Advanced Synthesis of Cholestenyl-TTF Liquid Crystals for Next-Generation Electronic Materials
Advanced Synthesis of Cholestenyl-TTF Liquid Crystals for Next-Generation Electronic Materials
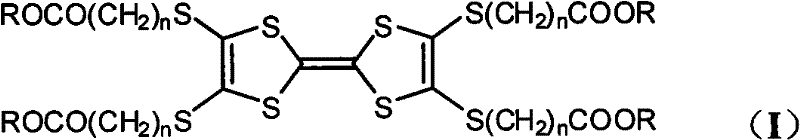
The rapid evolution of organic electronics demands materials that combine robust thermal stability with precise molecular ordering, a challenge effectively addressed by the innovations detailed in patent CN102127139A. This pivotal intellectual property introduces a sophisticated class of liquid crystal compounds based on the tetrathiafulvalene (TTF) structure, specifically functionalized with cholestenyl groups to enhance mesomorphic behavior. The core breakthrough lies in the ability to synthesize these complex architectures through a streamlined three-step process that ensures high purity and reproducible phase transitions, critical for consistent performance in display and semiconductor technologies. By integrating rigid TTF cores with flexible chiral cholesterol derivatives, the resulting materials exhibit unique smectic A and ordered hexagonal columnar phases that are essential for advanced optoelectronic applications. This report analyzes the technical merits of this synthesis route, offering strategic insights for R&D directors seeking high-performance intermediates and procurement managers aiming for supply chain optimization in the electronic chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized tetrathiafulvalene derivatives for liquid crystal applications has been plagued by inefficient multi-step sequences that suffer from low overall yields and difficult purification protocols. Traditional routes often rely on harsh reaction conditions that can degrade sensitive chiral side chains, leading to racemization and a loss of the desired mesophase properties. Furthermore, conventional methods frequently struggle to achieve the high degree of molecular order required for stable hexagonal columnar phases, resulting in materials with broad, ill-defined phase transitions that are unsuitable for precision electronic devices. The presence of sulfur-containing impurities and unreacted precursors in older methodologies necessitates extensive chromatographic separation, driving up production costs and extending lead times for commercial scale-up. These technical bottlenecks have limited the widespread adoption of TTF-based liquid crystals in high-value applications where material consistency is paramount.
The Novel Approach
The methodology outlined in the patent data presents a transformative solution by utilizing a specific condensation and self-coupling strategy that preserves the integrity of the cholestenyl moieties while efficiently constructing the TTF core. This novel approach leverages the reactivity of zinc dithiolene salts with omega-bromo fatty acid esters to install the side chains early in the synthesis, ensuring uniform substitution patterns that are critical for liquid crystallinity. The subsequent desulfurization and coupling steps are optimized to proceed under controlled thermal conditions, minimizing side reactions and maximizing the formation of the target disubstituted and tetrasubstituted products. By employing triethyl sulfite as both solvent and reagent in the final coupling stage, the process eliminates the need for exotic catalysts, simplifying the workup and enhancing the scalability of the manufacturing process. This streamlined pathway not only improves chemical efficiency but also guarantees the formation of highly ordered mesophases with sharp transition temperatures.
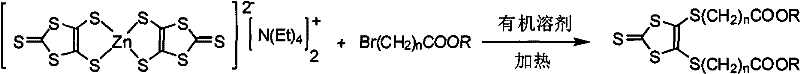
Mechanistic Insights into Zinc-Mediated Condensation and Self-Coupling
The initial stage of the synthesis involves a nucleophilic substitution reaction where the bis(1,3-dithiole-2-thione-4,5-dithiol)zinc bis(tetraethylammonium) salt acts as a potent nucleophile against the omega-bromo fatty acid cholesteryl ester. This step is mechanistically significant as it establishes the carbon-sulfur bonds that tether the bulky cholestenyl groups to the dithiole ring, a structural feature that dictates the spacing and packing of the molecules in the liquid crystal state. The reaction proceeds efficiently in polar aprotic solvents like acetonitrile or DMF, where the zinc complex remains soluble and reactive, allowing for the formation of the 4,5-bis(cholestenyloxycarbonylalkylthio)-1,3-dithiole-2-thione intermediate with yields consistently exceeding 70 percent. Careful control of the stoichiometry and temperature during this phase is essential to prevent dialkylation issues or hydrolysis of the ester linkage, ensuring that the final product maintains its chiral purity and thermal stability profile.
Following the installation of the side chains, the transformation of the thione group to a carbonyl group via mercuric acetate treatment serves as a crucial activation step for the final dimerization. This desulfurization process converts the 1,3-dithiole-2-thione into the corresponding 1,3-dithiol-2-one, which is the active species required for the subsequent self-coupling reaction to form the tetrathiafulvalene backbone. The final coupling occurs in a triethyl sulfite medium at elevated temperatures between 100°C and 160°C, facilitating the elimination of sulfur and the formation of the central carbon-carbon double bond characteristic of the TTF core. This thermal self-coupling mechanism is highly advantageous as it avoids the use of transition metal catalysts that could contaminate the final electronic material, thereby simplifying the purification process and enhancing the electrical properties of the resulting liquid crystal compound.
