Advanced Palladium-Catalyzed Synthesis of Indolinone Thioesters for Pharmaceutical Applications
Advanced Palladium-Catalyzed Synthesis of Indolinone Thioesters for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective methodologies for constructing complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN115403505A, which discloses a novel preparation method for thioester compounds containing an indolinone structure. Indolinones are privileged structures found in numerous bioactive molecules and natural products, serving as critical intermediates in the synthesis of various therapeutic agents. The traditional reliance on transition metal-catalyzed cascade cyclization reactions has often been hampered by limitations in sulfur sources and carbonylation efficiency. This new technology addresses these challenges by introducing a palladium-catalyzed cyclization/thiocarbonylation protocol that utilizes sulfonyl chlorides as the sulfur source and molybdenum carbonyl as both the carbonyl source and reducing agent. This approach not only simplifies the operational complexity but also significantly broadens the substrate scope, offering a reliable pathway for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds via transition metal-catalyzed thiocarbonylation has heavily relied on thiols or disulfides as the sulfur source. While effective in certain contexts, these reagents present substantial drawbacks for industrial application. Thiols are notorious for their strong, unpleasant odors and their tendency to act as catalyst poisons due to their high affinity for transition metals like palladium. This catalyst deactivation often necessitates the use of excessive catalyst loading, driving up production costs and complicating downstream purification. Furthermore, the handling of gaseous carbon monoxide (CO) in traditional carbonylation reactions poses severe safety risks, requiring specialized high-pressure equipment and rigorous safety protocols. These factors collectively create bottlenecks in cost reduction in pharmaceutical intermediate manufacturing, limiting the feasibility of these routes for large-scale production.
The Novel Approach
The methodology described in patent CN115403505A represents a paradigm shift by replacing problematic thiols with sulfonyl chlorides. Sulfonyl chlorides are inexpensive, commercially available, and operationally simple solids or liquids that do not suffer from the same catalyst poisoning effects as thiols. In this innovative system, the reaction proceeds through a palladium-catalyzed cascade involving oxidative addition, alkene insertion, and carbonylation. Crucially, the use of molybdenum hexacarbonyl [Mo(CO)6] eliminates the need for external CO gas; it decomposes under the reaction conditions to release CO in situ while simultaneously acting as a reducing agent to maintain the catalytic cycle. This dual functionality streamlines the process, enhances safety, and improves overall reaction efficiency, making it an ideal candidate for high-purity pharmaceutical intermediate production.
Mechanistic Insights into Pd-Catalyzed Cyclization/Thiocarbonylation
The core of this synthetic strategy lies in the intricate interplay between the palladium catalyst, the phosphine ligand, and the unique reagents. The reaction initiates with the oxidative addition of the aryl iodide substrate to the active Pd(0) species, generated in situ from palladium acetate and tricyclohexylphosphine (PCy3). The bulky and electron-rich nature of PCy3 is critical for stabilizing the palladium center and facilitating the subsequent migratory insertion of the alkene moiety. Following the formation of the alkyl-palladium intermediate, the carbon monoxide released from the thermal decomposition of Mo(CO)6 inserts into the Pd-C bond. This step is pivotal for constructing the carbonyl functionality of the thioester. The sulfonyl chloride then participates in the cycle, likely undergoing desulfitative chlorination or direct interaction to introduce the sulfur component, ultimately leading to the formation of the indolinone ring via intramolecular cyclization.
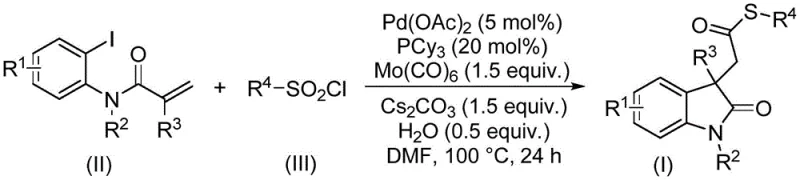
Understanding the impurity profile is essential for R&D directors focused on product quality. The mild reaction conditions (90-110°C) and the specific choice of base (cesium carbonate) help minimize side reactions such as homocoupling of the aryl iodide or polymerization of the alkene. The presence of water in the system, though in small amounts, may play a role in facilitating the hydrolysis of intermediate sulfonyl species or stabilizing the ionic transitions. The robustness of this catalytic system allows for a wide range of substituents on the aromatic ring (R1), the nitrogen atom (R2), and the sulfur source (R4), as demonstrated by the successful synthesis of various derivatives. This tolerance ensures that the final high-purity indolinone thioesters meet stringent quality specifications required for downstream drug synthesis.
