Scalable Synthesis of High-Purity L-alpha-GPC Using Stable Chiral Intermediates for Commercial Production
Scalable Synthesis of High-Purity L-alpha-GPC Using Stable Chiral Intermediates for Commercial Production
The pharmaceutical industry continuously seeks robust synthetic routes for critical neuroprotective agents, and the methodology disclosed in patent CN103087091A represents a significant advancement in the production of L-alpha-Glycerophosphorylcholine (L-alpha-GPC). This vital compound serves as a precursor for acetylcholine and phosphatidylcholine, playing a crucial role in cognitive function and membrane integrity. The patent introduces a streamlined chemical pathway that circumvents the historical reliance on unstable epoxide intermediates or complex extraction processes from natural lecithin sources. By leveraging a direct nucleophilic substitution strategy using (R)-(-)-3-chloro-1,2-propanediol, the inventors have established a protocol that balances high stereochemical fidelity with operational simplicity. This technical breakthrough addresses long-standing challenges regarding raw material stability and purification efficiency, positioning this method as a superior candidate for industrial-scale manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-alpha-GPC has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up of complex phospholipids. Early methods relied on the extraction of the compound from biological sources such as bovine pancreas or egg yolk lecithin, processes inherently limited by low yields, complex isolation procedures, and inconsistent purity profiles that fail to meet stringent regulatory standards for active pharmaceutical ingredients. Subsequent synthetic attempts, such as those described in EP0486100, utilized D-acetone glycerol and 2-chloro-2-oxy-1,3,2-dioxaphospholane, necessitating a hazardous ring-opening reaction with trimethylamine under pressurized conditions. This approach not only involves expensive and difficult-to-prepare starting materials but also introduces severe safety risks associated with high-pressure operations and the handling of moisture-sensitive cyclic phosphates. Furthermore, alternative routes employing glycidol derivatives, as seen in WO2007145476 and CN101544667, suffer from the intrinsic instability of the epoxide starting materials, which often require freezing preservation and rigorous purification via vacuum distillation prior to use, thereby inflating production costs and complicating supply chain logistics.
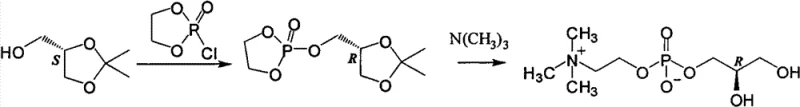
The Novel Approach
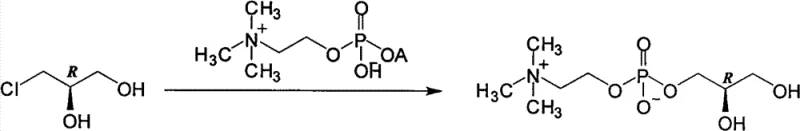
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN103087091A offers a remarkably direct and atom-economical solution by utilizing (R)-(-)-3-chloro-1,2-propanediol as the chiral building block. This specific intermediate is not only chemically stable at ambient temperatures, eliminating the need for energy-intensive cryogenic storage, but it is also a widely available commodity chemical used in the synthesis of other drugs like levodropropizine, ensuring a reliable supply chain. The core innovation lies in the one-step substitution reaction between this stable chlorohydrin and phosphorylcholine tetramethyl ammonium salt, which proceeds smoothly under standard reflux conditions in common solvents such as water, alcohols, or acetonitrile. This eliminates the requirement for specialized high-pressure reactors and avoids the generation of toxic byproducts associated with cyclic phosphate precursors. The resulting crude product is then subjected to a highly effective purification step using ion exchange resins, which efficiently removes mineral ions and residual chloride to yield the final sterling product with exceptional optical and chemical purity.
Mechanistic Insights into Nucleophilic Substitution and Ion Exchange Purification
The chemical elegance of this process is rooted in a classic bimolecular nucleophilic substitution (SN2) mechanism, where the phosphate oxygen of the phosphorylcholine tetramethyl ammonium salt acts as a potent nucleophile attacking the chiral carbon bearing the chlorine atom in the (R)-(-)-3-chloro-1,2-propanediol. Since the chlorine atom is an excellent leaving group, the reaction proceeds with inversion of configuration or retention depending on the specific coordination environment, but critically, the use of the pre-defined (R)-enantiomer ensures that the final L-alpha-GPC product maintains the requisite stereochemistry for biological activity. The reaction kinetics are favorable under reflux temperatures, allowing for complete conversion without the need for exotic catalysts or extreme pressure conditions that often degrade sensitive phospholipid structures. The choice of solvent plays a pivotal role in facilitating this interaction; polar protic solvents like water or alcohols help solvate the ionic phosphorylcholine species while maintaining the solubility of the organic chlorohydrin, creating a homogeneous reaction medium that maximizes collision frequency and reaction rates.
