Revolutionizing N-Substituted Glycine Production: A Safe, Scalable Route for Global Supply Chains
Revolutionizing N-Substituted Glycine Production: A Safe, Scalable Route for Global Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the dual imperatives of environmental sustainability and operational efficiency. A pivotal advancement in this domain is documented in patent CN1091097C, which outlines a novel preparation method for N-substituted glycine and glycine esters. This technology represents a significant departure from legacy synthetic pathways that have long relied on hazardous reagents. By leveraging the reactivity of glyoxylate hemiacetals with amines followed by a controlled hydrogenation step, this process offers a robust alternative for producing critical intermediates used in the synthesis of agrochemicals, pharmaceuticals, and dyes. For global procurement leaders and R&D directors, understanding the nuances of this patent is essential, as it unlocks potential for safer, more cost-effective, and higher-purity supply chains. The methodology not only addresses the toxicity concerns associated with traditional chloroacetic acid routes but also streamlines the production of complex molecules like indigo derivatives, positioning it as a cornerstone technology for modern industrial chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-phenylglycine, a crucial precursor for indigo and various pharmaceutical intermediates, has been fraught with significant safety and environmental challenges. Traditional protocols, such as those detailed in Ullmann's Encyclopedia, typically involve the reaction of monochloroacetic acid with aniline or anthranilic acid. Alternatively, the hydrolysis of N-cyanomethylaniline has been employed, which necessitates the use of cyanic acid or sodium cyanide. These reagents are notoriously hazardous; cyanides pose acute toxicity risks to personnel and require stringent waste management protocols to prevent environmental contamination. Furthermore, the handling of monochloroacetic acid involves corrosive risks and often results in complex impurity profiles that demand energy-intensive purification steps. From a supply chain perspective, the reliance on these restricted or highly regulated chemicals introduces volatility, as regulatory changes can abruptly impact availability and cost. Consequently, manufacturers have long sought a greener, more sustainable alternative that maintains high yield without compromising on safety or product quality.
The Novel Approach
The innovative strategy presented in the patent data fundamentally shifts the synthetic paradigm by utilizing glyoxylate hemiacetals as the primary building block. Instead of introducing toxic cyanide groups or corrosive chloro-acetyl moieties, this method reacts a glyoxylate or glyoxylic acid hemiacetal directly with an amine. This condensation reaction forms an intermediate that is subsequently subjected to catalytic hydrogenation. The elegance of this approach lies in its atom economy and the benign nature of the byproducts. The process operates under relatively mild conditions, typically between 0°C and the reflux temperature of the solvent, and utilizes standard hydrogenation catalysts like nickel. This transition eliminates the need for handling extremely toxic cyanides entirely, thereby drastically simplifying the safety infrastructure required at the manufacturing site. Moreover, the resulting N-substituted glycine esters exhibit exceptional purity, often negating the need for complex downstream purification, which translates directly into reduced operational expenditures and a smaller environmental footprint for the facility.
Mechanistic Insights into Ni-Catalyzed Reductive Amination
The core of this technological breakthrough relies on a sophisticated reductive amination mechanism facilitated by transition metal catalysis. Initially, the amine nucleophile attacks the carbonyl carbon of the glyoxylate hemiacetal, forming an unstable imine or hemiaminal intermediate. This step is critical as it sets the stereochemical and structural foundation for the final product. The reaction is conducted in a diluent, preferably an alcohol such as methanol or ethanol, which serves both as a solvent and potentially participates in the stabilization of the hemiacetal species. Following the formation of the intermediate, the mixture undergoes hydrogenation in the presence of a catalyst, with nickel being the preferred active component due to its balance of activity and cost-effectiveness. The hydrogenation step effectively reduces the C=N double bond or cleaves the hemiacetal functionality to yield the stable N-substituted glycine ester. This catalytic cycle is highly efficient, operating effectively at hydrogen pressures ranging from 40 to 80 bar and temperatures between 20°C and 130°C. The choice of nickel catalysts, often supported on carriers, ensures high turnover frequencies while minimizing metal leaching, which is crucial for meeting the stringent heavy metal specifications required in pharmaceutical applications.
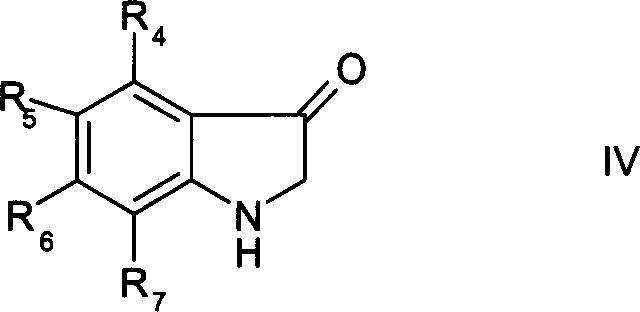
Beyond the primary reaction pathway, the control of impurities is a defining feature of this mechanism. Traditional routes often suffer from over-alkylation or the formation of polymeric byproducts due to the high reactivity of chloroacetic acid. In contrast, the glyoxylate hemiacetal route offers superior selectivity. The reaction conditions are tuned such that the formation of the desired N-substituted glycine is favored thermodynamically and kinetically. The patent data indicates that the reaction can be monitored via chromatography, ensuring that the disappearance of the starting hemiacetal correlates directly with the formation of the target intermediate. Furthermore, the subsequent cyclization of N-arylglycine esters to 3-hydroxyindole derivatives (Formula IV) demonstrates remarkable chemoselectivity. As illustrated in the structural representation below, the ester group remains intact during the initial synthesis but can be directly cyclized under molten alkali conditions without prior hydrolysis. This unique reactivity profile minimizes side reactions and ensures that the final impurity profile is clean, facilitating easier isolation and higher overall yields for downstream customers.
