Advanced Catalytic Synthesis of N(2)9-Diacetylguanine for Scalable Acyclovir Intermediate Production
Introduction to Patented Acyclovir Intermediate Technology
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical antiviral intermediates, and the recent disclosure in patent CN111777611A offers a transformative approach to synthesizing N(2),9-diacetylguanine. This compound serves as a pivotal precursor in the production of acyclovir, a widely prescribed antiviral medication used to treat herpes simplex and varicella-zoster virus infections. The traditional reliance on guanine or guanosine starting materials has long presented challenges regarding purity consistency and environmental impact, prompting the need for innovative chemical engineering solutions. This new methodology leverages a pyrimidine-based starting material to construct the purine ring system more efficiently, bypassing the limitations inherent in older extraction or semi-synthetic routes. By integrating selective acylation and optimized cyclization steps, the process ensures a high degree of molecular control that translates directly to improved product quality and operational safety for manufacturers. The strategic shift away from heavy metal catalysts and high-risk hydrogenation steps marks a significant advancement in green chemistry principles applied to pharmaceutical intermediate production. Stakeholders in the fine chemical sector must recognize this technological pivot as a key driver for future supply chain resilience and cost efficiency in antiviral drug manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of N(2),9-diacetylguanine has relied heavily on the direct acetylation of guanine or guanosine, a practice that has persisted for decades despite its inherent inefficiencies. As illustrated in the traditional reaction pathways, these methods often struggle to achieve purity levels exceeding 98%, resulting in products with suboptimal appearance that require extensive and costly purification procedures. The reliance on guanine as a starting material introduces significant variability, as the upstream synthesis of guanine itself involves high-energy consumption and the generation of substantial inorganic salt waste. Furthermore, conventional routes frequently necessitate the use of large excesses of acetic anhydride, which complicates solvent recovery and leads to the accumulation of acidic waste liquids that are difficult to treat. The presence of heavy metal catalysts in upstream guanine production, such as palladium or nickel used in hydrogenation steps, adds another layer of complexity regarding safety protocols and residual metal removal in the final API. These cumulative factors create a manufacturing bottleneck that increases both the environmental footprint and the overall cost of goods sold for pharmaceutical companies relying on these legacy supply chains.
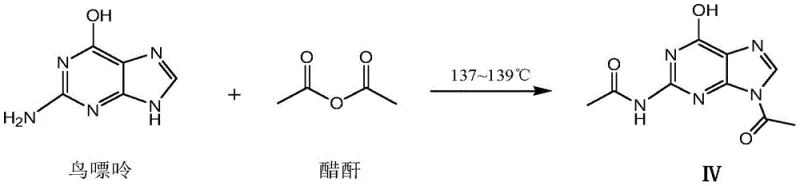
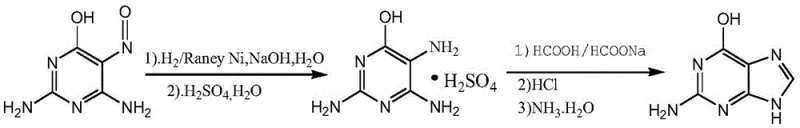
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes 2,4-diamino-6-hydroxy-5-carboxamidopyrimidine as a foundational building block, effectively circumventing the purity and safety issues associated with guanine. This strategic selection of starting material allows for a stepwise construction of the molecule where selectivity is rigorously controlled through precise temperature management and solvent engineering. The process eliminates the need for high-pressure hydrogenation equipment, thereby reducing capital expenditure and operational risk for manufacturing facilities. By avoiding the introduction of large amounts of water and inorganic salts, the new method simplifies the downstream workup and significantly reduces the volume of wastewater requiring treatment. The ability to recycle solvents such as acetic acid and xylene further enhances the economic viability of the process, creating a closed-loop system that minimizes raw material consumption. This paradigm shift not only addresses the technical limitations of yield and purity but also aligns with modern regulatory expectations for sustainable and environmentally responsible chemical manufacturing practices in the pharmaceutical sector.
