydrogenation, offering superior control over the reaction kinetics. Crucially, this pathway inherently suppresses the formation of double alkylation impurities, resulting in a cleaner crude reaction mixture. This improvement in selectivity drastically simplifies downstream purification, allowing manufacturers to achieve stringent quality specifications with fewer processing steps and reduced solvent usage.
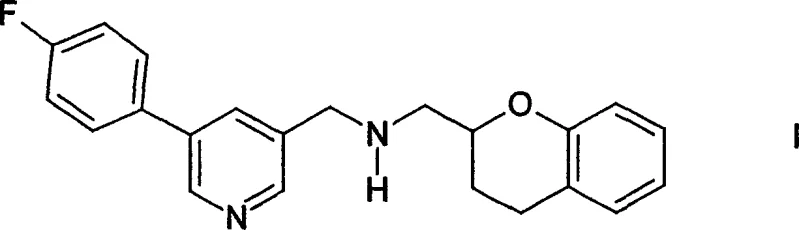
Mechanistic Insights into Reductive Amination and Chiral Resolution
The core of this technological advancement lies in the mechanistic elegance of the reductive amination cycle. Unlike standard literature methods that often require the isolation of an aldimine intermediate through water elimination, this process allows for the direct combination of the aldehyde and amine components. The reaction initiates with the formation of an iminium ion intermediate in situ, which is immediately reduced by the hydride source to form the secondary amine bond. This one-pot transformation avoids the thermal stress typically required to drive water removal, thereby preventing typical aldehyde side reactions such as disproportionation into alcohols and acids or oligomerization into trioxanes. The ability to utilize the amine directly from its storage-stable salt form, such as the hydrochloride salt, further enhances the mechanism's efficiency. The base required to liberate the free amine is generated in situ or added concurrently, removing the necessity for a separate liquid-liquid extraction step to isolate the free base prior to reaction. This telescoping of steps reduces the overall process mass intensity and minimizes waste generation.
A critical aspect of producing biologically active chroman derivatives is the control of stereochemistry, as the molecule contains a chiral center at the 2-position of the chroman ring. The patent details a sophisticated resolution strategy to access enantiomerically pure forms, which are often required for optimal pharmacological activity. This is achieved by reacting racemic 2-aminomethylchroman with a chiral resolving agent, specifically (S)-N-tosylproline. The interaction between the racemic amine and the chiral acid generates a pair of diastereomeric salts with distinct physical properties, particularly solubility. Through careful fractional crystallization, one diastereomer can be selectively precipitated while the other remains in solution. The X-ray structural analysis confirms the absolute configuration of these salts, ensuring that the subsequent liberation of the free amine yields the desired (R) or (S) enantiomer with high optical purity. This resolution technique is robust and scalable, providing a reliable supply of single-enantiomer intermediates essential for modern drug development.
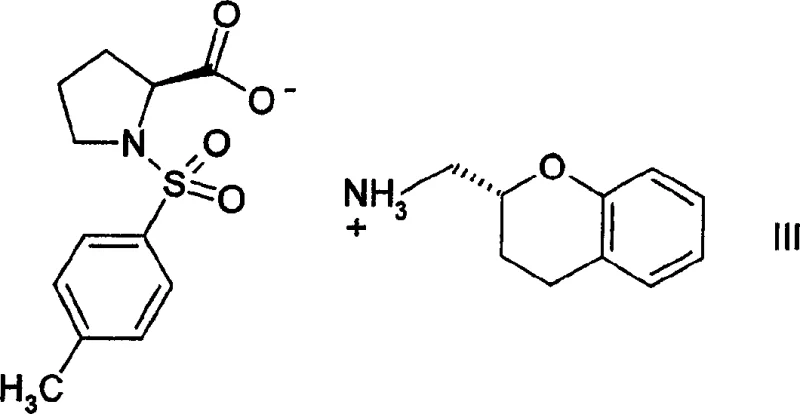
How to Synthesize 2-[5-(4-fluorophenyl)-3-pyridylmethylaminomethyl]chroman Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The process begins with the preparation of the key aldehyde building block, 5-(4-fluorophenyl)pyridine-3-carbaldehyde, which can be derived from commercially available nicotinic acid precursors through reduction and subsequent oxidation. Once the aldehyde is secured, it is combined with the aminomethylchroman hydrochloride salt in a protic solvent such as ethanol or methanol. The addition of a reducing agent like sodium borohydride facilitates the coupling, typically proceeding at mild temperatures between 20°C and 35°C to maintain selectivity. Following the reaction, the product can be isolated directly as a salt, such as the dihydrochloride hemihydrate, by adjusting the pH with hydrochloric acid. For applications requiring enantiopure material, the resolution step using N-tosylproline is integrated prior to the final coupling or applied to the amine precursor. The detailed standardized synthesis steps see the guide below.
