Advanced Synthetic Route for High-Purity Geranylgeraniol: Commercial Scalability and Cost Efficiency
The global demand for high-value diterpenoids continues to surge, driven by their critical roles in the synthesis of life-saving pharmaceuticals such as Coenzyme Q10, Vitamin K series, and the anticancer agent Paclitaxel. At the heart of this supply chain lies Geranylgeraniol (all-trans-Geranylgeraniol), a pivotal building block whose efficient production has long been a bottleneck for the fine chemical industry. Patent CN1218918C introduces a transformative synthetic methodology that addresses the historical limitations of yield, safety, and cost associated with traditional routes. By leveraging a novel binary oxidant system and optimizing key coupling and reduction steps, this technology offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. For R&D directors and procurement leaders seeking a reliable geranylgeraniol supplier, understanding the technical nuances of this patent is essential for securing a competitive advantage in the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of geranylgeraniol has been plagued by significant technical and economic hurdles that hindered its widespread industrial adoption. Early methodologies, such as those proposed by Klinge Susanne, relied heavily on the use of lithium aluminum hydride, a reagent known for its extreme hazard profile and high cost, rendering the process impractical for large-scale operations. Furthermore, the widely cited route by Kikumasa Sato, while conceptually sound, suffered from severe inefficiencies in critical steps. Specifically, the use of stoichiometric selenium dioxide in the initial oxidation phase resulted in low yields of approximately 44% and generated substantial colloidal waste that complicated downstream processing. Additionally, the reliance on n-butyllithium and hexamethylphosphoramide (HMPA) for the condensation step introduced prohibitive costs and safety risks due to the pyrophoric nature of the reagents and the toxicity of the solvent. Perhaps most critically, the final desulfonation step in conventional methods utilized a lithium-ethylamine system that yielded only 48% of the target product while generating difficult-to-separate double-bond migration isomers, severely impacting the purity profile required for high-purity geranylgeraniol applications.
The Novel Approach
The methodology disclosed in CN1218918C represents a paradigm shift in diterpenoid synthesis by systematically replacing hazardous and inefficient reagents with optimized, cost-effective alternatives. The innovation begins with the substitution of stoichiometric selenium dioxide with a selenium dioxide-tert-butyl hydroperoxide binary oxidant system. This modification not only drastically reduces the consumption of selenium reagents but also simplifies the workup procedure by preventing colloid formation, thereby enhancing the overall yield of the initial oxidation-reduction sequence. In the crucial carbon-carbon bond-forming step, the process replaces the expensive n-butyllithium base with potassium tert-butoxide in dimethylformamide. This change allows the condensation to proceed under much milder conditions (-20°C to 0°C versus -78°C), significantly lowering energy costs and operational complexity. Finally, the implementation of a lithium-methylamine reduction system for the final desulfonation step ensures exceptional selectivity, eliminating the formation of isomeric by-products and boosting the yield of the final step to an impressive 89%, effectively doubling the output compared to legacy methods.
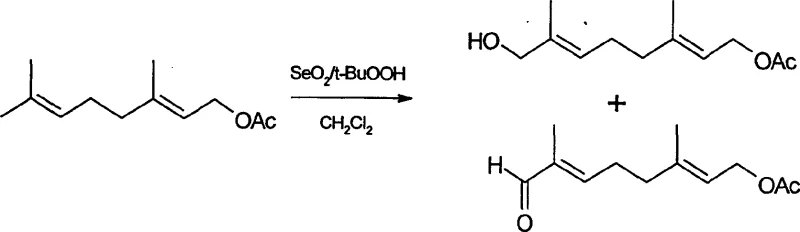
Mechanistic Insights into SeO2/t-BuOOH Binary Oxidation and Reductive Desulfonation
The core chemical innovation of this process lies in the mechanistic efficiency of the binary oxidant system employed in the first step. Traditional allylic oxidation using selenium dioxide often requires stoichiometric amounts of the reagent, leading to the generation of elemental selenium sludge that is difficult to filter and dispose of. In this novel approach, tert-butyl hydroperoxide acts as a co-oxidant, regenerating the active selenium species in situ or facilitating the oxidation cycle with a significantly lower loading of selenium dioxide. The molar ratio of selenium dioxide to tert-butyl hydroperoxide is optimized between 1:4 and 1:10, ensuring that the oxidation of the (E)-methyl group of geranyl acetate proceeds selectively to form a mixture of allylic carbonyl and hydroxyl products. This mixture is then seamlessly converted to the single desired alcohol intermediate, trans-8-hydroxyacetic acid geranyl ester, via sodium borohydride reduction. This tandem oxidation-reduction strategy maximizes atom economy and minimizes waste, a critical factor for cost reduction in pharmaceutical intermediates manufacturing.