How to Synthesize N-Substituted Glycine Esters Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the dissolution of the amine component in a suitable alcoholic solvent, followed by the addition of the glyoxylate hemiacetal. The mixture is then heated to initiate the condensation, with progress tracked via thin-layer chromatography to ensure complete consumption of the starting materials. Once the intermediate is formed, the reaction mixture is transferred to a hydrogenation vessel where the nickel catalyst is introduced. The system is pressurized with hydrogen and heated to facilitate the reduction. This streamlined workflow eliminates the need for isolating unstable intermediates, allowing for a telescoped process that saves time and solvent. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- React glyoxylate hemiacetal with the corresponding amine in an alcoholic diluent at temperatures ranging from 0°C to reflux.
- Subject the reaction mixture to catalytic hydrogenation using a nickel-based catalyst at pressures between 40-80 bar and temperatures of 20-130°C.
- Separate the final N-substituted glycine ester or glycine by evaporating the diluent, optionally purifying via distillation or crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology offers compelling strategic advantages that extend beyond simple chemical transformation. The primary value proposition lies in the drastic simplification of the raw material portfolio. By eliminating the need for cyanides and monochloroacetic acid, companies can reduce their regulatory burden and insurance costs associated with storing and transporting hazardous materials. This shift not only enhances workplace safety but also insulates the supply chain from regulatory shocks that frequently disrupt the availability of restricted chemicals. Furthermore, the high purity of the crude product—often reaching levels sufficient for direct use in subsequent steps—means that fewer resources are allocated to purification processes like distillation or recrystallization. This efficiency gain translates into lower energy consumption and reduced waste generation, aligning perfectly with corporate sustainability goals and reducing the total cost of ownership for the intermediate.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and hazardous reagents. Traditional methods involving cyanides require specialized containment systems and extensive waste treatment facilities, which represent significant capital and operational expenditures. By switching to the glyoxylate hemiacetal route, manufacturers can bypass these costly infrastructure requirements. Additionally, the use of nickel catalysts, which are generally more affordable than precious metals like palladium or platinum, further optimizes the cost structure. The ability to telescope the reaction without isolating intermediates also reduces solvent usage and labor hours, leading to substantial overall cost savings in the manufacturing of complex amino acid derivatives.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for global enterprises, and this synthetic route enhances reliability by utilizing widely available starting materials. Glyoxylate hemiacetals and common amines are produced on a large scale by multiple suppliers, reducing the risk of single-source bottlenecks. In contrast, specialized cyanide reagents often have limited supply channels and are subject to strict transportation regulations that can cause delays. The robustness of the hydrogenation step, which tolerates a range of pressures and temperatures, ensures consistent production output even under varying operational conditions. This resilience makes the supply chain less vulnerable to external disruptions, ensuring that downstream production of APIs or agrochemicals remains uninterrupted.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden inefficiencies, but this method is inherently scalable. The reaction conditions are compatible with standard industrial hydrogenation reactors, and the exothermic nature of the reaction is manageable with conventional cooling systems. From an environmental perspective, the process generates significantly less hazardous waste. The absence of chloride ions and cyanide residues simplifies effluent treatment, making it easier to comply with increasingly stringent environmental regulations. This 'green' credential is not just a compliance checkbox but a market differentiator, allowing customers to market their final products as sustainably sourced, which is increasingly valued in the global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps R&D teams assess feasibility and allows procurement teams to negotiate better terms based on the inherent efficiencies of the process. The answers cover aspects ranging from catalyst selection to downstream processing capabilities.
Q: What are the safety advantages of this new glycine synthesis method compared to traditional routes?
A: Unlike conventional methods that rely on hazardous monochloroacetic acid or toxic cyanides, this process utilizes glyoxylate hemiacetals and amines, significantly reducing environmental risks and handling hazards.
Q: Can N-arylglycine esters be directly converted to indigo precursors without hydrolysis?
A: Yes, the patent highlights that N-arylglycine esters can undergo direct ring closure to form 3-hydroxyindole derivatives in molten alkali hydroxides, eliminating the need for a separate hydrolysis step.
Q: What level of purity can be achieved with this hydrogenation process?
A: The process yields N-substituted glycine esters with exceptionally high purity, often reaching up to 99% as confirmed by gas chromatography, minimizing the need for extensive downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Substituted Glycine Supplier
The technical potential of patent CN1091097C is immense, offering a pathway to safer and more efficient chemical manufacturing. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovation to the global market. Our facility is equipped with state-of-the-art high-pressure hydrogenation reactors and stringent purity specifications are maintained through our rigorous QC labs. We understand that transitioning to a new synthetic route requires a partner who can guarantee consistency and quality at scale. Our team of expert chemists is ready to adapt this proprietary knowledge to your specific production needs, ensuring that you receive high-purity intermediates that meet the exacting standards of the pharmaceutical and agrochemical industries.
We invite you to explore how this advanced synthesis method can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current production volumes. We are prepared to provide specific COA data and route feasibility assessments to demonstrate the tangible benefits of switching to this superior technology. Let us collaborate to build a more sustainable and resilient supply chain for your critical chemical intermediates.
