Advanced Synthesis of Benzofuranone Derivatives for High-Value Pharmaceutical Applications
Advanced Synthesis of Benzofuranone Derivatives for High-Value Pharmaceutical Applications
The pharmaceutical and agrochemical industries are constantly seeking robust scaffolds that offer high biological activity combined with synthetic accessibility. Patent CN103864734A introduces a groundbreaking methodology for the preparation of novel substituted 4,6-dihydroxy-3(2H)-benzofuranone derivatives, a class of compounds structurally related to aurones. These molecules are not merely academic curiosities; they represent a critical frontier in the development of anti-tumor agents, antioxidants, and herbicides. By leveraging a strategic sequence of Friedel-Crafts acylation, base-catalyzed cyclization, and aldol condensation, this technology provides a reliable pathway to access complex heterocyclic systems that were previously difficult to synthesize with high regioselectivity. For R&D directors and procurement managers alike, understanding the nuances of this synthesis is key to securing a stable supply of high-purity intermediates for next-generation drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of aurone and benzofuranone derivatives has relied heavily on extraction from natural plant sources such as flowers and fruits, where these secondary metabolites occur in trace amounts. This reliance on natural occurrence creates severe bottlenecks for industrial application, including inconsistent batch-to-batch quality, seasonal availability issues, and exorbitant costs associated with isolation and purification. Furthermore, structural modification of naturally extracted compounds is often chemically challenging due to the presence of multiple reactive hydroxyl groups that require tedious protection and deprotection strategies. Traditional synthetic routes often suffer from poor regioselectivity during ring closure, leading to complex mixtures of isomers that drastically reduce overall yield and complicate downstream processing, thereby inflating the cost of goods sold for any final active pharmaceutical ingredient derived from these cores.
The Novel Approach
The methodology outlined in the patent data revolutionizes this landscape by establishing a linear, high-yielding synthetic route starting from the inexpensive and commercially abundant raw material, phloroglucinol. This approach bypasses the unpredictability of natural extraction entirely, offering a fully synthetic pathway that allows for precise engineering of the molecular architecture. By systematically introducing functional groups through controlled electrophilic substitutions and condensations, manufacturers can access a diverse library of derivatives with specific electronic and steric properties tailored for biological targets. This shift from extraction to rational synthesis ensures that supply chains are decoupled from agricultural variables, providing a consistent, scalable source of material that meets the rigorous purity standards required for clinical trial applications and commercial manufacturing.
Mechanistic Insights into the Multi-Step Synthetic Cascade
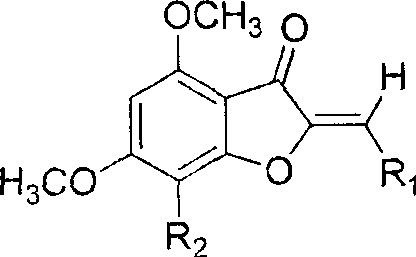
The core of this technological advancement lies in the meticulous orchestration of five distinct chemical transformations, beginning with a Friedel-Crafts acylation that sets the foundation for the entire molecular framework. In this initial step, phloroglucinol reacts with chloroacetyl chloride in the presence of aluminum chloride, a Lewis acid catalyst, to form 2-chloro-1-(2,4,6-trihydroxyphenyl)ethanone. This reaction is critical because it installs the two-carbon side chain necessary for subsequent ring closure while maintaining the symmetry of the phloroglucinol core. Following acylation, the process moves to a base-catalyzed intramolecular cyclization using sodium acetate in methanol. This elegant transformation closes the furan ring, generating the 4,6-dihydroxy-3(2H)-benzofuranone scaffold with impressive efficiency, reportedly achieving yields as high as 86 percent in laboratory settings.
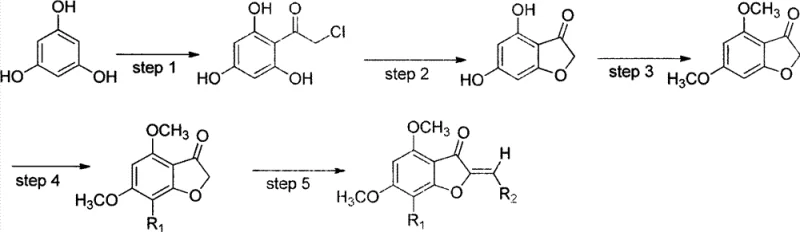
Subsequent steps focus on functionalizing this core to enhance its utility as a building block. The hydroxyl groups at the 4 and 6 positions are methylated using methyl iodide and potassium carbonate, which serves a dual purpose: it protects these sensitive positions from unwanted side reactions and modulates the lipophilicity of the final molecule. A crucial innovation in this pathway is the regioselective halogenation at the 7-position using N-iodosuccinimide. This introduces a heavy atom handle (iodine or bromine) that is invaluable for further cross-coupling reactions or for enhancing binding affinity in biological assays through halogen bonding interactions. Finally, the synthesis culminates in an aldol condensation with various aromatic aldehydes under basic conditions, forming the exocyclic double bond characteristic of aurones. This final step allows for immense structural diversity, enabling the rapid generation of analog libraries for structure-activity relationship studies without altering the core synthetic infrastructure.
