Advanced Synthetic Route for High-Purity Decaprenol: A Scalable Solution for Coenzyme Q10 Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical intermediates like Decaprenol, the essential side-chain precursor for Coenzyme Q10 synthesis. Patent CN1226260C introduces a transformative methodology that addresses long-standing challenges in terpenoid synthesis, specifically focusing on the construction of the all-trans-polyisoprenoid chain. This innovation leverages a sophisticated combination of in-situ halogen exchange and reductive desulfonylation to achieve high yields and exceptional purity. Unlike earlier methods that struggled with stereoselectivity or relied on hazardous reagents, this approach utilizes stable, industrially available starting materials to construct the complex carbon skeleton efficiently. The strategic use of a chloro-intermediate, activated via halogen exchange, represents a significant leap forward in process chemistry, ensuring that the final product meets the rigorous standards required for pharmaceutical applications.

Decaprenol, chemically known as (2E, 6E, 10E, 14E, 18E, 22E, 26E, 30E, 34E)-3, 7, 11, 15, 19, 23, 27, 31, 35, 39-decamethyl-2, 6, 10, 14, 18, 22, 26, 30, 34, 38-tetracontadecaen-1-ol, serves as the vital lipophilic tail for Coenzyme Q10, a molecule indispensable for mitochondrial energy production and cellular health. As the global demand for CoQ10 surges due to its therapeutic potential in treating cardiovascular diseases and neurodegenerative disorders, the pressure on supply chains to deliver high-quality Decaprenol has never been higher. Traditional extraction from natural sources is insufficient due to extremely low content, making total synthesis the only viable commercial route. This patent provides a blueprint for a reliable pharmaceutical intermediate supplier to meet this demand by offering a route that balances economic feasibility with chemical precision, ensuring a steady flow of this critical building block for the nutraceutical and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of long-chain polyisoprenoids like Decaprenol has been plagued by significant technical hurdles that impede commercial scale-up. Early approaches, such as the C2+C3 synthesis sequence described by R.Ruegg, suffered from a lack of stereoselectivity, resulting in a mixture of cis-trans isomers that necessitated complex and yield-reducing separation processes. Furthermore, these routes often relied on dangerous reagents like sodium acetylide, posing severe safety risks in a manufacturing environment. Subsequent improvements attempted to address stereochemistry but introduced new bottlenecks; for instance, methods utilizing 4-bromo-3-methyl-2-buten-1-ol acetate faced critical supply chain issues because this key intermediate is chemically unstable, difficult to preserve, and hard to synthesize on an industrial scale. Additionally, the widespread use of sodium-amalgam for reductive desulfonylation introduced unacceptable levels of toxic mercury residues, rendering such processes unsuitable for drug substance manufacturing under modern GMP guidelines.
The Novel Approach
The methodology outlined in CN1226260C effectively dismantles these barriers by substituting problematic reagents with stable, commercially viable alternatives while maintaining high stereochemical integrity. By employing 4-chloro-3-methyl-2-buten-1-ol acetate, a compound that is economically feasible and stable for long-term storage, the process eliminates the supply volatility associated with bromo-analogs. The innovation lies in the clever activation of this less reactive chloro-species through an in-situ Finkelstein halogen exchange, allowing the condensation to proceed smoothly without compromising safety or cost. Moreover, the replacement of toxic mercury-based reducers with a lithium-methylamine system ensures that the final product is free from heavy metal contamination, a critical requirement for high-purity pharmaceutical intermediates. This holistic improvement in reagent selection and reaction design paves the way for cost reduction in pharmaceutical intermediate manufacturing by simplifying post-processing and waste treatment.
Mechanistic Insights into Reductive Desulfonylation and Halogen Exchange
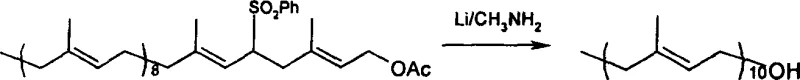
The core of this synthetic strategy relies on a dual-mechanism approach that maximizes reactivity while minimizing side reactions. In the first stage, the condensation of solanesyl sulfone with the chloro-acetate is facilitated by potassium tert-butoxide in a THF/DMF solvent system. Crucially, the addition of sodium iodide or sodium bromide triggers a Finkelstein reaction, converting the less reactive chloro-group into a highly reactive iodo- or bromo-species in situ. This dynamic equilibrium ensures that the nucleophilic attack by the sulfone carbanion occurs rapidly and efficiently, driving the formation of 5-sulfonyldecaprenyl acetate with yields reaching approximately 85% to 86%. The choice of potassium tert-butoxide over sodium tert-butoxide further enhances the reaction kinetics due to its higher basicity and solubility profile in the chosen solvent matrix, ensuring complete deprotonation of the sulfone precursor.
