Scalable Synthesis of 3-Acetylmercapto-2-methylpropanoic Acid for Commercial API Production
The pharmaceutical industry constantly seeks robust, cost-effective pathways for critical active pharmaceutical ingredient (API) intermediates, particularly for established drugs like Captopril. Patent CN111039838A introduces a transformative preparation method for 3-acetylmercapto-2-methylpropanoic acid, a pivotal building block in cardiovascular medicine. This technology addresses long-standing inefficiencies in legacy manufacturing by replacing hazardous, high-cost reagents with accessible inorganic alternatives. By leveraging a sequential hydrohalogenation and nucleophilic substitution strategy, the process achieves exceptional yields exceeding 90% in key steps while maintaining environmental compliance. For global procurement teams, this innovation represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced supply chain volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-acetylmercapto-2-methylpropanoic acid has relied heavily on the direct addition of thioacetic acid to methacrylic acid, a route fraught with significant operational and economic drawbacks. As illustrated in the conventional reaction pathway, this method necessitates the handling of thioacetic acid, a reagent known for its extreme volatility, pungent odor, and corrosive nature, which imposes severe restrictions on factory usage and worker safety. Furthermore, thioacetic acid is a relatively expensive specialty chemical compared to bulk inorganic salts, driving up the raw material costs substantially for large-scale production. The reliance on such sensitive reagents often complicates waste treatment protocols due to sulfur-containing byproducts, creating additional environmental burdens for manufacturing facilities striving for green chemistry standards.
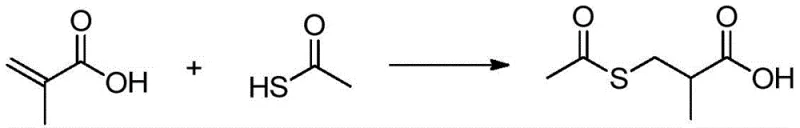
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent circumvents these issues by employing a three-step sequence starting from methacrylic acid and simple halogenated acids. This innovative route first converts methacrylic acid into a halo-intermediate, which is subsequently transformed into a mercapto-acid using sodium hydrosulfide or sodium sulfide before final acetylation. This strategic shift eliminates the need for thioacetic acid entirely, replacing it with abundant, low-cost inorganic sulfides that are easier to handle and store. The process flow demonstrates remarkable flexibility, allowing for the use of either hydrochloric or hydrobromic acid, and accommodates various solvent systems including water and DMF, thereby enhancing cost reduction in API manufacturing by optimizing solvent recovery and reuse cycles.
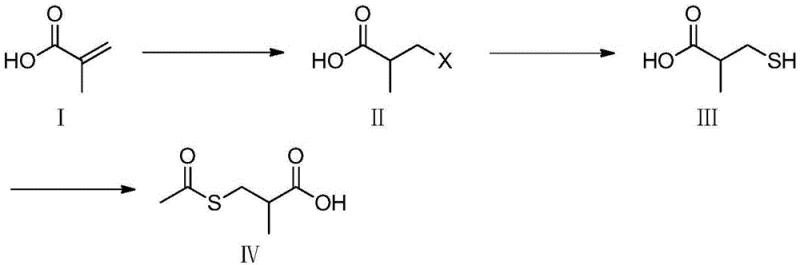
Mechanistic Insights into Copper-Catalyzed Nucleophilic Substitution
The core chemical transformation in this synthesis lies in the conversion of the halo-intermediate to the mercapto-acid, a step facilitated by a sophisticated copper-catalyzed nucleophilic substitution mechanism. The patent details the use of cuprous iodide (CuI) in conjunction with phase transfer catalysts like tetrabutylammonium bromide to drive the reaction between the organic halo-acid and inorganic sodium hydrosulfide. This catalytic system is crucial for overcoming the solubility mismatch between the organic substrate and the inorganic sulfide source, ensuring efficient mass transfer and high reaction kinetics. The presence of the copper catalyst likely activates the carbon-halogen bond, lowering the activation energy required for the nucleophilic attack by the hydrosulfide ion, which results in the observed high yields of over 90% for this critical step.
