Advanced Water-Soluble Diamine Ligands for Green Asymmetric Catalysis and Commercial Scale-Up
Introduction to Novel Water-Soluble Chiral Ligands
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for greener, more sustainable manufacturing processes. Patent CN1566084A introduces a groundbreaking class of optically pure water-soluble diamines and their derivatives, specifically designed to facilitate efficient asymmetric catalysis in aqueous media. This technology addresses a critical bottleneck in the synthesis of chiral pharmaceutical intermediates by replacing hazardous organic solvents with water, thereby enhancing safety and environmental compliance. The core innovation lies in the structural modification of 1,2-diphenyl ethylene diamine through strategic sulfonation, rendering the ligand soluble in water while maintaining its stereochemical integrity. 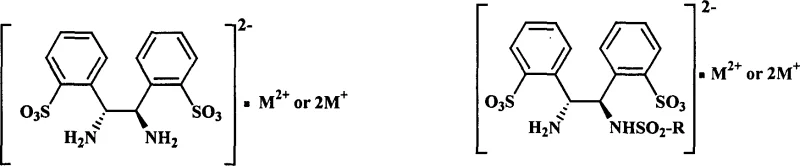 This structural adaptation allows the resulting metal complexes, particularly those with Ruthenium, Rhodium, and Iridium, to operate effectively in water-organic biphasic systems. For R&D directors and process chemists, this represents a paradigm shift towards more robust and scalable catalytic systems that do not compromise on enantioselectivity, achieving values up to 98% ee in specific transformations.
This structural adaptation allows the resulting metal complexes, particularly those with Ruthenium, Rhodium, and Iridium, to operate effectively in water-organic biphasic systems. For R&D directors and process chemists, this represents a paradigm shift towards more robust and scalable catalytic systems that do not compromise on enantioselectivity, achieving values up to 98% ee in specific transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral alcohols and amines, which are vital building blocks for beta-adrenoceptor antagonists and other active pharmaceutical ingredients, has relied heavily on stoichiometric chiral reagents or homogeneous catalysis in organic solvents. A prominent example is the use of chiral oxazaborolidine reagents (such as CBS catalysts) for the reduction of ketones. While effective, these methods suffer from significant drawbacks including the high cost of stoichiometric reagents, the generation of substantial chemical waste, and the difficulty in separating the product from the reaction mixture. Furthermore, the reliance on volatile organic compounds (VOCs) poses serious safety hazards and environmental liabilities, complicating regulatory compliance and increasing operational expenditures related to solvent recovery and waste treatment. The inability to easily recycle expensive transition metal catalysts in homogeneous organic systems further exacerbates the cost burden, making these processes less attractive for large-scale industrial applications where margin compression is a constant concern.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by leveraging the unique properties of water-soluble ligands. By introducing sulfonic acid groups onto the chiral diamine backbone, the ligand becomes inherently compatible with aqueous environments. This enables the formation of stable metal complexes that can catalyze asymmetric transfer hydrogenation reactions directly in water or water-organic biphasic systems. The use of water as a solvent not only eliminates the fire hazards associated with organic solvents but also simplifies the work-up procedure; products can often be extracted with immiscible organic solvents while the catalyst remains in the aqueous phase. 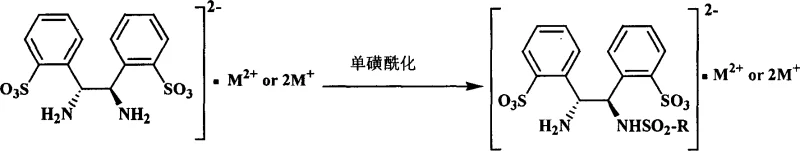 Moreover, the addition of surfactants or phase-transfer catalysts, such as cationic surfactants, dramatically enhances reaction rates and enantioselectivity, mimicking enzymatic environments. This methodology offers a cleaner, safer, and potentially more cost-effective route to high-value chiral intermediates, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Moreover, the addition of surfactants or phase-transfer catalysts, such as cationic surfactants, dramatically enhances reaction rates and enantioselectivity, mimicking enzymatic environments. This methodology offers a cleaner, safer, and potentially more cost-effective route to high-value chiral intermediates, aligning perfectly with the principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into Aqueous Asymmetric Transfer Hydrogenation
The mechanistic foundation of this technology rests on the precise engineering of the ligand environment around the transition metal center. The synthesis begins with the direct sulfonation of optical purity 1,2-diphenyl ethylene diamine using oleum, followed by a selective mono-sulfonylation step. This dual functionalization ensures that the ligand possesses both the necessary chirality for stereocontrol and the hydrophilicity for water solubility. When complexed with Group 8 transition metals like Ruthenium, the resulting catalyst operates via a concerted outer-sphere mechanism typical of Noyori-type transfer hydrogenation. In this mechanism, a metal-hydride species is generated in situ using a hydrogen donor such as sodium formate or isopropanol. The hydride is then transferred to the prochiral ketone substrate in a highly stereoselective manner, dictated by the chiral pocket formed by the diamine ligand. The presence of the sulfonate groups does not hinder this coordination; rather, it facilitates the interaction between the catalyst and the substrate within the aqueous micellar environment created by surfactants.
