Advancing Fullerene Functionalization: A Novel One-Step Route to N-Phenyl [60] Fullerene Pyrrolines
The landscape of fullerene functionalization has long been dominated by challenges regarding regioselectivity and the limited scope of amine substrates. A significant breakthrough in this domain is detailed in patent CN113004190B, which discloses a robust preparation method for N-phenyl [60] fullerene pyrroline derivatives. This innovation addresses a critical gap in the synthesis of organic photovoltaic materials by enabling the direct use of aromatic primary amines, a substrate class that has historically proven recalcitrant in standard cycloaddition protocols. By leveraging a synergistic catalytic system comprising manganese acetate and DMAP, this methodology achieves a streamlined one-step transformation under aerobic conditions. For R&D directors and procurement specialists seeking a reliable fullerene derivative supplier, understanding the mechanistic nuances and operational simplicity of this patent is essential for evaluating its potential in next-generation electronic material manufacturing.
![General reaction scheme for N-phenyl [60] fullerene pyrroline synthesis using aldehyde and aromatic amine](/insights/img/n-phenyl-fullerene-pyrroline-synthesis-pharma-supplier-20260315125526-02.webp)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art in fullerene functionalization, particularly the synthesis of fulleropyrrolidines and pyrrolines, has heavily relied on copper-mediated pathways. As highlighted in the background of the patent, previous studies utilizing copper(II) acetate demonstrated significant limitations when attempting to incorporate aromatic amines. The fundamental issue lies in the electronic nature of aromatic amines; the direct conjugation between the aryl ring and the amino group drastically diminishes the nucleophilicity of the nitrogen atom. This electronic deactivation impedes the formation of the crucial alpha-hydroxylamine intermediate, which is the precursor to the imine species necessary for the cycloaddition. Consequently, traditional methods often result in poor conversion rates, averaging around 60%, and generate substantial quantities of by-products, necessitating cumbersome purification processes that erode overall process efficiency and increase waste generation.
The Novel Approach
The methodology described in CN113004190B represents a paradigm shift by replacing the traditional copper catalyst with a manganese acetate and DMAP combination. This new approach successfully overcomes the nucleophilicity barrier associated with aromatic amines, allowing for the efficient synthesis of N-phenyl [60] fullerene pyrroline derivatives in a single step. The reaction proceeds smoothly in solvents such as chlorobenzene or o-dichlorobenzene at temperatures between 100°C and 120°C under an air atmosphere, eliminating the need for stringent inert gas protection. This simplification not only enhances operational safety but also significantly reduces the complexity of the reaction setup. Furthermore, the protocol demonstrates broad substrate tolerance, accommodating various alpha-unsubstituted aldehydes and substituted anilines, thereby offering a versatile platform for generating diverse libraries of high-purity OLED material precursors.
Mechanistic Insights into Manganese-Catalyzed Cyclization
The success of this transformation hinges on the unique activation mode provided by the manganese catalyst. Unlike copper systems that may struggle with the specific electronic demands of aromatic amines, the manganese(II) species acts as a potent Lewis acid. It coordinates with the C=C double bonds of the [60] fullerene cage, effectively lowering the LUMO energy level and rendering the fullerene more susceptible to nucleophilic attack. Simultaneously, the presence of DMAP likely facilitates the condensation between the aldehyde and the aromatic amine, promoting the formation of the enamine intermediate despite the reduced nucleophilicity of the amine. This enamine species then undergoes a nucleophilic addition to the activated fullerene double bond, initiating the cascade that leads to ring closure.
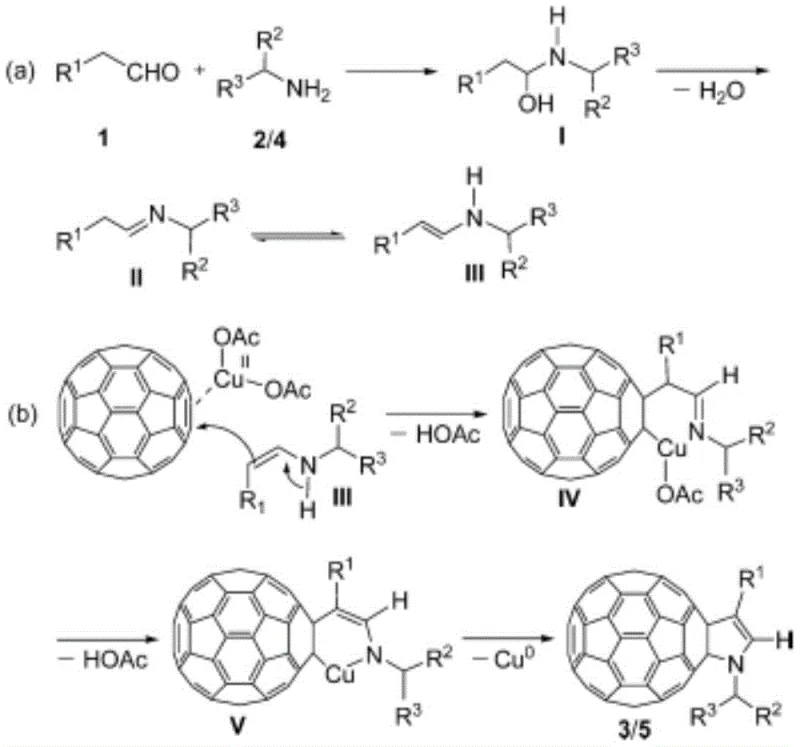
Following the initial addition, the reaction proceeds through an intramolecular cyclization accompanied by the elimination of acetic acid molecules, eventually yielding a manganese-fullerene complex. The final step involves a reductive elimination that releases the desired N-phenyl [60] fullerene pyrroline product and regenerates the catalytic species or precipitates as Cu0 in comparative systems, though here the manganese cycle is optimized for turnover. This mechanistic pathway ensures that side reactions are minimized, leading to a cleaner impurity profile. For quality control teams, this implies a more predictable杂质谱 (impurity profile), which is critical when sourcing materials for sensitive applications like organic photovoltaics where trace metal contaminants or structural isomers can degrade device performance.