From an impurity control perspective, this mechanistic approach offers superior selectivity compared to non-catalyzed thermal substitutions which often lead to elimination byproducts or polymerization. The mild reaction temperatures, typically ranging from 65°C to 85°C, prevent the degradation of the sensitive carboxylic acid moiety and minimize the formation of disulfide impurities that can plague thiol chemistry. Furthermore, the subsequent acetylation step is carefully controlled under alkaline conditions or in organic solvents like dichloromethane to ensure the selective protection of the thiol group without affecting the carboxylic acid. This precise control over reaction parameters ensures that the final product meets the stringent purity specifications required for pharmaceutical applications, with gas chromatography data confirming purity levels above 98.5%.
How to Synthesize 3-Acetylmercapto-2-methylpropanoic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for producing this valuable intermediate with high efficiency and consistency. The process begins with the hydrohalogenation of methacrylic acid, followed by the copper-catalyzed sulfuration, and concludes with a straightforward acetylation. Each step has been optimized for yield and ease of workup, utilizing standard separation techniques such as liquid-liquid extraction and reduced pressure distillation. For detailed operational parameters, including specific molar ratios, temperature profiles, and workup procedures, please refer to the standardized synthesis guide below which encapsulates the critical process controls necessary for successful replication.
- React methacrylic acid with a halogenated acid (HCl or HBr) to form the corresponding halo-acid intermediate.
- Perform a copper-catalyzed nucleophilic substitution using sodium hydrosulfide or sodium sulfide to generate the mercapto-acid.
- Conduct an acetylation reaction using acetic anhydride or acetyl chloride under alkaline or organic solvent conditions to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers compelling strategic benefits that extend beyond simple unit price reductions. By shifting away from volatile organic sulfur reagents to stable inorganic salts, manufacturers can significantly mitigate supply risks associated with specialty chemical shortages. The use of commodity chemicals like methacrylic acid and hydrochloric acid ensures a robust supply base with multiple global sources, effectively reducing lead time for high-purity carboxylic acid derivatives and insulating the supply chain from market fluctuations. Additionally, the simplified waste profile and lower toxicity of reagents streamline regulatory compliance and reduce the overhead costs associated with environmental health and safety management.
- Cost Reduction in Manufacturing: The elimination of thioacetic acid, a high-cost reagent, fundamentally alters the cost structure of the synthesis, leading to substantial cost savings in raw material procurement. The ability to use water as a primary solvent in several steps further reduces expenditure on organic solvents and simplifies the energy-intensive solvent recovery processes typically required in fine chemical synthesis. Moreover, the high reaction yields reported in the patent minimize material loss, ensuring that a greater proportion of input materials are converted into saleable product, thereby maximizing overall process efficiency and margin potential.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on widely available bulk chemicals rather than niche intermediates that may have limited production capacity. The robustness of the reaction conditions, which do not require exotic catalysts or extreme pressures, means that production can be easily distributed across multiple manufacturing sites without significant requalification efforts. This flexibility enhances supply continuity, ensuring that downstream API production schedules are not disrupted by upstream intermediate shortages, a critical factor for maintaining just-in-time inventory models in the pharmaceutical sector.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard reactor configurations and ambient pressure conditions that are safe and easy to operate at multi-ton scales. The reduced generation of hazardous waste and the avoidance of noxious sulfur odors contribute to a smaller environmental footprint, aligning with modern sustainability goals and reducing the burden on effluent treatment plants. This environmental compatibility facilitates faster regulatory approvals and permits for new production lines, accelerating the time-to-market for generic versions of Captopril and related therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these details is essential for R&D teams planning technology transfer and for procurement teams negotiating supply agreements based on process capabilities.
Q: Why is this new synthesis route preferred over the traditional thioacetic acid method?
A: The traditional method relies on thioacetic acid, which is volatile, has a strong unpleasant odor, and is costly. The new route uses inexpensive, stable raw materials like methacrylic acid and inorganic sulfides, significantly improving operational safety and cost efficiency.
Q: What represents the key quality metric for this intermediate in the patent?
A: The patent highlights that the final product achieves a gas chromatography purity of greater than 98.5% through simple distillation, meeting the stringent requirements for downstream ACE inhibitor synthesis.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process utilizes common industrial reagents and mild reaction conditions (0-100°C) without requiring specialized high-pressure equipment, making it highly adaptable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Acetylmercapto-2-methylpropanoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of consistent quality and reliable supply in the pharmaceutical value chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet their exact volume requirements without compromise. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-acetylmercapto-2-methylpropanoic acid delivers the performance expected in downstream ACE inhibitor synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced process chemistry. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your project needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can support your long-term production goals and enhance your competitive position in the global market.
