Advanced Metal-Free Synthesis of Diaminoalkenes via NBS-Promoted Polarity Inversion
Introduction to Patent CN113045508B
The landscape of organic synthesis is constantly evolving, driven by the need for safer, more efficient, and cost-effective methodologies for constructing complex molecular architectures. A significant breakthrough in this domain is documented in Chinese Patent CN113045508B, which details a novel method for synthesizing diaminoalkenes through NBS-promoted polarity inversion of enaminones. This technology represents a paradigm shift away from traditional transition-metal catalysis and hazardous oxidative conditions, offering a robust pathway for generating highly functionalized C(sp2)–N bonds. For R&D directors and procurement specialists alike, understanding the nuances of this metal-free approach is critical, as it directly impacts the purity profiles of active pharmaceutical ingredients and the overall economic viability of large-scale manufacturing processes. The patent highlights a strategic utilization of N-bromosuccinimide (NBS) as a promoter, enabling the transformation of electron-rich enaminones into electrophilic intermediates that readily undergo substitution with various nitrogen-containing heterocycles.
This innovation addresses long-standing challenges in the functionalization of olefinic systems, particularly the difficulty of achieving chemoselective amination without relying on precious metal catalysts that require rigorous removal steps. By leveraging a polarity inversion strategy, often referred to as umpolung, the process allows for the direct coupling of enaminones with a wide array of nitrogen nucleophiles under relatively mild thermal conditions. The implications for the supply chain are profound, as the elimination of heavy metals simplifies downstream processing and waste management, aligning with increasingly stringent environmental regulations. Furthermore, the use of commercially available and inexpensive reagents positions this methodology as a highly attractive option for the industrial production of pharmaceutical intermediates, where cost control and supply continuity are paramount concerns for decision-makers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the development of this NBS-promoted technique, the synthesis of substituted alkenes via C(sp2)–N bond formation was often fraught with significant operational and safety hurdles. One prominent conventional strategy involved the use of transition-metal catalysts for coupling aryl or alkenyl halides with nitrogen nucleophiles, a process that invariably introduces the risk of metal contamination in the final product. For pharmaceutical applications, removing trace amounts of palladium or copper to meet regulatory standards adds substantial complexity and cost to the purification workflow. Additionally, alternative metal-free oxidative coupling methods frequently relied on hazardous oxidants such as tert-butyl hydroperoxide (TBHP), which is known for its explosive potential and requires careful handling at elevated temperatures around 100°C.
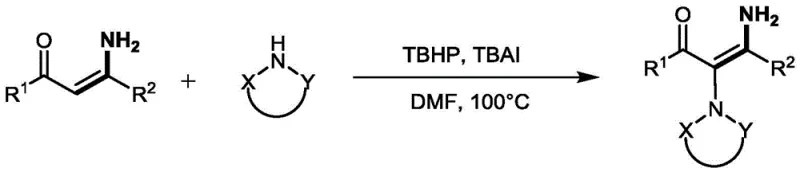
Another notable limitation in the prior art was the reliance on expensive hypervalent iodine reagents, such as iodosylbenzene (PhIO), to mediate the polarity inversion of enaminones. While effective, these reagents are costly to produce and purchase, making them economically unfeasible for large-scale commercial manufacturing. Moreover, the substrate scope for these iodine-mediated reactions was often narrow, typically restricted to specific types of alkynamines, thereby limiting their utility in the diverse landscape of drug discovery and development. The combination of high reagent costs, safety risks associated with peroxides, and the environmental burden of metal waste created a pressing need for a more sustainable and economically viable synthetic alternative.
The Novel Approach
The methodology described in Patent CN113045508B offers a compelling solution to these historical constraints by employing N-bromosuccinimide (NBS) as a safe and effective promoter for polarity inversion. This novel approach operates under metal-free conditions, completely bypassing the need for transition-metal catalysts and the associated purification burdens. The reaction proceeds efficiently at a moderate temperature of 80°C in common solvents like DMF or toluene, significantly reducing energy consumption compared to higher-temperature protocols. By generating an electrophilic intermediate in situ, the system facilitates a smooth substitution reaction with a broad spectrum of nitrogen-containing heterocycles, including benzotriazoles, tetrazoles, and imides, yielding the desired diaminoalkene compounds with high efficiency.
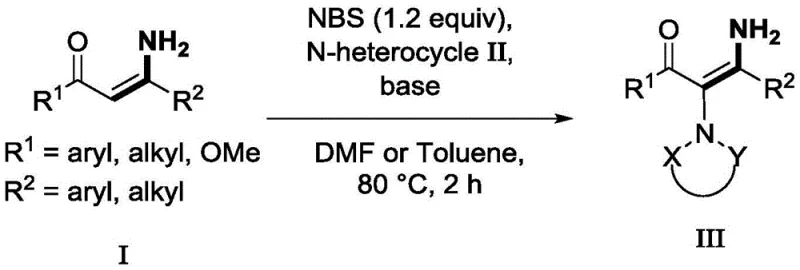
From a commercial perspective, this new route is transformative because it utilizes inexpensive and readily available starting materials, drastically lowering the barrier to entry for production. The simplicity of the operation, which involves mixing reagents at room temperature followed by heating, makes it highly amenable to scale-up in standard reactor vessels without requiring specialized equipment for handling explosives or sensitive catalysts. The broad substrate tolerance demonstrated in the patent examples, covering various aryl, alkyl, and heteroaryl substituents, ensures that this platform technology can be adapted for the synthesis of a wide range of complex intermediates. This versatility, combined with the inherent safety and cost advantages, establishes the NBS-promoted method as a superior choice for modern fine chemical manufacturing.
