Revolutionizing Febuxostat Impurity Production: A High-Yield Commercial Synthesis Strategy
The pharmaceutical industry constantly seeks efficient pathways for synthesizing critical reference standards and impurities to ensure drug safety and regulatory compliance. Patent CN110878064A, published in March 2020, introduces a transformative high-yield synthesis method for a specific impurity of febuxostat, a potent xanthine oxidase inhibitor used in gout treatment. This technical disclosure addresses a longstanding bottleneck in the production of this complex pharmaceutical intermediate, where traditional methods suffered from abysmally low overall yields. By strategically reordering the synthetic steps and optimizing reaction conditions, the inventors have achieved a total yield of approximately 43%, a staggering improvement over the prior art's mere 0.2%. This breakthrough not only facilitates better quality control for febuxostat manufacturers but also opens new avenues for cost-effective production of related thiazole-based scaffolds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
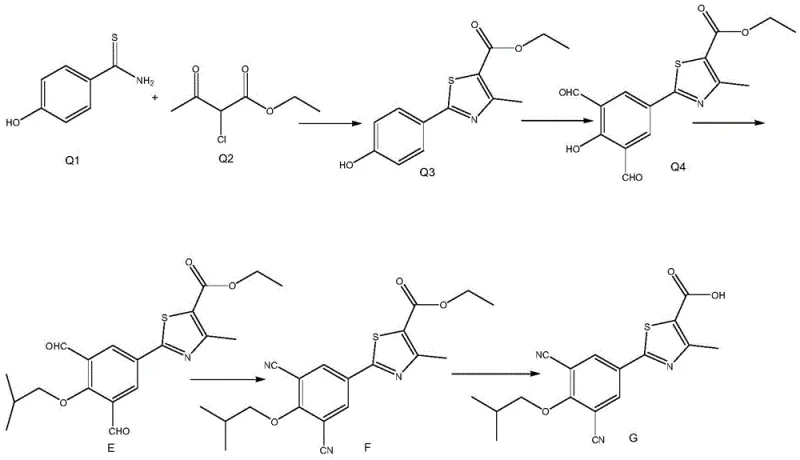
The historical approach to synthesizing this specific febuxostat impurity, as detailed in the background section of the patent, relies on a linear sequence that encounters severe steric and electronic hurdles early in the process. The conventional route begins with the formation of the thiazole ring (Q3) from precursors Q1 and Q2, which proceeds reasonably well with a 90% yield. However, the subsequent step involves introducing two aldehyde groups onto the benzene ring of this already bulky thiazole-substituted intermediate (Q3) using hexamethylenetetramine (HMTA). This formylation step is critically inefficient, yielding only about 3% of the desired dialdehyde Q4 due to significant steric hindrance imposed by the adjacent thiazole moiety. Furthermore, the following etherification step to convert Q4 to intermediate E suffers from a similarly poor yield of only 10%, creating a compounding effect that renders the entire process economically unviable for large-scale operations.
The Novel Approach
In stark contrast, the novel methodology described in CN110878064A ingeniously bypasses these steric constraints by reversing the order of functional group installation. Instead of attempting to formylate a crowded thiazole-substituted benzene ring, the new process starts with a simpler phenol derivative (P1) and immediately performs the Duff reaction with HMTA to introduce the two aldehyde groups, generating intermediate P2 with an impressive yield of 85% to 90%. This early-stage functionalization takes advantage of the lower steric hindrance of the starting material. Subsequently, the thiazole ring component (P3) is coupled to this pre-functionalized core via a palladium-catalyzed reaction, achieving a robust 70% yield. This strategic pivot transforms a process previously plagued by single-digit yields into a highly efficient, scalable workflow suitable for industrial application.
Mechanistic Insights into Pd-Catalyzed Coupling and Formylation
The core of this synthetic advancement lies in the mechanistic understanding of electrophilic aromatic substitution and transition metal catalysis. In the initial step, the reaction of P1 with HMTA in trifluoroacetic acid (TFA) at 120°C follows the classic Duff reaction mechanism. The acidic conditions facilitate the formation of an iminium ion from HMTA, which acts as a potent electrophile attacking the ortho positions of the phenol ring relative to the hydroxyl group. Because the starting phenol P1 lacks the bulky thiazole substituent at this stage, the ortho positions are highly accessible, allowing for the efficient introduction of two formyl groups to create the dialdehyde P2. This contrasts sharply with the prior art where the thiazole ring blocked access to these reactive sites, drastically reducing the reaction rate and yield.