Mechanistic Insights into Selective Acylation and Cyclization
The core innovation of this synthesis lies in the meticulous control of reaction kinetics during the initial selective acylation step, where 2,4-diamino-6-hydroxy-5-carboxamidopyrimidine is converted into 2-acetamido-4-amino-6-hydroxy-5-carboxamidopyrimidine. By dissolving the原料 in acetic acid and dropwise adding a diluted acetic anhydride solution, the reaction system maintains a low local concentration of the acylating agent, which is critical for preventing over-acetylation and di-substitution byproducts. This precise control ensures that the acetylation occurs selectively at the desired amino group, setting the stage for a clean cyclization reaction in the subsequent step. The use of specific catalysts, such as sodium acetate or tetrabutylammonium bromide, further facilitates the ring-closing reaction by promoting dehydration under reflux conditions without degrading the sensitive purine structure. The mechanistic pathway is designed to maximize the formation of the 2-acetamido-6-hydroxypurine intermediate, which serves as the direct precursor to the final target molecule. This level of mechanistic understanding allows process chemists to fine-tune reaction parameters such as temperature and addition rates to optimize yield and minimize the formation of difficult-to-remove impurities.
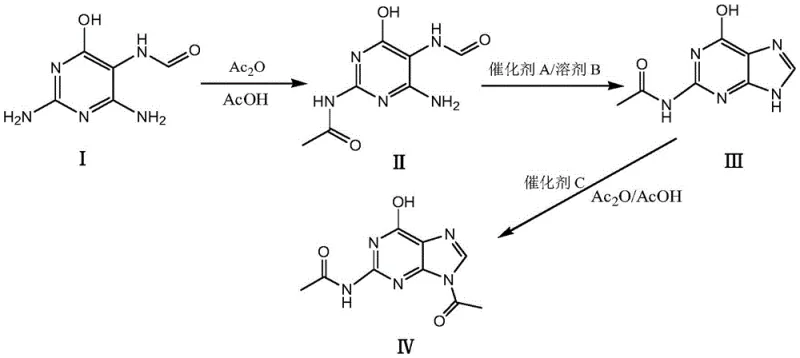
Impurity control is inherently built into the design of this catalytic cycle, particularly through the management of solvent systems and catalyst loading in the final acylation step. The transition from the purine intermediate to N(2),9-diacetylguanine is achieved using a mixed solvent system of acetic anhydride and acetic acid, which provides the necessary solubility and reactivity balance for high conversion rates. The selection of Catalyst C, often a mild organic base like triethylamine, ensures that the acylation proceeds efficiently without promoting side reactions that could compromise the structural integrity of the molecule. By avoiding the harsh conditions associated with traditional methods, the process maintains a narrow impurity profile that is easier to manage during crystallization and filtration. The resulting product consistently demonstrates high purity and excellent physical appearance, meeting stringent quality standards required for pharmaceutical intermediates. This robust control over the chemical environment ensures that the process is not only scientifically sound but also practically viable for consistent commercial production.
How to Synthesize N(2)9-Diacetylguanine Efficiently
The implementation of this synthesis route requires a clear understanding of the operational parameters defined in the patent to ensure optimal results in a production setting. The process is divided into three distinct stages, each with specific requirements for temperature, solvent ratios, and catalyst concentrations that must be adhered to for successful scale-up. Operators must focus on the precise dropwise addition of reagents in the first step to maintain selectivity, followed by careful temperature control during the reflux cyclization to drive water removal effectively. The final acylation step demands attention to solvent composition to ensure complete conversion while facilitating easy product isolation through crystallization. Detailed standard operating procedures derived from the patent data provide the necessary framework for translating this laboratory-scale innovation into a reliable industrial process.
- Dissolve 2,4-diamino-6-hydroxy-5-carboxamidopyrimidine in acetic acid and perform selective acylation with acetic anhydride solution to obtain the mono-acetylated intermediate.
- Conduct a cyclization reaction using Catalyst A in a refluxing organic solvent to form the purine ring structure of 2-acetamido-6-hydroxypurine.