Mechanistic Insights into NBS-Promoted Polarity Inversion
The core chemical innovation driving this synthesis lies in the concept of polarity inversion, or umpolung, of the enaminone substrate. Typically, the beta-carbon of an enaminone is nucleophilic due to the electron-donating effect of the amino group. However, in the presence of NBS, the system undergoes a fundamental electronic reversal. The NBS acts as a source of electrophilic bromine, which initially interacts with the electron-rich double bond of the enaminone. This interaction generates a reactive brominated intermediate, effectively converting the beta-carbon from a nucleophilic center into an electrophilic one. This transient electrophile is then primed for attack by the nitrogen-containing heterocycle, which acts as the nucleophile in the subsequent substitution step.
This mechanistic pathway is crucial for controlling impurity profiles, a key concern for R&D directors. Because the reaction avoids radical chains often associated with peroxide initiators, the formation of side products is minimized, leading to cleaner reaction mixtures. The use of a base, such as sodium carbonate or DBU, plays a vital role in neutralizing the succinimide byproduct and facilitating the deprotonation of the nitrogen nucleophile, ensuring the reaction proceeds to completion. The absence of transition metals means there is no risk of metal-catalyzed decomposition pathways or metal-ligand complexation issues that can plague other coupling reactions. Consequently, the resulting diaminoalkenes exhibit high structural integrity and purity, reducing the need for extensive chromatographic purification and enhancing the overall yield of the process.
How to Synthesize Diaminoalkenes Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The general protocol involves dissolving the enaminone substrate in a polar aprotic solvent like DMF or a non-polar solvent like toluene, depending on the solubility profile of the specific reactants. NBS is added first to allow for the initial activation of the enaminone at room temperature, a step that is critical for forming the reactive intermediate before the introduction of the nucleophile. After a brief stirring period, the nitrogen heterocycle and the base are introduced, and the mixture is heated to 80°C for approximately two hours. This standardized approach ensures consistent results across different substrate combinations.
- Mix NBS (1.2 equiv) and enaminone (1.0 equiv) in DMF or toluene solvent at room temperature and stir for 10-20 minutes.
- Add the nitrogen-containing heterocyclic compound (1.2 equiv) and a base such as Na2CO3 (1.2 equiv) to the reaction mixture.
- Stir the mixture at 80°C for 2 hours under air atmosphere, then perform aqueous workup and silica gel column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NBS-promoted synthesis offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By replacing expensive and potentially supply-constrained hypervalent iodine reagents or precious metal catalysts with commodity chemicals like NBS and sodium carbonate, manufacturers can secure a more stable and predictable sourcing strategy. This shift reduces exposure to price volatility in the specialty chemical market and ensures that production schedules are not disrupted by the scarcity of exotic reagents. Furthermore, the metal-free nature of the process eliminates the need for expensive scavenging resins or complex extraction protocols designed to remove trace metals, streamlining the downstream processing workflow.
- Cost Reduction in Manufacturing: The economic impact of switching to this methodology is significant, primarily driven by the replacement of high-cost reagents with low-cost alternatives. Hypervalent iodine compounds are notoriously expensive, and their stoichiometric use in previous methods imposed a heavy financial burden on production budgets. In contrast, NBS is a bulk chemical with a well-established global supply chain and a fraction of the cost. Additionally, the elimination of transition metals removes the capital and operational expenditures associated with metal recovery and waste treatment facilities. The simplified workup procedure, which involves basic aqueous washes and standard crystallization or chromatography, further reduces labor and solvent costs, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available reagents that are not subject to the same geopolitical or logistical bottlenecks as specialized catalysts. NBS and common bases like sodium carbonate are produced by numerous chemical suppliers worldwide, ensuring redundancy in sourcing options. This diversity mitigates the risk of single-source dependency, a critical factor for maintaining continuous production of high-purity pharmaceutical intermediates. Moreover, the stability of the reagents allows for longer storage times and easier transportation, reducing the logistical complexity of inventory management. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing an additional layer of security against supply fluctuations.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is straightforward due to the absence of hazardous peroxides and the use of standard heating protocols. The reaction operates at a moderate 80°C, which is easily achievable in standard glass-lined or stainless steel reactors without requiring high-pressure or cryogenic equipment. From an environmental standpoint, the metal-free aspect significantly reduces the toxic load of the waste stream, simplifying compliance with environmental discharge regulations. The byproducts, primarily succinimide and inorganic salts, are relatively benign and easier to treat than heavy metal waste. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NBS-promoted synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the safety advantages of this NBS method compared to previous TBHP protocols?
A: Unlike previous methods that utilized explosive tert-butyl hydroperoxide (TBHP) at high temperatures (100°C), this NBS-promoted protocol operates at a milder 80°C without hazardous peroxides, significantly enhancing operational safety.
Q: How does this method improve cost efficiency over hypervalent iodine strategies?
A: This approach eliminates the need for expensive hypervalent iodine reagents like iodosylbenzene (PhIO), utilizing commercially available and inexpensive NBS instead, which drastically reduces raw material costs.
Q: What is the substrate scope for the nitrogen-containing heterocycles in this reaction?
A: The method demonstrates broad applicability, successfully reacting with benzotriazoles, tetrazoles, triazoles, succinimides, and phthalimides, offering significant versatility for diverse pharmaceutical intermediate synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaminoalkenes Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the NBS-promoted polarity inversion technology for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of volume. Our state-of-the-art facilities are equipped to handle metal-free synthesis routes with precision, adhering to stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest industry standards. We understand that the transition to a new synthetic route requires confidence in both the chemistry and the partner executing it.
We invite you to collaborate with us to leverage this cost-effective and scalable methodology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating exactly how this metal-free approach can optimize your budget. Please contact us to request specific COA data and route feasibility assessments, and let us help you streamline your supply chain while maintaining the highest levels of quality and safety.
