Optimizing ADC Linker Production: A Technical Analysis of Patent CN113527418B for Commercial Scale-Up
Introduction to Next-Generation ADC Linker Technology
The rapid evolution of Antibody-Drug Conjugates (ADCs) has placed immense pressure on the supply chain for high-quality linkers, which are critical for maintaining stability in systemic circulation while ensuring efficient payload release. Patent CN113527418B introduces a robust, five-step synthetic methodology designed to address the persistent challenges of yield optimization and impurity control in pharmaceutical intermediates manufacturing. This technical disclosure outlines a precise route starting from Fmoc-glycyl-glycine (Compound A) and culminating in a maleimide-functionalized linker (Compound J), utilizing a unique copper-catalyzed oxidation strategy followed by sequential peptide couplings. For R&D directors and procurement specialists, understanding the mechanistic nuances of this patent is essential for evaluating its potential to streamline production workflows.
The significance of this invention lies not merely in the final structure but in the operational simplicity embedded within each transformation. By leveraging specific catalytic ratios, such as the optimized molar percentage of copper acetate relative to the starting material, the process mitigates the formation of difficult-to-remove byproducts that often plague traditional linker synthesis. Furthermore, the strategic implementation of crystallization as a primary purification tool at multiple intermediate stages (Compounds B, D, G, and H) offers a distinct advantage over chromatography-dependent routes. This approach directly translates to reduced solvent waste and lower operational expenditures, making it a compelling candidate for commercial scale-up of complex peptide linkers.
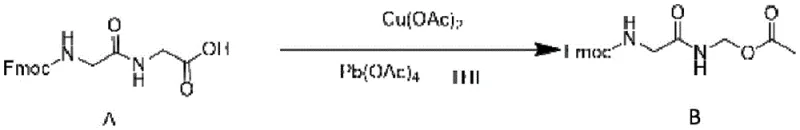
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ADC linkers has been fraught with inefficiencies, particularly regarding the introduction of the initial spacer arm and the subsequent peptide elongation. Conventional routes often rely on stoichiometric oxidants or harsh conditions that generate significant quantities of side products, necessitating extensive and costly purification steps like preparative HPLC. In many legacy processes, the removal of protecting groups such as Fmoc or Cbz can lead to racemization or incomplete deprotection, resulting in an impurity profile that complicates regulatory approval. Moreover, the reliance on column chromatography for intermediate purification creates a bottleneck in manufacturing throughput, increasing both the lead time and the environmental footprint of the production facility.
The Novel Approach
The methodology disclosed in CN113527418B circumvents these bottlenecks through a carefully engineered sequence that prioritizes crystallization-driven purification. The novel approach initiates with a copper-mediated transformation that effectively constructs the acetoxy-methyl scaffold with high selectivity, avoiding the generation of impurities that share similar mass characteristics with the target molecule. Subsequent steps utilize mild acid catalysts like PPTS for esterification and modern coupling reagents like HATU for amide bond formation, ensuring high fidelity in the peptide backbone construction. By integrating a two-step reaction protocol for the formation of Compound G, the process actively suppresses side reactions between the base (DBU) and the activated acid species, thereby preserving the integrity of the chiral centers and maximizing the overall yield of the final high-purity ADC linker.
Mechanistic Insights into Copper-Catalyzed Oxidative Decarboxylation
The cornerstone of this synthetic route is the initial transformation of Compound A to Compound B, which employs a copper(II) acetate catalyst in conjunction with lead tetraacetate. Mechanistically, this reaction likely proceeds through a radical decarboxylation pathway where the copper species facilitates the single-electron transfer necessary to cleave the carboxylic acid moiety of the glycine residue. The presence of lead tetraacetate serves as the terminal oxidant, regenerating the active copper species and providing the acetoxy group that functionalizes the alpha-position. This catalytic cycle is finely tuned by the molar ratio of copper to substrate, with the patent specifying an optimal range that balances reaction kinetics against the formation of over-oxidized byproducts. Such precision in catalyst loading is critical for maintaining a clean reaction profile, which is a prerequisite for the subsequent crystallization steps.
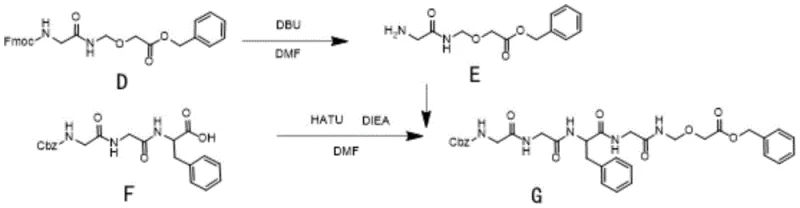
Impurity control is further enhanced in the later stages of the synthesis, specifically during the assembly of the tripeptide segment. The patent describes a sequential addition protocol where the deprotected amine (Compound E) is generated in situ using DBU before being introduced to the activated acid species (Compound F + HATU). This temporal separation prevents the base-induced degradation of the activated ester or the racemization of the phenylalanine residue, which are common failure modes in solid-phase and solution-phase peptide synthesis. By rigorously controlling the pH during the workup and employing specific solvent systems like ethyl acetate and methyl tert-butyl ether for anti-solvent crystallization, the process ensures that the final product meets the stringent purity specifications required for clinical-grade pharmaceutical intermediates.
