Advanced Photooxidative Synthesis of Quinoline-5,8-Diones for Scalable Pharmaceutical Manufacturing
Advanced Photooxidative Synthesis of Quinoline-5,8-Diones for Scalable Pharmaceutical Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for complex heterocyclic scaffolds. Patent CN1139575C introduces a transformative methodology for the preparation of quinoline-5,8-diones, a critical class of intermediates utilized in the synthesis of antimalarial agents and Maillard reaction inhibitors. This technology leverages a sophisticated photooxidation strategy that converts readily available 8-hydroxyquinoline derivatives into valuable quinone structures with high efficiency. By shifting away from traditional stoichiometric oxidants towards a catalytic, light-driven process, this innovation addresses significant pain points regarding toxicity, cost, and operational safety in modern chemical manufacturing. The following analysis details the mechanistic elegance and commercial viability of this approach for global supply chains.
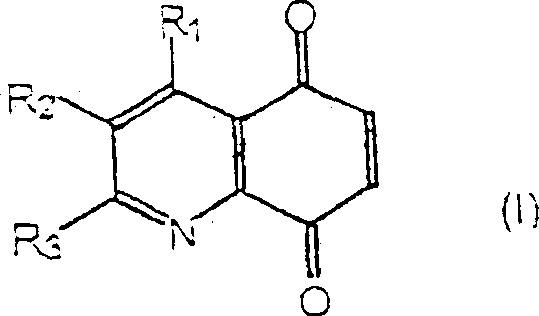
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinoline-5,8-diones has been fraught with significant chemical and economic challenges that hinder efficient large-scale production. Traditional pathways, such as those described by Bracher, often rely on the oxidation of 5-amino-8-hydroxyquinolines using heavy metal oxidants like dichromates. These reagents are not only highly toxic and carcinogenic, posing severe occupational health risks, but they also generate substantial quantities of hazardous chromium waste that requires expensive disposal protocols. Furthermore, these conventional routes frequently necessitate starting materials bearing multiple functional groups, which are inherently expensive and difficult to source in bulk quantities. The reliance on harsh oxidative conditions can also lead to poor selectivity, resulting in complex impurity profiles that complicate downstream purification and reduce overall process yield, thereby inflating the cost of goods for the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast, the methodology disclosed in CN1139575C offers a paradigm shift by utilizing a mild, catalytic photooxidation process that circumvents the drawbacks of legacy technologies. This novel approach employs molecular oxygen as the terminal oxidant, driven by visible light irradiation in the presence of catalytic amounts of photosensitizers such as tetraphenylporphine (TPP) or Rose Bengal. This transition to aerobic oxidation eliminates the need for stoichiometric heavy metal reagents, drastically simplifying the waste stream and enhancing the environmental profile of the synthesis. Moreover, the process utilizes simple 8-hydroxyquinoline precursors which are commercially abundant and cost-effective, removing the dependency on scarce, multi-functionalized starting materials. The reaction proceeds under ambient temperature conditions, typically between 15 to 25°C, which minimizes energy consumption and reduces the thermal stress on sensitive molecular architectures, ensuring higher fidelity in the final product structure.
Mechanistic Insights into Photosensitized Oxygenation
The core of this technological advancement lies in the generation of singlet oxygen through energy transfer from an excited state photosensitizer to ground-state triplet oxygen. When the reaction mixture containing the 8-hydroxyquinoline substrate and the photosensitizer is irradiated with visible light, the sensitizer absorbs photons and transitions to an excited triplet state. This excited species then transfers energy to dissolved molecular oxygen, generating highly reactive singlet oxygen (1O2). Unlike ground-state oxygen, singlet oxygen acts as a potent electrophile capable of undergoing cycloaddition or ene reactions with electron-rich aromatic systems. In this specific transformation, the singlet oxygen attacks the electron-rich quinoline ring system, specifically targeting the positions adjacent to the hydroxyl group, leading to the formation of a transient hydroperoxide intermediate. This mechanistic pathway is particularly elegant because it avoids the radical chain reactions often associated with autoxidation, providing greater control over the reaction trajectory and minimizing the formation of polymeric byproducts.
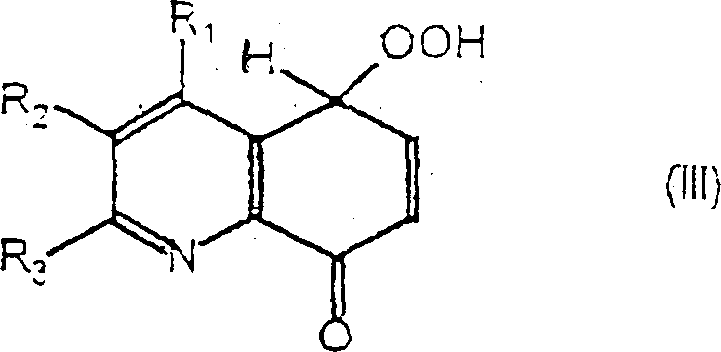
Following the initial oxygenation event, the stability and subsequent decomposition of the hydroperoxide intermediate are critical for the successful isolation of the target quinone. The patent data indicates that the formed hydroperoxide, characterized by the presence of an -OOH group on the quinoline ring, is relatively unstable and undergoes spontaneous decomposition upon stirring, particularly in the presence of drying agents like anhydrous sodium sulfate. This decomposition step effectively aromatizes the ring system and establishes the conjugated dione functionality essential for the biological activity of the final compound. Understanding this mechanism is vital for process optimization, as the rate of hydroperoxide formation versus its decomposition must be balanced to prevent the accumulation of potentially explosive peroxide species. The ability to drive this decomposition under mild conditions without requiring strong acids or bases further underscores the chemoselectivity of the process, preserving other sensitive functional groups such as esters, nitriles, or halides that may be present on the quinoline scaffold.