- Preparation of 5-(4-fluorophenyl)pyridine-3-carbaldehyde from commercially available nicotinic acid derivatives via reduction and oxidation.
- Direct reductive amination of the aldehyde with 2-aminomethylchroman hydrochloride using sodium borohydride or catalytic hydrogenation.
- Optional chiral resolution of the amine precursor using N-tosylproline salts to obtain enantiomerically pure products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the transition to this novel synthetic route offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for the hazardous 3-(chloromethyl)-5-(4-fluorophenyl)pyridine, manufacturers remove a significant bottleneck associated with sourcing specialized, high-risk alkylating agents. This shift not only mitigates regulatory compliance burdens related to hazardous substance handling but also ensures greater continuity of supply, as the aldehyde precursors are more stable and widely available. Furthermore, the improved impurity profile resulting from the suppression of double alkylation means that less material is lost during purification stages. This increase in effective yield translates directly into better cost efficiency, allowing for more competitive pricing structures without compromising on quality standards. The ability to use amine salts directly also reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing process.
- Cost Reduction in Manufacturing: The elimination of hazardous chloromethyl reagents removes the need for expensive containment infrastructure and specialized waste treatment protocols, leading to significant operational cost savings. Additionally, the suppression of double alkylation by-products reduces the burden on purification units, minimizing the loss of valuable intermediates during chromatography or recrystallization. The telescoping of the free base generation and coupling steps further reduces solvent usage and energy consumption, driving down the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Sourcing stable aldehyde precursors is inherently less risky than managing thermally unstable haloamino compounds, ensuring a more consistent flow of raw materials. The robustness of the reductive amination process allows for flexible manufacturing schedules, as the reaction is less sensitive to minor fluctuations in temperature or mixing compared to exothermic alkylation reactions. This reliability is crucial for maintaining long-term supply contracts with pharmaceutical partners who demand uninterrupted delivery of critical intermediates.
- Scalability and Environmental Compliance: The process operates under mild conditions with common reducing agents, making it highly amenable to scale-up in standard stainless steel reactors without requiring exotic metallurgy. The reduction in solvent volume and the avoidance of toxic alkylating agents align perfectly with green chemistry principles, facilitating easier permitting and environmental compliance. This scalability ensures that production can be ramped up from pilot plant quantities to multi-ton commercial volumes seamlessly to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on process safety, stereochemical control, and purification efficiency. Understanding these nuances is vital for technical teams evaluating the feasibility of adopting this route for their own production pipelines. The answers reflect the consensus on best practices for handling the specific reagents and conditions outlined in the intellectual property.
Q: Why is the reductive amination route preferred over the conventional alkylation method for this chroman derivative?
A: The conventional method utilizes 3-(chloromethyl)-5-(4-fluorophenyl)pyridine, which is a known skin irritant and prone to exothermic decomposition. The new reductive amination route avoids these hazardous haloamino compounds, significantly improving operational safety and reducing the formation of double-alkylation by-products.
Q: How does the patent address the challenge of obtaining enantiomerically pure intermediates?
A: The process employs a highly effective chiral resolution strategy using (S)-N-tosylproline. By forming diastereomeric salts with racemic 2-aminomethylchroman, the desired enantiomer can be isolated through fractional crystallization, ensuring high optical purity for the final active pharmaceutical ingredient.
Q: What are the purification advantages of this novel synthetic pathway?
A: By suppressing double alkylation side reactions, the new method yields a crude product with a cleaner impurity profile. Furthermore, the ability to use the amine salt directly in situ eliminates the need for a separate liquid-liquid extraction step to generate the free base, simplifying the overall workup and reducing solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-[5-(4-fluorophenyl)-3-pyridylmethylaminomethyl]chroman Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the highest quality and safety standards for pharmaceutical intermediates. Our technical team has extensively analyzed the pathways described in CN1622944A and possesses the expertise to implement this reductive amination strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise determination of enantiomeric excess for chiral intermediates. We are committed to delivering high-purity chroman derivatives that meet the exacting demands of global drug development programs.
We invite you to collaborate with us to leverage these technological advancements for your specific projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. We look forward to supporting your supply chain with reliable, high-quality intermediates produced through state-of-the-art chemical processes.