How to Synthesize 4,6-Dimethoxy-7-halo-3(2H)-benzofuranone Efficiently
Executing this synthesis requires careful attention to reaction conditions, particularly regarding moisture control during the Friedel-Crafts acylation and temperature management during the cyclization phase. The protocol dictates the use of anhydrous solvents and inert atmospheres to prevent catalyst deactivation and hydrolysis of the acid chloride. While the laboratory examples demonstrate the feasibility on a gram scale, transitioning to pilot and commercial scales necessitates robust engineering controls to manage the exothermic nature of the acylation and the handling of volatile solvents like dichloromethane and methanol. The detailed standardized operating procedures for scaling this pathway from 100 kgs to 100 MT annual production are critical for ensuring safety and consistency.
- Perform Friedel-Crafts acylation of phloroglucinol with chloroacetyl chloride using aluminum chloride catalyst in 1,2-dichloroethane.
- Execute base-catalyzed cyclization of the acylated intermediate using sodium acetate in anhydrous methanol to form the benzofuranone core.
- Conduct electrophilic substitution via methylation using methyl iodide and potassium carbonate in DMF to protect hydroxyl groups.
- Introduce a halogen handle (iodine or bromine) at the 7-position using N-iodosuccinimide or similar halogenating agents.
- Finalize the structure through aldol condensation with various aromatic aldehydes under basic conditions to yield the target aurone derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic route offers profound strategic advantages beyond mere technical feasibility. The primary driver of value is the utilization of phloroglucinol, a commodity chemical with a stable global supply chain, as the starting material. Unlike specialized natural extracts that are subject to harvest fluctuations and geopolitical instability, phloroglucinol is produced via established industrial processes, ensuring long-term availability and price stability. This foundational security allows for accurate long-term forecasting and budget planning, eliminating the volatility often associated with bio-sourced intermediates. Furthermore, the high yields reported in the early stages of the synthesis (consistently above 85 percent) mean that less raw material is wasted, directly translating to a lower cost basis per kilogram of the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in favor of common Lewis acids like aluminum chloride significantly reduces the cost of reagents. Additionally, the high atom economy of the cyclization and condensation steps minimizes waste generation, which lowers the burden on waste treatment facilities and reduces disposal costs. By avoiding complex chromatographic purifications in favor of crystallization and filtration, the process becomes inherently more suitable for large-scale batch processing, driving down operational expenditures associated with solvent consumption and energy usage.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis allows for the stocking of key intermediates, such as the methylated benzofuranone core, which can be rapidly converted into various final derivatives based on demand. This flexibility acts as a buffer against market fluctuations, enabling suppliers to respond quickly to urgent requests from pharmaceutical partners without restarting the entire synthesis from scratch. The robustness of the reaction conditions also means that production is less susceptible to minor variations in utility supply or environmental conditions, ensuring consistent on-time delivery performance.
- Scalability and Environmental Compliance: The synthetic route avoids the use of highly toxic heavy metals or persistent organic pollutants, aligning well with modern green chemistry principles and stringent environmental regulations. The solvents used, such as methanol and ethyl acetate, are readily recyclable, further enhancing the sustainability profile of the manufacturing process. This compliance reduces the regulatory risk for downstream customers, facilitating smoother approval processes for new drug applications that rely on these intermediates, and positioning the supply chain as a partner in sustainable development goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzofuranone derivatives. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: What are the primary biological activities associated with these benzofuranone derivatives?
A: According to patent CN103864734A, these derivatives exhibit significant potential as anti-tumor agents, showing selective inhibitory effects against K562, HepG2, and HT-29 cell lines. Additionally, they possess antioxidant and herbicidal properties, making them versatile candidates for both pharmaceutical and agrochemical development.
Q: How does this synthetic route improve upon traditional extraction methods?
A: Traditional isolation from natural plant sources is limited by low abundance and complex purification requirements. This synthetic approach utilizes readily available phloroglucinol as a starting material, offering a scalable, reproducible pathway with high yields (up to 86% in key steps) and precise control over substitution patterns, ensuring consistent supply chain reliability.
Q: Is the halogenation step compatible with large-scale manufacturing?
A: Yes, the halogenation step utilizes standard reagents like N-iodosuccinimide in DMF under mild conditions (room temperature). This avoids the need for extreme temperatures or pressures, facilitating easier heat management and safety protocols during commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Dihydroxy-3(2H)-benzofuranone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the reliability and quality of the underlying chemical supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 4,6-dihydroxy-3(2H)-benzofuranone derivatives meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance ensures that your R&D timelines are protected from supply-related delays.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Whether you require custom synthesis of specific analogs or bulk supply of the core scaffold, our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your path to market while optimizing your overall production costs.