Impurity control is another critical aspect where this mechanism excels. The biphasic nature of the reaction system acts as a built-in purification step. Since the catalyst is water-soluble and the product is typically organic-soluble, simple phase separation effectively removes the bulk of the metal catalyst from the product stream. This minimizes the risk of metal contamination in the final API, a stringent requirement for pharmaceutical manufacturing. Additionally, the mild reaction conditions (typically 0°C to 45°C) prevent thermal degradation of sensitive functional groups, ensuring a cleaner impurity profile compared to harsher reduction methods. The ability to tune the reaction outcome, such as selectively obtaining chiral epoxides versus chiral alcohols from omega-bromo ketones by adjusting temperature, further demonstrates the precise control afforded by this catalytic system, providing R&D teams with a versatile tool for complex molecule synthesis.
How to Synthesize Optical Pure Water-Soluble Diamines Efficiently
The preparation of these advanced ligands involves a streamlined two-step sequence that is amenable to scale-up. The process starts with the sulfonation of the parent diamine, followed by selective protection of one amine group via sulfonylation. This protocol ensures high optical purity is maintained throughout the synthesis. The detailed standardized synthesis steps, including specific molar ratios, temperature controls, and purification techniques like recrystallization and column chromatography, are outlined below to guide process implementation.
- Sulfonate optical purity 1,2-diphenyl ethylene diamine with oleum at 0-35°C to obtain the 2,2'-disulfonic acid substituted derivative.
- React the disulfonic derivative with sulfonyl chloride under alkaline conditions with surfactants to achieve selective mono-sulfonylation.
- Complex the resulting ligand with Ruthenium, Rhodium, or Iridium precursors for use in aqueous asymmetric transfer hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous catalytic technology presents compelling economic and operational advantages. The shift from organic solvents to water drastically reduces raw material costs, as water is significantly cheaper and more readily available than high-purity organic solvents like dichloromethane or THF. Furthermore, the elimination of large volumes of VOCs simplifies waste management protocols, leading to substantial cost savings in disposal and environmental compliance fees. The ability to recycle the catalyst multiple times without significant loss of activity means that the consumption of expensive precious metals like Ruthenium and Rhodium is minimized, directly impacting the cost of goods sold (COGS). This efficiency translates into a more competitive pricing structure for the final chiral intermediates.
- Cost Reduction in Manufacturing: The transition to an aqueous-based system fundamentally alters the cost structure of chiral synthesis. By eliminating the need for expensive stoichiometric chiral reagents and reducing the dependency on volatile organic solvents, the overall material costs are significantly lowered. The catalyst recycling capability, demonstrated to maintain efficiency over multiple cycles, ensures that the high initial cost of the transition metal precursor is amortized over a much larger production volume. This leads to a drastic reduction in the cost per kilogram of the chiral product, enhancing profit margins for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium mitigates risks associated with the supply and price volatility of organic solvents. Water is universally available and不受 geopolitical supply chain disruptions that often affect specialty chemicals. Additionally, the simplified work-up procedure, which often requires only phase separation and extraction, reduces the processing time and equipment complexity. This streamlined workflow enhances throughput and reliability, ensuring consistent delivery schedules for critical pharmaceutical intermediates even during periods of high demand.
- Scalability and Environmental Compliance: The inherent safety of water-based reactions makes this technology highly scalable. The absence of flammable organic solvents reduces the risk of fire and explosion, allowing for larger batch sizes and more aggressive production schedules without compromising safety standards. From an environmental perspective, the reduced generation of hazardous waste and the lower carbon footprint associated with solvent production and disposal align with global sustainability goals. This positions manufacturers using this technology favorably regarding regulatory compliance and corporate social responsibility initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this water-soluble diamine technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of using water-soluble diamine ligands over traditional organic soluble ligands?
A: Water-soluble ligands enable reactions in aqueous media, significantly reducing the need for volatile organic solvents. This simplifies product isolation through phase separation, allows for catalyst recycling, and aligns with green chemistry principles by lowering environmental impact and waste disposal costs.
Q: Can the catalyst system described in CN1566084A be recycled?
A: Yes, the patent demonstrates that the catalyst retains high transformation efficiency and enantioselectivity after multiple cycles. Specifically, the aqueous catalyst phase can be reused up to six times without significant loss in performance, offering substantial economic benefits for large-scale production.
Q: What types of substrates are compatible with this asymmetric transfer hydrogenation method?
A: The method is highly versatile, effectively reducing various aromatic alkyl ketones including acetophenone derivatives, p-nitroacetophenone, and omega-bromo aromatic alkyl ketones. It can selectively produce chiral alcohols or epoxides depending on temperature control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Disulfonic Substituted 1,2-Diphenyl Ethylene Diamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the water-soluble diamine technology described in CN1566084A for the future of green pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition to this advanced catalytic system is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chiral intermediate delivered meets the highest industry standards for enantiomeric excess and chemical purity.
We invite you to collaborate with our technical team to explore how this innovative aqueous catalysis route can optimize your specific synthesis challenges. By leveraging our expertise, you can achieve significant process improvements and cost efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments tailored to your project requirements. Let us help you build a more sustainable and profitable supply chain for your critical chiral building blocks.