Following the formylation, the coupling of P2 with the thiazole ester P3 represents a sophisticated application of palladium chemistry. The reaction utilizes Pd(dppf)Cl2 as the primary catalyst alongside cuprous bromide (CuBr) and potassium bicarbonate in a toluene solvent system. The presence of isobutyric acid as an additive is crucial, likely serving to solubilize the inorganic bases and stabilize the catalytic cycle. The mechanism likely involves an oxidative addition of the aryl halide (from P3) to the Pd(0) species, followed by transmetallation with the organometallic species derived from P2, and finally reductive elimination to forge the carbon-carbon bond. This step achieves a 70% yield, demonstrating that coupling a functionalized benzene ring with the thiazole fragment is far more kinetically favorable than attempting to functionalize the ring after the coupling has occurred.
How to Synthesize Febuxostat Specific Impurity Efficiently
The synthesis of this high-value pharmaceutical intermediate requires precise control over reaction parameters to maximize the benefits of the new route. The process is divided into four distinct stages: initial formylation, palladium-catalyzed coupling, oxime formation, and final hydrolysis. Each step has been optimized in the patent examples to ensure purity levels exceeding 99%, which is critical for reference standards. The detailed standardized synthesis steps, including exact reagent ratios, temperature profiles, and workup procedures, are outlined in the guide below to assist R&D teams in replicating this high-yield protocol.
- Perform Duff reaction on starting phenol P1 with HMTA in TFA at 120°C to generate dialdehyde P2 with 85% yield.
- Execute Pd-catalyzed coupling between P2 and thiazole ester P3 using CuBr and isobutyric acid to form intermediate E.
- Convert intermediate E to oxime F using hydroxylamine hydrochloride in formic acid, followed by hydrolysis to final impurity G.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from the conventional 0.2% yield process to this new 43% yield methodology represents a paradigm shift in cost structure and supply reliability. The primary driver of value here is the drastic reduction in raw material waste. In the old process, nearly all starting materials were lost during the low-yield formylation and etherification steps, necessitating the processing of massive quantities of feedstock to obtain negligible amounts of product. The new route eliminates this inefficiency, meaning that the same amount of final product can be generated with a fraction of the input materials, leading to substantial cost savings in raw material procurement.
- Cost Reduction in Manufacturing: The economic impact of increasing the total yield from 0.2% to 43% cannot be overstated in terms of unit cost. By avoiding the bottlenecks that required extensive recycling or disposal of failed batches, the manufacturing overhead per kilogram of product is significantly reduced. Furthermore, the new route utilizes cheap and readily available starting materials like P1 and P3, avoiding the need for expensive, custom-synthesized precursors that might have been required to bypass the steric issues in the old route. The elimination of difficult purification steps, which often involve costly chromatography or multiple recrystallizations to remove complex byproduct mixtures, further streamlines the operational expenditure.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by processes that rely on low-yield steps, as any slight deviation in reaction conditions can result in total batch failure. The robust nature of the new synthesis, with individual step yields ranging from 70% to 90%, provides a much wider operating window for manufacturing teams. This reliability ensures that delivery schedules can be met consistently without the risk of unexpected production delays caused by poor conversion rates. Additionally, the use of common solvents like toluene, ethanol, and dichloromethane, along with standard catalysts, ensures that the supply chain is not dependent on niche or hard-to-source reagents.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the new method offers distinct advantages. The high atom economy resulting from improved yields means less chemical waste is generated per unit of product, simplifying wastewater treatment and hazardous waste disposal protocols. The patent explicitly notes that the method is suitable for large-scale synthesis, indicating that the reaction exotherms and mixing requirements have been managed effectively in the examples provided. This scalability allows manufacturers to respond quickly to surges in demand for febuxostat quality control materials without needing to build entirely new production lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical disclosures within patent CN110878064A, providing clarity on the feasibility and advantages of adopting this new methodology for producing febuxostat impurities.
Q: Why does the new synthesis route offer significantly higher yields than conventional methods?
A: The new route reverses the synthesis order by introducing aldehyde groups early on a less sterically hindered phenol ring (P1), achieving 85% yield compared to the difficult 3% yield of formylating the bulky thiazole-substituted ring in the old method.
Q: What are the critical reaction conditions for the coupling step?
A: The coupling reaction requires Pd(dppf)Cl2 as a catalyst, cuprous bromide as a co-catalyst, and isobutyric acid as an additive in toluene solvent at 110°C for 16 hours to ensure optimal conversion.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly states the method is suitable for large-scale synthesis due to the use of cheap, readily available starting materials and the elimination of difficult purification steps associated with low-yield intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Febuxostat Impurity Supplier
The technical advancements detailed in this report underscore the complexity and precision required to produce high-purity pharmaceutical intermediates like the febuxostat specific impurity. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate such patented laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the lab are maintained at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch meets the exacting standards required for regulatory submissions and drug safety testing.
We invite global pharmaceutical partners to collaborate with us to leverage this high-yield synthesis technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your procurement strategy is built on the most efficient and reliable chemical foundations available in the market.