- Perform the final acylation on the purine intermediate using Catalyst C in a mixed acetic anhydride and acetic acid solvent to yield high-purity N(2)9-diacetylguanine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial advantages by fundamentally altering the cost structure and risk profile of intermediate manufacturing. The elimination of expensive noble metal catalysts and the reduction in hazardous waste generation directly translate to lower operational expenditures and simplified compliance management. By enabling the recycling of key solvents like acetic acid and xylene, the process significantly reduces the consumption of raw materials, leading to a more sustainable and cost-effective supply chain. The improved safety profile, characterized by the absence of high-pressure hydrogenation, lowers insurance costs and reduces the need for specialized safety infrastructure, making it an attractive option for manufacturing partners. These efficiencies collectively contribute to a more stable supply of high-quality intermediates, mitigating the risk of production delays caused by regulatory or environmental constraints. Companies adopting this technology can expect a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The process achieves cost optimization through the elimination of costly heavy metal catalysts and the implementation of solvent recovery systems that minimize raw material waste. By avoiding the generation of large volumes of inorganic salts and acidic wastewater, the expense associated with waste treatment and disposal is drastically reduced. The high yield and purity of the product also mean less material is lost to purification steps, further enhancing the overall economic efficiency of the manufacturing process. These factors combine to create a leaner production model that offers significant competitive advantages in price-sensitive markets.
- Enhanced Supply Chain Reliability: The simplified reaction conditions and reduced safety risks contribute to a more reliable supply chain by minimizing the potential for unplanned shutdowns or regulatory interventions. The use of readily available starting materials and common solvents ensures that procurement teams can source inputs easily without facing supply bottlenecks. The robustness of the process allows for consistent production schedules, ensuring that downstream API manufacturers receive their intermediates on time and to specification. This reliability is crucial for maintaining the continuity of drug production and meeting patient needs without interruption.
- Scalability and Environmental Compliance: The design of this synthesis route inherently supports scalability, as it avoids complex unit operations that are difficult to replicate on a large scale. The reduced environmental footprint, characterized by lower waste generation and solvent recycling, ensures that the process remains compliant with increasingly stringent environmental regulations. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity. Facilities can scale production with confidence, knowing that the process is both economically and environmentally sustainable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis method for N(2),9-diacetylguanine. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to provide accurate guidance for stakeholders. Understanding these details is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks. The insights provided here aim to clarify the operational advantages and quality improvements associated with this novel approach.
Q: How does this new method improve product purity compared to traditional guanine-based routes?
A: The patented process avoids the use of crude guanine starting materials which often contain hard-to-remove impurities. By building the purine ring from a pyrimidine precursor, the method achieves purity levels exceeding 98% with superior appearance, eliminating the need for complex decolorization steps associated with traditional hydrogenation methods.
Q: What are the environmental benefits of this synthesis route for large-scale manufacturing?
A: This process significantly reduces the generation of inorganic salts and acidic wastewater. Unlike traditional methods that require large excesses of acetic anhydride and produce difficult-to-recycle waste, this route allows for the recovery and reuse of acetic acid solvents, resulting in a much lower environmental footprint and reduced waste disposal costs.
Q: Is this process suitable for commercial scale-up without high-risk safety measures?
A: Yes, the method eliminates the need for high-pressure catalytic hydrogenation and hazardous noble metal catalysts like palladium or platinum. The reaction conditions operate at atmospheric pressure with manageable temperatures, significantly lowering safety risks and simplifying the engineering requirements for commercial scale-up in standard chemical reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N(2)9-Diacetylguanine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of implementing advanced synthetic methodologies like the one described in patent CN111777611A to deliver superior pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this green chemistry route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of N(2),9-diacetylguanine meets the highest industry standards for antiviral drug synthesis. Our commitment to technical excellence allows us to navigate the complexities of catalytic acylation and cyclization with precision, delivering a product that supports the efficient manufacturing of life-saving medications. Partnering with us means gaining access to a supply chain that is both innovative and reliable, driven by a deep understanding of chemical process engineering.
We invite procurement leaders and technical directors to engage with our Customized Cost-Saving Analysis to explore how this optimized synthesis route can benefit your specific production needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate the tangible value of our manufacturing capabilities. By collaborating with NINGBO INNO PHARMCHEM, you secure a supply of high-purity intermediates that are produced with a focus on sustainability and cost-efficiency. Contact us today to discuss how we can support your supply chain optimization goals with our advanced chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