Following the substitution reaction, the control of impurities is achieved through a sophisticated yet operationally simple ion exchange purification strategy that is critical for meeting high-purity L-alpha-GPC specifications. The crude reaction mixture typically contains residual mineral ions, such as calcium or sodium, originating from the initial preparation of the phosphorylcholine salt, as well as chloride ions released during the substitution event. By passing the aqueous solution of the crude product through strong acid cation exchange resins like Dowex 509H, Amberlite IR-120, or Liwatit S100, these unwanted cationic species are tightly bound to the resin matrix while the zwitterionic L-alpha-GPC molecule passes through unaffected. This selective adsorption mechanism effectively scrubs the product stream of inorganic contaminants that could otherwise catalyze degradation or interfere with downstream formulation. The result is a highly purified aqueous solution that, upon concentration under reduced pressure, yields a white crystalline solid with a chemical purity of 99% and an enantiomeric excess (ee) value of 99%, demonstrating the robustness of this purification protocol in delivering pharmaceutical-grade material.
How to Synthesize L-alpha-Glycerophosphorylcholine Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing the importance of precise stoichiometry and temperature control to maximize yield. The process begins with the in-situ generation of the reactive phosphorylcholine tetramethyl ammonium species, followed by the addition of the chiral chlorohydrin and subsequent reflux. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-efficiency route.
- Preparation of phosphorylcholine tetramethyl ammonium by reacting phosphorylcholine calcium salt with oxalic acid and tetramethylammonium hydroxide.
- Execution of the substitution reaction between the prepared ammonium salt and (R)-(-)-3-chloro-1,2-propanediol in a suitable solvent under reflux conditions.
- Purification of the crude product using ion exchange resins such as Dowex 509H or Amberlite IR-120 to remove mineral ions and achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic benefits that extend far beyond simple chemical yield improvements. By shifting away from unstable epoxide intermediates like glycidol, which require specialized cold-chain logistics and have limited shelf lives, manufacturers can drastically simplify their raw material inventory management and reduce the risk of production stoppages due to reagent degradation. The use of (R)-(-)-3-chloro-1,2-propanediol, a stable and commercially abundant chemical, ensures a consistent and reliable supply of starting materials, mitigating the volatility often associated with niche chiral building blocks. Furthermore, the elimination of high-pressure reaction steps reduces the capital expenditure required for specialized reactor vessels and lowers the operational overhead related to safety compliance and maintenance, contributing to substantial cost savings in API manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the removal of expensive and complex reagents such as D-acetone glycerol and cyclic phosphates, which traditionally drive up the bill of materials. The ability to perform the reaction in common, low-cost solvents like water, ethanol, or acetone further decreases the operational expenditure associated with solvent procurement and recovery. Additionally, the simplified purification workflow using ion exchange resins avoids the need for costly chromatographic columns or extensive recrystallization cycles, streamlining the downstream processing phase and reducing labor and utility consumption per kilogram of finished product.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the key chiral intermediate allows for bulk purchasing and long-term storage without the fear of quality deterioration, providing a buffer against market fluctuations and supply disruptions. The robustness of the reaction conditions means that production can be scaled up in standard glass-lined or stainless steel reactors available in most multipurpose chemical facilities, reducing the lead time for high-purity choline derivatives by eliminating the need for custom equipment fabrication. This flexibility enables manufacturers to respond more agilely to market demand surges for neuroprotective supplements and pharmaceutical formulations.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly lower than previous methods, as it avoids the generation of hazardous waste streams associated with tosylate activation or pressurized amine handling. The aqueous workup and ion exchange purification generate minimal organic waste, aligning with modern green chemistry principles and simplifying wastewater treatment protocols. The high atom economy of the direct substitution reaction ensures that the majority of the starting mass is incorporated into the final product, minimizing waste disposal costs and enhancing the overall sustainability profile of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method, derived directly from the experimental data and comparative analysis presented in the patent documentation. These insights are intended to clarify the operational advantages and quality parameters associated with this novel production route.
Q: Why is (R)-(-)-3-chloro-1,2-propanediol preferred over glycidol for L-alpha-GPC synthesis?
A: Unlike (R)-(+)-glycidol, which is unstable at normal temperatures and requires freezing preservation and distillation, (R)-(-)-3-chloro-1,2-propanediol possesses stable chemical properties and is easily obtainable as a key intermediate for other pharmaceuticals like levodropropizine.
Q: What purification method ensures high optical purity in this process?
A: The process utilizes ion exchange resin purification (e.g., Dowex 509H, Amberlite IR-120) to effectively remove mineral ions and chloride byproducts, achieving a chemical purity of 99% and an ee value of 99% without complex chromatography.
Q: Does this synthesis route require high-pressure equipment?
A: No, unlike older methods involving trimethylamine ring-opening reactions that require pressurization, this novel approach operates under standard atmospheric reflux conditions, significantly simplifying equipment requirements and enhancing operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-alpha-GPC Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent quality and availability of essential pharmaceutical intermediates like L-alpha-GPC. Our technical team has thoroughly analyzed the pathway described in CN103087091A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to translate this laboratory success into industrial reality. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch for chemical purity and enantiomeric excess, ensuring that your downstream formulations perform reliably.
We invite you to collaborate with us to optimize your supply chain for neuroprotective agents and leverage our expertise in chiral synthesis. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