How to Synthesize ADC Linker Efficiently
The synthesis of the target ADC linker involves a logical progression of functional group manipulations that transform simple amino acid derivatives into a complex, multifunctional molecule capable of bridging an antibody and a cytotoxic payload. The process begins with the oxidative modification of a dipeptide, followed by esterification to install the cleavable spacer, and concludes with the attachment of the maleimide warhead. Each step has been optimized to maximize recovery through precipitation or crystallization, minimizing the need for resource-intensive chromatographic separations. For process chemists looking to implement this route, attention to the specific solvent volumes and temperature controls detailed in the patent examples is paramount to reproducing the reported yields.
- Prepare Compound B via copper acetate-catalyzed oxidation of Fmoc-Gly-Gly using lead tetraacetate.
- Synthesize Compound D by reacting Compound B with benzyl glycolate using PPTS catalyst in DCM.
- Couple Compound E (derived from D) with Cbz-Gly-Gly-Phe using HATU/DIEA to form Compound G.
- Perform hydrogenolysis on Compound G using Pd/C to remove Cbz and benzyl groups, yielding Compound H.
- Conjugate Compound H with Maleimidohexanoate succinimidyl ester to obtain the final ADC Linker (Compound J).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers substantial strategic benefits, primarily driven by the simplification of the manufacturing workflow. The shift away from complex purification techniques towards crystallization-based isolation significantly reduces the consumption of expensive chromatography media and high-grade solvents, leading to a direct reduction in the cost of goods sold (COGS). Furthermore, the use of commercially available and relatively stable reagents such as copper acetate and HATU ensures a reliable supply of raw materials, mitigating the risk of production delays caused by specialized reagent shortages. This robustness is essential for maintaining continuity in the supply of critical pharmaceutical intermediates to downstream drug manufacturers.
- Cost Reduction in Manufacturing: The elimination of extensive column chromatography steps in favor of crystallization drastically lowers operational costs associated with solvent recovery and waste disposal. By optimizing catalyst loading and reaction times, the process achieves high conversion rates, reducing the amount of starting material lost to side reactions. This efficiency translates into significant economic savings over large production batches, making the final linker more cost-competitive in the global market without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on standard organic synthesis operations such as reflux, filtration, and drying means that the process can be executed in a wide range of multipurpose chemical facilities without requiring specialized equipment. This flexibility allows for diversified manufacturing strategies, including the potential for multi-vendor sourcing, which strengthens the resilience of the supply chain against geopolitical or logistical disruptions. The consistent quality of the intermediates also reduces the need for extensive incoming quality control testing, speeding up the release of materials for production.
- Scalability and Environmental Compliance: The protocol is inherently scalable, as demonstrated by the gram-to-kilogram progression described in the examples, with no indication of exothermic runaways or hazardous gas evolution that would complicate scale-up. The reduced solvent intensity and the ability to recycle mother liquors from crystallization steps align with green chemistry principles, helping manufacturers meet increasingly strict environmental regulations. This sustainability profile is becoming a key differentiator for suppliers seeking partnerships with major pharmaceutical companies committed to reducing their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ADC linker synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: How does this patent address impurity control in ADC linker synthesis?
A: The patent utilizes a specific copper acetate catalyst system in Step 1 to avoid impurities that match the main component's mass. Additionally, Step 3 employs a two-stage reaction sequence to minimize side reactions between Compound F and DBU, ensuring higher purity before crystallization.
Q: What are the key advantages of the purification method described?
A: Unlike traditional methods relying heavily on column chromatography, this process emphasizes crystallization (e.g., in DMF or ethyl acetate/MTBE systems) at multiple stages (Compounds B, D, G, H). This significantly simplifies downstream processing and reduces solvent consumption.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the protocol uses standard reagents like Cu(OAc)2, Pb(OAc)4, and HATU under manageable conditions (e.g., 45-65°C reflux). The reliance on filtration and crystallization rather than complex separations makes it highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ADC Linker Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality linkers play in the success of next-generation ADC therapies. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of high-purity ADC linker we deliver meets the exacting standards required for clinical and commercial applications. Our commitment to technical excellence allows us to navigate the complexities of peptide coupling and sensitive functional group manipulation with precision.
We invite you to collaborate with us to leverage this advanced synthetic technology for your drug development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of reliability, quality, and scientific innovation. Let us help you accelerate your ADC pipeline with a supply partner dedicated to your success.
