Revolutionizing Tetrahydrozoline Production: A Deep Dive into Scalable API Intermediate Manufacturing
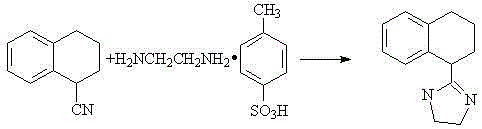
The pharmaceutical industry is constantly seeking more efficient pathways for synthesizing critical active pharmaceutical ingredient (API) intermediates, and the technology disclosed in patent CN103224468A represents a significant leap forward in the manufacturing of Tetrahydrozoline. This specific patent outlines a groundbreaking synthetic method that transitions away from the cumbersome, multi-step processes historically associated with this compound, opting instead for a streamlined, high-yield approach. By leveraging a direct condensation and cyclization strategy between 1-cyanotetralin and ethylenediamine monotosylate, the inventors have successfully addressed long-standing issues regarding yield optimization and process complexity. For R&D directors and procurement managers alike, this development signals a potential paradigm shift in how we approach the supply chain for vasoconstrictor intermediates, offering a route that is not only chemically elegant but also commercially viable for large-scale operations. The elimination of intermediate isolation steps and the ability to control reaction progress simply through thermal parameters make this a highly attractive candidate for modern green chemistry initiatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tetrahydrozoline has been plagued by inefficient methodologies that rely on the condensation of indane carboxylic acid derivatives with tetraols under harsh conditions. These traditional routes typically necessitate the use of organic solvents and specific catalysts to drive the initial condensation, followed by a separate ring-closure reaction and a final salification step with hydrochloric acid. This multi-stage process is inherently fraught with operational difficulties, including severe reaction conditions that demand rigorous safety controls and energy inputs. Furthermore, the requirement to isolate and purify intermediates between each step drastically increases the overall production time and generates substantial amounts of chemical waste, leading to higher disposal costs and environmental burdens. The cumulative effect of these inefficiencies is a lower total yield and a higher cost of goods sold (COGS), making the conventional method less competitive in a market that increasingly demands cost-effective and sustainable manufacturing solutions.
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a direct reaction between 1-cyanotetralin and ethylenediamine monotosylate, effectively bypassing the need for pre-functionalized carboxylic acid precursors. This innovative strategy employs a 'one-pot' synthesis technique where the reactants are heated together in a controlled mass ratio, allowing the cyclization to occur directly without the intervention of external solvents or complex catalytic systems. The simplicity of this method is its greatest strength; by merging the condensation and cyclization into a single thermal event, the process significantly reduces the number of unit operations required. This consolidation not only accelerates the production timeline but also minimizes the potential for material loss during transfer and purification stages. The result is a robust manufacturing protocol that delivers high-purity Tetrahydrozoline with exceptional efficiency, setting a new benchmark for what is achievable in intermediate synthesis.
Mechanistic Insights into Thermal Cyclization of Nitriles
The core of this synthetic breakthrough lies in the thermal activation of the nitrile group within 1-cyanotetralin, which serves as the electrophilic center for the subsequent nucleophilic attack by the amine functionality of ethylenediamine monotosylate. Under the elevated temperatures specified in the patent, ranging from 120°C to 210°C, the kinetic energy of the molecules increases sufficiently to overcome the activation barrier for the addition reaction. The monotosylate salt form of ethylenediamine plays a crucial role here, likely acting as a stabilized source of the diamine that prevents premature polymerization or side reactions while ensuring a steady release of the nucleophile. As the reaction proceeds, the initial adduct undergoes an intramolecular cyclization, driven by the proximity of the second amine group to the imine intermediate formed after the initial attack. This cascade of events leads to the formation of the imidazoline ring system characteristic of Tetrahydrozoline, all occurring within the molten reaction mass without the dilution effects of a solvent.
Controlling impurities in this solvent-free environment is achieved through precise modulation of reaction time and temperature, which are the two critical variables governing the extent of conversion and the formation of by-products. If the temperature is too low or the time too short, unreacted starting materials may remain, whereas excessive heat or prolonged reaction times could lead to degradation products or oligomerization. The patent data indicates that optimizing these parameters allows for a clean reaction profile, where the crude solid obtained upon cooling is already of high quality. Subsequent purification steps, such as recrystallization or pH-controlled extraction, are then highly effective at removing trace impurities, ensuring that the final product meets the stringent purity specifications required for pharmaceutical applications. This level of control demonstrates a deep understanding of the reaction kinetics and thermodynamics involved in nitrile-amine cyclizations.
