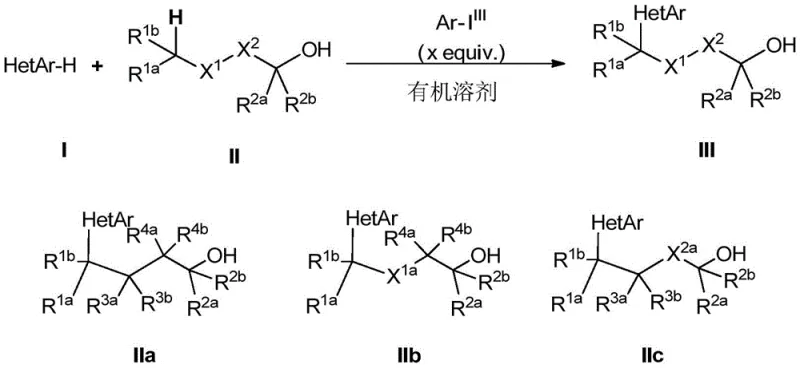
Advanced Metal-Free Synthesis of 4-Azaaryl Alkanols for Scalable Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex molecular architectures with high precision and efficiency. A significant breakthrough in this domain is documented in Chinese Patent CN108658853B, which discloses a novel synthesis method for 4-azaaryl alkanol compounds. This technology addresses a long-standing challenge in synthetic chemistry: the remote activation and functionalization of inert C(sp3)-H bonds. Traditionally, modifying aliphatic chains attached to heteroaromatic systems required multi-step sequences or harsh conditions that compromised sensitive functional groups. The disclosed invention leverages hypervalent iodine chemistry combined with photochemical or thermal energy to achieve direct intermolecular coupling. This approach not only streamlines the synthetic route but also aligns with modern green chemistry principles by avoiding toxic heavy metal catalysts. For organizations aiming to secure a reliable pharmaceutical intermediate supplier, understanding the mechanistic depth and operational simplicity of this patent is crucial for integrating these building blocks into next-generation drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of heteroaromatic rings has relied heavily on classical strategies such as the Minisci reaction. While effective for certain substrates, conventional Minisci protocols typically necessitate the use of stoichiometric amounts of transition metal oxidants, such as silver nitrate, along with strong acids and elevated temperatures. These conditions pose significant drawbacks for industrial applications, including the generation of heavy metal waste streams that require costly removal processes to meet stringent regulatory purity standards for active pharmaceutical ingredients (APIs). Furthermore, the radical species generated in traditional methods often lack precise regiocontrol, leading to mixtures of isomers that are difficult to separate. The reliance on pre-functionalized alkylating agents or harsh radical initiators also limits the scope of compatible functional groups, often resulting in decomposition of sensitive moieties like esters or halides. Consequently, the cost reduction in API manufacturing is hindered by low atom economy and complex purification workflows associated with these legacy technologies.
The Novel Approach
In stark contrast, the methodology described in CN108658853B introduces a paradigm shift by utilizing hypervalent iodine(III) reagents, such as bis(trifluoroacetoxy)iodobenzene (PIFA), to mediate the reaction under remarkably mild conditions. This novel approach enables the direct use of free alkanols as radical precursors, eliminating the need for pre-activation steps that reduce overall yield. The reaction can be driven by visible light irradiation, specifically using blue LEDs, or simply by mild thermal energy, offering flexibility in process engineering. By operating at room temperature or slightly elevated temperatures (50-80°C), the method preserves the integrity of thermally labile functional groups. The metal-free nature of this transformation is a distinct advantage, as it removes the burden of residual metal testing and cleanup, thereby significantly enhancing the efficiency of the production workflow. This innovation provides a practical strategy for the late-stage functionalization of complex molecules, making it an invaluable tool for medicinal chemists.
Mechanistic Insights into Hypervalent Iodine Mediated C-H Activation
The core of this technological advancement lies in the unique ability of hypervalent iodine compounds to generate alkoxy radicals under mild conditions. Upon interaction with the alcohol substrate, the iodine(III) reagent forms a reactive intermediate, likely a hypoiodite species, which undergoes homolytic cleavage upon exposure to light or heat. This cleavage generates a transient alkoxy radical, a highly reactive species capable of abstracting hydrogen atoms from unactivated C-H bonds. What makes this mechanism particularly sophisticated is the intramolecular 1,5-hydrogen atom transfer (HAT) process. The alkoxy radical selectively abstracts a hydrogen atom from the delta-position (the 4-position relative to the oxygen), generating a carbon-centered radical at a specific remote site. This selectivity is governed by the favorable six-membered transition state geometry, ensuring that functionalization occurs precisely where intended, regardless of the bond dissociation energies of other C-H bonds in the molecule.
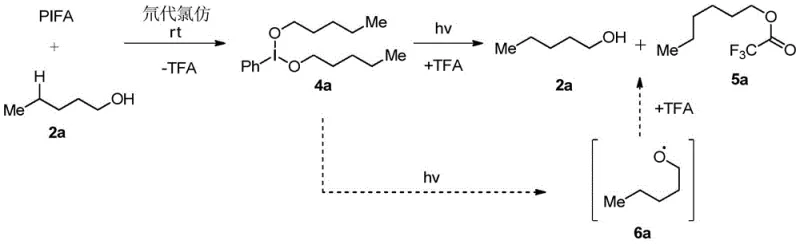
Following the generation of the remote carbon radical, the species adds to the electron-deficient heteroaromatic ring, forming a new carbon-carbon bond. The resulting cyclohexadienyl-type radical intermediate is then oxidized and deprotonated to restore aromaticity, yielding the final 4-azaaryl alkanol product. This cascade effectively bypasses the inherent inertness of aliphatic C-H bonds without requiring directing groups or chelating metals. The tolerance for various functional groups, including halides, esters, and even sensitive olefins, underscores the robustness of this radical manifold. For R&D teams focused on impurity control, understanding this mechanism is vital, as it predicts a clean impurity profile dominated by regioisomers rather than complex metal-ligand byproducts. The ability to tune the reaction outcome by adjusting the electronic properties of the hypervalent iodine reagent further enhances the versatility of this synthetic platform for diverse chemical spaces.