How to Synthesize Indolinone Thioesters Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction parameters to maximize yield and purity. The process is designed to be straightforward, utilizing standard organic synthesis techniques without the need for exotic equipment. The key to success lies in the precise ratio of the palladium catalyst to the ligand and the adequate loading of the carbonyl source to drive the reaction to completion. Below is a summary of the standardized protocol derived from the patent data, which serves as a foundation for process optimization.
- Combine palladium acetate, tricyclohexylphosphine, molybdenum carbonyl, cesium carbonate, water, iodoarene, and sulfonyl chloride in a sealed tube.
- Add N,N-dimethylformamide (DMF) as the solvent and stir the mixture to ensure homogeneous dissolution of all reactants.
- Heat the reaction mixture to 90-110°C for approximately 24 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits beyond mere chemical elegance. The shift from thiols to sulfonyl chlorides fundamentally alters the cost structure and logistical footprint of the manufacturing process. By eliminating the need for specialized high-pressure CO reactors and mitigating the risks associated with toxic and malodorous thiol handling, the operational overhead is significantly reduced. This translates directly into a more competitive pricing model for the final intermediates. Furthermore, the reliance on commercially available, shelf-stable reagents ensures a consistent and reliable supply chain, reducing the risk of production delays caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the substitution of expensive or hazardous reagents with commodity chemicals. Sulfonyl chlorides are generally less expensive and easier to source in bulk quantities compared to specialized thiols. Additionally, the use of Mo(CO)6 as a solid CO surrogate removes the capital expenditure associated with high-pressure gas infrastructure and its ongoing maintenance. The simplified workup procedure, which involves basic filtration and standard chromatography, reduces labor costs and solvent consumption, contributing to substantial overall cost savings in the production of these valuable intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials. Iodoarenes and sulfonyl chlorides are produced by numerous chemical suppliers globally, ensuring that sourcing is not dependent on a single vendor. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the continuity of supply. This reliability is crucial for maintaining uninterrupted production schedules for downstream API manufacturing, thereby reducing lead times for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles. The avoidance of gaseous CO reduces the carbon footprint and safety hazards associated with the process. The reaction generates fewer hazardous byproducts compared to traditional thiol-based methods, simplifying waste treatment and disposal. The straightforward scale-up potential, evidenced by the successful synthesis of diverse derivatives in the patent, indicates that this process can be seamlessly transferred from gram-scale laboratory synthesis to multi-kilogram or ton-scale commercial production without significant re-engineering.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific applications, we have compiled answers to common questions regarding the reaction scope, safety, and operational details. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear picture of what to expect during process implementation.
Q: What is the primary advantage of using sulfonyl chlorides over thiols in this synthesis?
A: Sulfonyl chlorides avoid the catalyst poisoning issues commonly associated with thiols due to their strong thiolophilicity towards transition metals. Additionally, they are generally cheaper, easier to handle, and do not possess the unpleasant odor characteristic of many thiol reagents.
Q: What role does Molybdenum Hexacarbonyl play in the reaction mechanism?
A: Molybdenum hexacarbonyl [Mo(CO)6] serves a dual function: it acts as the solid source of carbon monoxide (CO) required for the carbonylation step, eliminating the need for hazardous high-pressure CO gas, and simultaneously acts as a reducing agent to regenerate the active palladium catalyst species.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes commercially available and inexpensive raw materials, operates at moderate temperatures (90-110°C), and employs a simple workup procedure involving filtration and column chromatography, making it highly amenable to scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolinone Thioester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced synthetic methodologies play in accelerating drug discovery and development. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN115403505A and is fully equipped to leverage this palladium-catalyzed thiocarbonylation route for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to manufacturing is seamless. Our state-of-the-art facilities are designed to handle complex organometallic reactions with precision, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the highest quality standards for every batch.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis. Whether you require custom synthesis of specific indolinone derivatives or a comprehensive evaluation of this route for your existing pipeline, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis, where we will evaluate your specific needs and provide detailed projections on efficiency gains. We encourage you to reach out today to obtain specific COA data and route feasibility assessments tailored to your project requirements.