How to Synthesize Quinoline-5,8-Dione Efficiently
Implementing this photooxidative protocol requires careful attention to reaction parameters to ensure reproducibility and high purity. The general procedure involves dissolving the specific 8-hydroxyquinoline derivative in a suitable organic solvent, with dichloromethane being the preferred medium due to its ability to dissolve both the substrate and the photosensitizer effectively. A catalytic amount of tetraphenylporphine (approximately 0.5 mol%) is added to the solution, which is then placed in a Pyrex vessel to filter out UV radiation that might cause side reactions. The mixture is maintained at a controlled temperature of 20°C using a water bath while being continuously sparged with oxygen gas to maintain a high concentration of the oxidant. Irradiation is performed using a high-power xenon arc lamp equipped with filters to ensure only visible light greater than 495nm reaches the reaction mixture, preventing degradation of the sensitizer. Reaction progress is monitored via TLC until the starting material is consumed, typically requiring 2 to 8 hours depending on the electronic nature of the substituents.
- Dissolve the 8-hydroxyquinoline starting material in an organic solvent such as dichloromethane with a catalytic amount of photosensitizer like tetraphenylporphine.
- Irradiate the solution with visible light (>495nm) while sparging oxygen at room temperature (15-25°C) for 2 to 8 hours to form the hydroperoxide intermediate.
- Decompose the formed hydroperoxide by stirring with drying agents like sodium sulfate, followed by filtration and purification via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photooxidative technology presents a compelling value proposition centered on risk mitigation and operational efficiency. The elimination of hexavalent chromium and other heavy metal oxidants removes a significant regulatory burden associated with the handling, storage, and disposal of hazardous chemicals. This shift not only aligns with increasingly stringent global environmental regulations but also substantially reduces the overhead costs linked to waste management and environmental compliance auditing. By transitioning to a catalytic system driven by light and air, manufacturers can achieve a leaner operational model that requires less specialized infrastructure for hazard containment. This simplification of the chemical process directly translates to enhanced supply chain reliability, as the dependency on volatile markets for toxic reagents is removed, ensuring a more stable and predictable production schedule for critical pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the substitution of expensive, multi-step precursor synthesis with readily available commodity chemicals. Traditional methods often require 5-amino-8-hydroxyquinolines, which involve complex nitration and reduction sequences to produce, whereas this method starts from simple 8-hydroxyquinolines that are produced on a massive industrial scale. Additionally, the removal of heavy metal oxidants eliminates the costly downstream processing steps required to scrub trace metals from the final product to meet pharmaceutical purity standards. The use of ambient temperature conditions further drives down utility costs by removing the need for extensive heating or cryogenic cooling systems, resulting in a significantly lower energy footprint per kilogram of product manufactured.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures consistent output quality and volume. The reliance on molecular oxygen and visible light means that the key reagents are essentially inexhaustible and immune to supply chain disruptions that often plague specialty chemical markets. The mild reaction conditions also reduce the wear and tear on reactor equipment, extending asset life and minimizing unplanned maintenance downtime. This reliability is crucial for long-term supply agreements with multinational pharmaceutical companies, where consistency of supply is often valued higher than marginal price fluctuations. The ability to source starting materials from multiple global vendors further de-risks the supply chain, preventing single-source bottlenecks that could halt production lines.
- Scalability and Environmental Compliance: Scaling photochemical reactions has historically been a challenge due to light penetration limits, but modern flow chemistry and advanced reactor designs have largely overcome these barriers, making this technology viable for ton-scale production. The process generates minimal hazardous waste, primarily consisting of spent solvent which can be easily recovered and recycled, supporting a circular economy model within the manufacturing plant. This high level of environmental compliance facilitates faster regulatory approvals for new drug filings, as the impurity profile is cleaner and free from genotoxic metal residues. Consequently, this method supports the rapid commercialization of new therapeutic candidates by shortening the timeline from process development to GMP manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photooxidative synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process transfer and for procurement specialists assessing vendor capabilities. The answers reflect the specific advantages of the catalytic cycle and the structural scope of the substrates tolerated by this method.
Q: What are the primary advantages of this photooxidation method over traditional dichromate oxidation?
A: The photooxidation method eliminates the use of toxic heavy metal oxidants like dichromate, significantly reducing environmental hazards and waste treatment costs. Furthermore, it utilizes readily available 8-hydroxyquinoline precursors rather than expensive, functionally complex starting materials required by older methods.
Q: What specific reaction conditions are required for high yield conversion?
A: The process operates under mild conditions, specifically at room temperature (15-25°C) using visible light irradiation (>495nm) and continuous oxygen sparging. The reaction typically completes within 2 to 8 hours depending on the specific substituents on the quinoline ring.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, the use of ambient temperature and standard organic solvents like dichloromethane makes the process highly amenable to scale-up. The absence of extreme thermal conditions or hazardous reagents simplifies reactor design and enhances operational safety for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinoline-5,8-Dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of quinoline-5,8-dione meets the exacting standards required for pharmaceutical applications. Our commitment to process excellence means we can leverage advanced photochemical reactors to deliver this complex scaffold with superior consistency and reliability, supporting your pipeline from early-stage discovery through to market launch.
We invite you to collaborate with us to optimize your supply chain for these vital intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and security for your organization.