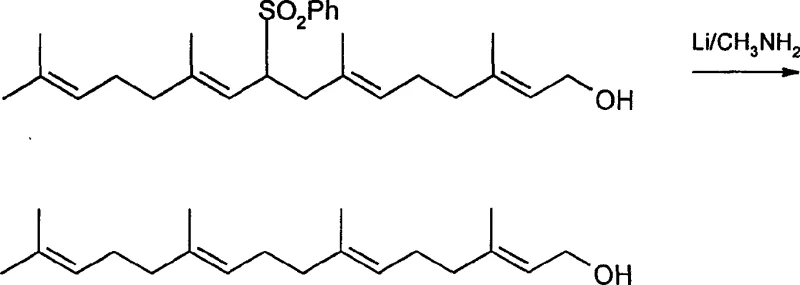
Furthermore, the mechanism of the final desulfonation step has been refined to address the persistent issue of stereochemical integrity. In previous methods using lithium-ethylamine, the higher reactivity of the reducing system often led to the migration of double bonds, creating cis-isomers that are structurally similar to the all-trans target and notoriously difficult to separate via standard chromatography. The use of lithium in methylamine provides a milder reducing environment with lower reactivity but higher selectivity. This specific solvent-reductant combination facilitates the cleavage of the sulfone group via a radical anion mechanism without disturbing the conjugated polyene system of the geranylgeranyl chain. As a result, the process delivers the target all-trans-geranylgeraniol with superior purity, eliminating the need for costly and yield-loss-inducing purification steps to remove geometric isomers.
How to Synthesize Geranylgeraniol Efficiently
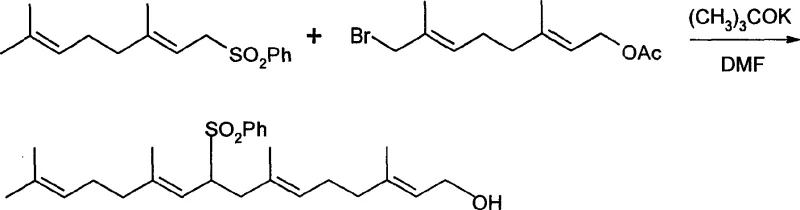
The synthesis of geranylgeraniol via this patented route involves a logical four-step sequence that transforms readily available geranyl acetate into the high-value diterpenol. The process is designed to be operationally simple, avoiding the need for cryogenic temperatures in the coupling step and utilizing common laboratory solvents. The initial phase focuses on the functionalization of the geranyl backbone through selective oxidation, followed by activation of the alcohol as a bromide leaving group. The subsequent coupling reaction builds the C20 carbon skeleton, and the final reductive step unveils the target molecule. For detailed laboratory protocols and specific reaction parameters, please refer to the standardized synthesis guide below.
- Selective allylic oxidation of geranyl acetate using a SeO2/t-BuOOH binary system followed by NaBH4 reduction.
- Conversion of the hydroxy intermediate to the corresponding bromide using PBr3 in anhydrous ether.
- Base-mediated condensation of geranyl sulfone with the bromo-intermediate using potassium tert-butoxide.