How to Synthesize 4-Azaaryl Alkanol Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and selectivity. The process generally involves dissolving the heteroaromatic substrate and the alcohol in a suitable organic solvent, such as dichloromethane or acetonitrile, under an inert atmosphere. The hypervalent iodine oxidant is then added, and the mixture is subjected to irradiation or heating. Detailed standard operating procedures regarding stoichiometry, light intensity, and workup protocols are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent data to ensure successful execution in a laboratory or pilot plant setting.
- Mix the azaaryl compound and alcohol substrate in an organic solvent under an inert atmosphere.
- Add the hypervalent iodo aryl compound (e.g., PIFA) to the reaction mixture.
- Irradiate with blue LEDs or apply heat to initiate the radical cascade and isolate the product after workup.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this metal-free synthesis technology offers transformative benefits that extend beyond mere chemical elegance. The elimination of transition metal catalysts fundamentally alters the cost structure of manufacturing complex intermediates. Traditional methods often incur significant expenses related to the purchase of precious metal catalysts and the subsequent implementation of scavenging technologies to remove metal residues to ppm levels. By removing this requirement entirely, the process achieves substantial cost savings in raw material acquisition and waste management. Furthermore, the simplified purification process reduces the consumption of silica gel and solvents during chromatography, contributing to a lower environmental footprint and reduced operational expenditures. This efficiency translates directly into improved margins for high-volume production campaigns.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by its high atom economy and the use of commercially abundant reagents. Unlike processes that require custom-synthesized radical precursors, this method utilizes simple, off-the-shelf alkanols and hypervalent iodine compounds. The avoidance of expensive noble metals like palladium or silver removes a major variable cost driver. Additionally, the mild reaction conditions reduce energy consumption associated with heating or cooling large-scale reactors. These factors combine to create a highly cost-effective manufacturing route that supports competitive pricing strategies for downstream API production without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the broad availability of the requisite starting materials. Hypervalent iodine reagents and common heteroaromatics are produced by multiple global suppliers, mitigating the risk of single-source bottlenecks. The robustness of the reaction conditions means that the process is less susceptible to variations in utility quality, such as fluctuations in cooling water temperature or power stability, which can plague sensitive catalytic processes. This reliability ensures consistent batch-to-batch quality and predictable delivery schedules, which are critical metrics for supply chain heads managing just-in-time inventory models for critical drug substances.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden challenges related to heat transfer and mixing, but this photochemical or thermal radical process is inherently scalable. The use of flow chemistry setups for the light-driven variant is particularly promising for ton-scale production, offering superior control over photon flux and reaction residence time. Moreover, the green chemistry profile of the method aligns with increasingly stringent environmental regulations. The reduction in heavy metal waste simplifies compliance with discharge permits and lowers the costs associated with hazardous waste disposal. This sustainability advantage enhances the corporate social responsibility profile of the manufacturing operation while future-proofing the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
To address common technical and commercial inquiries regarding this synthesis technology, we have compiled a set of frequently asked questions based on the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and strategic value of implementing this method within your existing manufacturing framework. Understanding these details will facilitate more informed decision-making regarding process adoption and vendor selection for your upcoming projects.
Q: What are the advantages of this metal-free C-H activation method over traditional Minisci reactions?
A: Unlike traditional Minisci reactions that often require transition metal catalysts like silver nitrate and harsh oxidative conditions, this method utilizes hypervalent iodine reagents under mild light or thermal conditions. This eliminates heavy metal contamination risks, simplifies downstream purification, and offers superior regioselectivity for remote functionalization.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process is designed for scalability. It operates at room temperature or mild heating (50-80°C) using commercially available reagents like PIFA. The absence of expensive transition metals and the use of simple light sources or standard heating equipment make it highly suitable for commercial scale-up of complex organic molecules.
Q: What is the regioselectivity profile of this alcohol functionalization reaction?
A: The reaction exhibits excellent regioselectivity for the 4-position relative to the hydroxyl group. This is achieved through a specific 1,5-hydrogen atom transfer (HAT) mechanism mediated by alkoxy radicals, allowing for precise functionalization of distant C(sp3)-H bonds that are typically inert in other chemical environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Azaaryl Alkanol Supplier
The technical potential of the 4-azaaryl alkanol synthesis described in CN108658853B represents a significant opportunity for advancing drug development programs. However, translating patent literature into commercial reality requires a partner with deep process engineering expertise and a commitment to quality. NINGBO INNO PHARMCHEM stands ready to support your initiatives as a trusted CDMO partner. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the highest industry standards for pharmaceutical intermediates.
We invite you to explore how this innovative chemistry can optimize your synthesis routes and reduce your overall time to market. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific molecule, evaluating the feasibility of adapting this metal-free protocol to your portfolio. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a supply chain that prioritizes innovation, quality, and long-term partnership, ensuring your projects proceed without interruption.