Following the condensation, the removal of the sulfone directing group is achieved through a dissolving metal reduction using lithium in liquid methylamine at cryogenic temperatures ranging from -78°C to -60°C. This specific reductive condition is paramount for preserving the delicate all-trans geometry of the polyisoprene chain. Unlike harsher reducing agents that might cause double bond migration or elimination side products, the lithium-methylamine system selectively cleaves the carbon-sulfur bond via a single-electron transfer mechanism. This precision prevents the formation of unwanted isomers and ensures that the terminal hydroxyl group is revealed without protecting group complications, as the acetate is concurrently hydrolyzed or managed during workup. The result is a clean conversion to Decaprenol with high purity, eliminating the need for extensive chromatographic purification and significantly streamlining the downstream processing workflow.
How to Synthesize Decaprenol Efficiently
The synthesis of Decaprenol via this patented route involves a carefully controlled two-step sequence that prioritizes safety and yield. The process begins with the condensation reaction where precise molar ratios of solanesyl sulfone, the chloro-acetate intermediate, and the halogen exchange catalyst are maintained in a mixed solvent system to optimize the in-situ activation. Following the isolation of the sulfone intermediate, the subsequent reduction step requires strict temperature control to manage the exothermic nature of the lithium reaction while preventing thermal degradation of the sensitive polyisoprenoid chain. Detailed standardized operating procedures for scaling this pathway from laboratory to commercial production are essential for maintaining batch-to-batch consistency.
- Condense solanesyl sulfone with 4-chloro-3-methyl-2-buten-1-ol acetate using potassium tert-butoxide and sodium iodide in THF/DMF to form 5-sulfonyldecaprenyl acetate.
- Perform reductive desulfonylation on the intermediate using lithium metal in liquid methylamine at temperatures between -78°C and -60°C.
- Quench the reaction with isoprene, methanol, and ammonium chloride, then extract and purify to obtain high-purity Decaprenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers profound strategic advantages that extend beyond simple chemical yield. The shift from unstable bromo-intermediates to stable chloro-precursors fundamentally alters the logistics of raw material management, allowing for bulk purchasing and long-term inventory storage without the risk of degradation. This stability translates directly into enhanced supply chain reliability, as manufacturers are no longer held hostage by the short shelf-life of critical reagents. Furthermore, the elimination of toxic mercury reagents drastically simplifies environmental compliance and waste disposal protocols, reducing the regulatory burden and associated costs of hazardous waste treatment. These factors combine to create a more resilient and cost-effective manufacturing ecosystem capable of sustaining high-volume production schedules.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive and difficult-to-handle reagents with commodity chemicals. By utilizing 4-chloro-3-methyl-2-buten-1-ol acetate, which is industrially feasible and cheaper than its bromo-counterpart, the raw material cost base is significantly lowered. Additionally, the avoidance of sodium-amalgam removes the need for specialized mercury containment and remediation infrastructure, leading to substantial capital expenditure savings. The high yields reported in the patent examples, consistently above 85%, further contribute to cost efficiency by maximizing the throughput of valuable solanesyl starting materials and minimizing waste generation per kilogram of product.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for any reliable pharmaceutical intermediate supplier, and this method secures that metric through reagent stability. The key chloro-intermediate is not only easier to synthesize but also possesses superior storage characteristics, mitigating the risk of production stoppages due to spoiled raw materials. This robustness allows for more flexible scheduling and larger batch sizes, ensuring that downstream CoQ10 manufacturers receive their shipments on time. The use of common solvents like THF and DMF, along with readily available lithium metal, further insulates the supply chain from the volatility associated with exotic or highly regulated specialty chemicals.
- Scalability and Environmental Compliance: Scaling complex polymer additives or long-chain terpenoids often encounters heat transfer and mixing limitations, but the mild conditions of this reaction (-15°C to 10°C for condensation, -78°C for reduction) are well within the capabilities of standard industrial reactors. The absence of heavy metals like mercury simplifies the environmental impact assessment, making it easier to obtain necessary permits for commercial scale-up of complex pharmaceutical intermediates. The cleaner reaction profile reduces the load on wastewater treatment facilities and lowers the overall environmental footprint of the manufacturing site, aligning with modern green chemistry principles and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decaprenol synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this route into their existing production lines.
Q: Why is the chloro-intermediate preferred over the bromo-intermediate in this synthesis?
A: The 4-chloro-3-methyl-2-buten-1-ol acetate is industrially economically feasible, chemically stable, and easy to store, whereas the bromo-analog is unstable and difficult to preserve, limiting large-scale application.
Q: How does this method improve safety compared to traditional routes?
A: This method eliminates the use of toxic sodium-amalgam (Na/Hg) reducing agents and dangerous reagents like diisopropylaluminum hydride, replacing them with a safer lithium-methylamine system.
Q: What is the stereochemical outcome of this synthetic route?
A: The process maintains the all-trans configuration of the polyisoprene chain, avoiding the formation of cis-trans isomer mixtures that require difficult separation in older C2+C3 synthesis sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decaprenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and robust infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of the lithium-methylamine reduction are managed with precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying the all-trans stereochemistry and absence of heavy metal residues, guaranteeing that every batch of Decaprenol meets the exacting standards required for Coenzyme Q10 synthesis. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients to bring life-saving therapies to market faster.
We invite global partners to collaborate with us to leverage this advanced synthetic technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project is built on a foundation of chemical excellence and supply chain security. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing.