- Final reductive removal of the sulfone group using lithium in methylamine to yield all-trans-geranylgeraniol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend far beyond simple chemical curiosity. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on specialized and hazardous reagents like n-butyllithium and HMPA, manufacturers can source inputs from a broader range of chemical suppliers, reducing the risk of supply disruptions. The replacement of these high-cost reagents with commodity chemicals like potassium tert-butoxide and methylamine directly translates to a lower bill of materials. Moreover, the significant improvement in yield, particularly in the final desulfonation step where efficiency nearly doubles from 48% to 89%, means that less starting material is required to produce the same amount of finished goods. This increase in material throughput effectively lowers the unit cost of production without requiring capital investment in new equipment, providing a clear path for cost reduction in pharmaceutical intermediates manufacturing.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as n-butyllithium and hexamethylphosphoramide (HMPA) fundamentally alters the cost structure of the synthesis. Potassium tert-butoxide is a widely available, solid base that is significantly cheaper and safer to handle than pyrophoric organolithium reagents. Additionally, the binary oxidant system reduces the consumption of selenium dioxide, a relatively costly heavy metal reagent, by allowing it to function more efficiently with tert-butyl hydroperoxide. The cumulative effect of these substitutions is a substantial decrease in raw material expenses, while the higher yields across all steps ensure that maximum value is extracted from every kilogram of input material.
- Enhanced Supply Chain Reliability: Reliance on exotic or highly regulated reagents often creates bottlenecks in the supply chain, leading to extended lead times and potential production stoppages. This novel method utilizes reagents that are staples of the fine chemical industry, such as geranyl acetate, sodium borohydride, and phosphorus tribromide, all of which enjoy stable and robust global supply networks. The shift away from cryogenic conditions for the coupling step (from -78°C to -20°C/0°C) also reduces the strain on facility infrastructure, allowing for more flexible scheduling and faster turnaround times. This reliability is crucial for maintaining continuous production schedules for downstream clients requiring high-purity geranylgeraniol for time-sensitive drug development programs.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is inherently more scalable. The reduction in selenium waste and the avoidance of carcinogenic solvents like HMPA simplify waste treatment protocols and reduce the regulatory burden on manufacturing facilities. The higher selectivity of the lithium-methylamine reduction minimizes the formation of isomeric impurities, which reduces the solvent and energy load associated with extensive purification processes like preparative HPLC. These factors combined make the transition from pilot scale to multi-ton commercial production smoother and more compliant with increasingly stringent environmental regulations, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route. These insights are derived directly from the comparative data and experimental results presented in the patent literature, highlighting the specific improvements over prior art methods. Understanding these details is vital for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing the quality and cost implications of sourcing intermediates produced via this technology.
Q: What are the key advantages of the SeO2/t-BuOOH binary oxidant system?
A: This system significantly reduces the consumption of selenium dioxide compared to stoichiometric methods, minimizes colloidal waste formation, and improves the yield of the initial oxidation step from roughly 44% to over 60%.
Q: How does this method improve upon the traditional Kikumasa Sato route?
A: It replaces hazardous and expensive reagents like n-butyllithium and HMPA with safer alternatives like potassium tert-butoxide. Furthermore, it utilizes a lithium-methylamine reduction system that prevents double-bond migration isomers, boosting the final step yield from 48% to 89%.
Q: Is this process suitable for large-scale manufacturing of Coenzyme Q10 precursors?
A: Yes, the use of mild reaction conditions (0°C to -20°C for coupling) and commercially available starting materials like geranyl acetate makes this route highly scalable and cost-effective for industrial production of diterpenoid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Geranylgeraniol Supplier
As the pharmaceutical and fine chemical industries continue to evolve, the need for efficient, scalable, and cost-effective synthetic routes for complex intermediates like geranylgeraniol has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced process technologies such as the one described in CN1218918C to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of geranylgeraniol meets the exacting standards required for the synthesis of Coenzyme Q10, Vitamin K, and other high-value bioactive compounds.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how switching to our optimized process can impact your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates that drive your innovation forward.