How to Synthesize N-phenyl [60] Fullerene Pyrroline Efficiently
The practical execution of this synthesis is designed for reproducibility and ease of handling, making it highly attractive for process chemistry teams aiming for rapid prototyping. The procedure involves dissolving the reactants—[60] fullerene, the chosen aldehyde, and the aromatic amine—in the specified chlorinated solvent, followed by the addition of the catalyst mixture. The reaction progress is conveniently monitored via thin-layer chromatography (TLC), allowing operators to quench the reaction precisely when product formation plateaus and by-product accumulation begins. This level of control is vital for maintaining consistent batch-to-batch quality in a commercial setting.
- Mix [60] fullerene, alpha-unsubstituted aldehyde, and aromatic primary amine in chlorobenzene or o-dichlorobenzene solvent.
- Add a catalytic mixture of manganese acetate and DMAP, then heat the reaction mixture to 100-120°C under air atmosphere.
- Monitor via TLC, filter through silica to remove metal salts, evaporate solvent, and purify the derivative via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this manganese-catalyzed route offers compelling economic and logistical benefits over legacy copper-mediated processes. The primary advantage stems from the drastic simplification of the synthetic sequence; by consolidating the functionalization into a one-step reaction, manufacturers can eliminate multiple unit operations, thereby reducing labor costs and facility occupancy time. Moreover, the use of manganese acetate and DMAP represents a shift towards more cost-effective catalytic additives compared to stoichiometric or high-loading copper reagents, directly contributing to cost reduction in organic photovoltaic material manufacturing. The ability to run the reaction under air further lowers infrastructure costs by removing the need for expensive nitrogen or argon blanketing systems.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of cheaper, earth-abundant manganese catalysts significantly lower the raw material and operational expenditure. The process avoids the need for expensive ligand systems or rigorous exclusion of oxygen, which translates to substantial cost savings in utility consumption and reactor maintenance. Additionally, the high conversion rates reported (up to 92%) mean that less starting material is wasted, optimizing the atom economy of the production line.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various aldehydes and aromatic amines, are commodity chemicals available from multiple global suppliers. This diversity in sourcing mitigates the risk of supply disruptions that often plague specialized reagent markets. The robustness of the reaction conditions also means that production schedules are less likely to be impacted by minor environmental fluctuations, ensuring a steady flow of high-purity N-phenyl [60] fullerene pyrroline to downstream customers.
- Scalability and Environmental Compliance: The straightforward workup procedure, which involves simple filtration to remove metal salts followed by standard chromatography, is inherently easier to scale than processes requiring delicate extractions or crystallizations. This simplicity facilitates the commercial scale-up of complex fullerene derivatives from gram-scale laboratory batches to multi-kilogram production runs. Furthermore, the reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, simplifying waste disposal logistics and reducing compliance overhead.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common inquiries regarding the process specifics and its applicability. These insights are derived directly from the experimental data and mechanistic studies presented in the patent documentation, ensuring accuracy and relevance for industrial application. Understanding these details is crucial for integrating this synthesis route into existing manufacturing workflows.
Q: Why are aromatic amines difficult to use in traditional fullerene pyrroline synthesis?
A: Traditional copper-catalyzed methods often fail with aromatic amines because the direct conjugation between the aryl ring and the amine group significantly reduces the nucleophilicity of the nitrogen atom, hindering the formation of the critical alpha-hydroxylamine intermediate required for cyclization.
Q: What are the key advantages of the manganese acetate/DMAP catalyst system?
A: This novel catalyst system enables a one-step reaction under air conditions with significantly reduced catalyst loading compared to previous methods. It effectively activates the C=C bond of C60 and facilitates the reaction with less nucleophilic aromatic amines, leading to higher conversion rates and simpler product isolation.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process utilizes cheap and easily obtained raw materials and avoids complex multi-step sequences. The simple workup involving filtration and standard column chromatography suggests strong potential for commercial scale-up of complex fullerene derivatives without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-phenyl [60] Fullerene Pyrroline Supplier
The technological advancements outlined in CN113004190B underscore the evolving capabilities in fullerene chemistry, presenting new opportunities for high-performance material development. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of fullerene derivative meets the exacting standards required for advanced electronic and pharmaceutical applications.
We invite you to explore how our expertise can optimize your supply chain for these critical intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and manufacturing goals, ensuring you stay ahead in the competitive landscape of specialty chemicals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