How to Synthesize Tetrahydrozoline Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and thermal profile to maximize yield and purity. The process begins with the precise weighing of 1-cyanotetralin and ethylenediamine monotosylate, adhering to the recommended mass ratio to ensure complete consumption of the limiting reagent. Once charged into the reaction vessel, the mixture is subjected to a controlled heating ramp, maintaining the target temperature for a duration sufficient to drive the reaction to completion as monitored by analytical methods. Following the reaction period, the mixture is cooled to induce crystallization of the product, which is then isolated via filtration and washed to remove soluble impurities. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the standardized synthesis steps outlined below.
- Charge 1-cyanotetralin and ethylenediamine monotosylate into a reaction vessel at a mass ratio between 2: 1 and 5:1.
- Heat the mixture to a temperature range of 120-210°C and maintain stirring for 1 to 5 hours to facilitate the cyclization reaction.
- Cool the reaction mixture to induce solidification, filter the crude product, wash with cold water, and purify via recrystallization or pH-controlled extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis method offers profound benefits for procurement managers and supply chain heads who are tasked with optimizing costs and ensuring reliable material flow. The elimination of organic solvents and expensive catalysts from the process equation translates directly into a reduction in raw material expenditures and a simplification of the supply chain logistics. Without the need to procure, store, and dispose of large volumes of volatile solvents, manufacturers can significantly lower their operational overheads and reduce their environmental footprint. Furthermore, the 'one-pot' nature of the reaction minimizes the requirement for complex reactor setups and intermediate storage tanks, allowing for a more compact and efficient production facility layout. These factors combined create a compelling economic case for switching to this technology, particularly for companies looking to enhance their margins in a competitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The most immediate financial impact of this technology is the drastic simplification of the production workflow, which inherently lowers labor and utility costs. By removing the need for solvent recovery systems and catalyst removal steps, the process reduces the energy intensity of the manufacturing cycle. Additionally, the high yield reported in the patent means that less raw material is wasted per kilogram of finished product, further driving down the variable cost of production. This efficiency gain allows suppliers to offer more competitive pricing structures to their downstream clients without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 1-cyanotetralin and ethylenediamine derivatives ensures a stable and resilient supply chain that is less susceptible to disruptions. Unlike specialized catalysts or exotic reagents that may have long lead times or single-source dependencies, the inputs for this process are commodity chemicals with robust global availability. This accessibility mitigates the risk of production stoppages due to raw material shortages, providing procurement teams with greater confidence in their inventory planning and delivery commitments to customers.
- Scalability and Environmental Compliance: Scaling this process from pilot batches to commercial tonnage is straightforward due to the absence of solvent handling hazards and the simplicity of the workup procedure. The solid-state nature of the reaction and the ease of filtration make it ideally suited for large-scale reactors, facilitating rapid capacity expansion to meet market demand. Moreover, the reduced generation of hazardous waste aligns with increasingly strict environmental regulations, helping manufacturers avoid costly compliance issues and fostering a more sustainable corporate image.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Tetrahydrozoline synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of adopting this technology within your existing manufacturing infrastructure.
Q: What are the primary advantages of the new Tetrahydrozoline synthesis method over traditional routes?
A: The novel method described in patent CN103224468A eliminates the need for organic solvents and complex catalysts required in traditional indane carboxylic acid routes. It utilizes a direct 'one-pot' melt reaction between 1-cyanotetralin and ethylenediamine monotosylate, significantly reducing processing steps, waste generation, and overall production time while achieving yields up to 80.3%.
Q: How is purity controlled in this solvent-free synthesis process?
A: Purity is rigorously managed through precise temperature control (120-210°C) and reaction time optimization (1-5h) to minimize side reactions. Post-reaction, the crude solid undergoes purification via either recrystallization or pH-value extraction, consistently delivering product purity levels exceeding 98%, which is critical for pharmaceutical grade intermediates.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for scalability. By avoiding volatile organic solvents and utilizing a straightforward filtration and washing workup, the method simplifies equipment requirements and safety protocols. The robust nature of the reaction conditions allows for easy transition from laboratory bench scales to multi-ton commercial production facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydrozoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in CN103224468A for enhancing the efficiency of pharmaceutical intermediate production. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and high-quality supply of critical materials. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards, guaranteeing that the Tetrahydrozoline we supply meets the exacting requirements of global regulatory bodies.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized synthesis route for your operations. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and reliability in your supply chain.