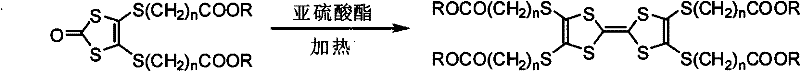
How to Synthesize Cholestenyl-TTF Derivatives Efficiently
The synthesis of these high-value electronic intermediates requires precise adherence to the patented three-step protocol to ensure the formation of the desired smectic or columnar phases with high fidelity. Operators must begin with the rigorous preparation of the dithiole-thione precursor, followed by careful desulfurization and finally the thermal coupling step, with intermediate purification via column chromatography to remove mercury salts and unreacted starting materials. The detailed standardized synthesis steps provided below outline the specific molar ratios, solvent choices, and temperature profiles necessary to replicate the high yields and purity levels reported in the patent documentation. Adhering to these parameters is critical for maintaining the structural integrity of the cholestenyl groups, which are responsible for the unique glass-forming and phase-stabilizing properties of the final material.
- Condense bis(1,3-dithiole-2-thione-4,5-dithiol)zinc salt with omega-bromo fatty acid cholesteryl ester in organic solvent to form the dithiole-2-thione intermediate.
- Perform desulfurization of the thione intermediate using mercuric acetate in dichloromethane to yield the corresponding 1,3-dithiol-2-one.
- Execute self-coupling of the dithiol-2-one in triethyl sulfite medium at elevated temperatures (100-160°C) to finalize the tetrathiafulvalene structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this patented synthesis route offers substantial benefits in terms of cost reduction in electronic chemical manufacturing by streamlining the production workflow and minimizing raw material waste. The high yields achieved at each step of the process, particularly the efficient coupling reaction, mean that less starting material is required to produce a kilogram of the final active ingredient, directly lowering the cost of goods sold. Furthermore, the use of commercially available reagents such as cholesteryl esters and zinc dithiolene salts ensures a stable supply chain that is not dependent on scarce or geopolitically sensitive catalysts, mitigating risks associated with raw material shortages. The simplicity of the purification process, which relies on standard chromatographic techniques rather than complex distillation or crystallization setups, further reduces the capital expenditure required for setting up production lines.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts in the final coupling step significantly lowers the direct material costs associated with producing these specialized liquid crystals. By utilizing triethyl sulfite as a dual-purpose reagent and solvent, the process reduces the volume of chemicals required and simplifies the downstream recovery and recycling operations, leading to substantial operational savings. Additionally, the high thermal stability of the intermediates allows for robust processing conditions that minimize batch failures and rework, contributing to a more predictable and cost-effective manufacturing environment.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals like fatty acid esters and basic sulfur heterocycles ensures that the supply chain for these TTF derivatives is resilient against market fluctuations. Since the synthesis does not require custom-synthesized catalysts or rare earth elements, procurement teams can secure long-term contracts for raw materials with multiple suppliers, guaranteeing continuity of supply for large-scale production runs. This reliability is crucial for meeting the demanding delivery schedules of downstream manufacturers in the display and semiconductor industries who require consistent quality and volume.
- Scalability and Environmental Compliance: The straightforward nature of the reaction sequence facilitates easy scale-up from laboratory gram quantities to multi-kilogram commercial batches without the need for specialized high-pressure equipment. The process generates manageable waste streams that can be treated using standard industrial effluent protocols, ensuring compliance with increasingly stringent environmental regulations regarding heavy metal discharge and organic solvent emissions. This scalability allows manufacturers to respond quickly to surges in demand for advanced organic electronic materials without compromising on quality or regulatory standing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrathiafulvalene-based liquid crystal compounds, derived directly from the experimental data and claims within the patent specification. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating these materials into new device architectures or existing manufacturing lines. The answers provided reflect the specific advantages of the patented method over prior art, focusing on yield, purity, and phase behavior characteristics that define the commercial value of the product.
Q: What are the thermal stability characteristics of these TTF-based liquid crystals?
A: The compounds exhibit high thermal stability with decomposition temperatures exceeding 290°C and maintain stable liquid crystal phases (Smectic A or Hexagonal Columnar) over wide temperature ranges, making them suitable for demanding electronic applications.
Q: How does the new synthesis method improve yield compared to traditional routes?
A: The patented three-step process achieves cumulative yields significantly higher than conventional multi-step syntheses, with individual step yields ranging from 70% to 89%, ensuring efficient material utilization and reduced waste generation.
Q: Are these compounds suitable for organic field-effect transistor (OFET) applications?
A: Yes, the rigid tetrathiafulvalene core combined with the ordered columnar or lamellar arrangement of the cholestenyl side chains provides excellent charge transport properties, ideal for organic semiconductor and OFET device fabrication.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrathiafulvalene Derivatives Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis for advanced electronic materials, leveraging deep expertise in sulfur chemistry and liquid crystal technology to deliver high-purity tetrathiafulvalene derivatives that meet the rigorous standards of the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your transition from R&D prototyping to full-scale manufacturing with seamless efficiency. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the phase transition temperatures and electrochemical properties of every batch, guaranteeing performance consistency for your end applications.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis routes can drive value for your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into how our manufacturing efficiencies translate into lower total costs for your supply chain. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your unique formulation needs, ensuring a successful partnership built on technical excellence and reliability.
