Advanced Synthesis of Bis(triisopropylcyclopentadienyl)strontium for High-Performance ALD Applications
Advanced Synthesis of Bis(triisopropylcyclopentadienyl)strontium for High-Performance ALD Applications
The semiconductor industry's relentless drive toward smaller node sizes has placed unprecedented demands on the purity and performance of Atomic Layer Deposition (ALD) precursors. A pivotal advancement in this domain is detailed in patent CN113563390B, which discloses a highly efficient preparation method for bis(triisopropylcyclopentadienyl)strontium, a critical strontium source material. This innovation addresses long-standing challenges in organometallic synthesis by utilizing a novel one-pot reaction system involving strontium hydride and hexamethyldisilazane. Unlike conventional routes that struggle with solvent coordination and metal impurities, this patented approach delivers a product with exceptional metal purity reaching 5N levels and yields as high as 87%. For R&D directors and procurement specialists alike, this represents a significant leap forward in securing reliable supply chains for high-k dielectric materials and ferroelectric memory applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkaline earth metal cyclopentadienyl derivatives has been fraught with operational complexities and purity limitations that hinder industrial scalability. Traditional methods often rely on the reaction of strontium iodide with sodium salts of cyclopentadiene derivatives in solvents like tetrahydrofuran (THF), or the direct reaction of metal strontium with cyclopentadienes in liquid ammonia. These legacy processes suffer from severe drawbacks, including the formation of stable solvent adducts where THF molecules coordinate tightly to the strontium center, necessitating rigorous and often damaging vacuum sublimation steps to remove them. Furthermore, the use of liquid ammonia requires cryogenic conditions around -40°C, introducing significant safety hazards and energy costs, while typically resulting in mediocre yields of only about 50%. The presence of residual solvent molecules and metal impurities from halide salts compromises the thermal stability and vapor pressure characteristics required for precise ALD processes.
The Novel Approach
The methodology outlined in CN113563390B fundamentally reimagines the synthetic pathway by employing strontium hydride and hexamethyldisilazane (HMDS) in a streamlined one-pot procedure. This innovative route eliminates the need for hazardous cryogenic reagents and avoids the introduction of halide contaminants entirely. By leveraging HMDS not merely as a solvent but as an active participant in the coordination sphere, the process facilitates a smooth ligand exchange where the bulky triisopropylcyclopentadienyl groups displace the silylamide ligands under reflux conditions. This strategic design prevents the entrapment of solvent molecules within the crystal lattice of the final product, thereby simplifying purification. The result is a robust manufacturing protocol that operates under mild conditions yet achieves superior outcomes, positioning it as a preferred choice for cost reduction in electronic chemical manufacturing where consistency and purity are paramount.
Mechanistic Insights into SrH2-Mediated Ligand Exchange
The core of this technological breakthrough lies in the unique dual role of hexamethyldisilazane, which acts as both the reaction medium and a transient ligand to stabilize the reactive strontium species. In the initial phase, strontium hydride dissolves in HMDS to form a coordinated intermediate that moderates the reactivity of the strontium center. When 1,2,4-triisopropylcyclopentadiene is introduced, the steric bulk of the isopropyl groups combined with the thermal energy of reflux drives the displacement of the HMDS ligands. This mechanism is crucial because direct reaction between strontium hydride and the cyclopentadiene without HMDS would be excessively exothermic, risking the decomposition of the sensitive cyclopentadiene structure. The boiling point differential further aids the process; as HMDS distills out during reflux, the equilibrium shifts towards the formation of the target bis(triisopropylcyclopentadienyl)strontium, effectively pushing the reaction to completion without requiring complex workup procedures.
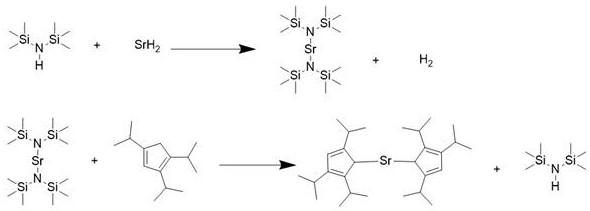
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity OLED material and semiconductor precursors. The absence of halide ions, which are common in iodide-based routes, eliminates a major source of corrosion and defect formation in downstream deposition chambers. Additionally, the volatility of the byproduct (HMDS) allows it to be easily separated from the non-volatile organometallic product during the concentration phase. This inherent selectivity ensures that the final sublimation step, conducted at 100-115°C under high vacuum, serves primarily to remove trace organic residues rather than breaking strong solvent-metal bonds. Consequently, the process yields a product with a metal purity of 5N, meeting the stringent specifications required for growing nanometer-thick SrO films with uniform step coverage and excellent electrical properties.
How to Synthesize Bis(triisopropylcyclopentadienyl)strontium Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize the 87% yield potential described in the patent data. The process begins with the dissolution of strontium hydride in HMDS under an inert atmosphere, followed by the controlled addition of the cyclopentadiene ligand. Maintaining the reaction temperature between 125-200°C during the reflux stage is critical to ensure complete ligand exchange while preventing thermal degradation. The following guide outlines the standardized operational parameters derived from the patent examples to assist process engineers in replicating these results.
- Under a protective atmosphere, dissolve strontium hydride solid powder in hexamethyldisilazane with stirring to form a mixed solution.
- Dropwise add 1,2,4-triisopropylcyclopentadiene to the mixture, controlling temperature below 30°C during addition, then heat to reflux at 125-200°C for 12-48 hours.
- After reaction completion, evaporate solvent under reduced pressure and purify the crude product via vacuum sublimation at 100-115°C to achieve 5N metal purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible benefits that extend beyond mere chemical yield. The elimination of liquid ammonia and the reduction of purification steps translate directly into lower operational expenditures and reduced safety compliance burdens. By adopting a process that utilizes commercially available and stable reagents like strontium hydride and HMDS, manufacturers can mitigate the risks associated with sourcing hazardous cryogenic fluids. This stability enhances supply chain reliability, ensuring consistent production schedules without the interruptions often caused by the handling complexities of traditional methods. Furthermore, the high purity achieved reduces the need for extensive quality control testing and reprocessing, streamlining the path from reactor to shipment.
- Cost Reduction in Manufacturing: The one-pot nature of this synthesis significantly simplifies the production workflow by removing the need for multiple solvent exchanges and complex adduct dissociation steps. By avoiding the use of expensive strontium iodide and eliminating the energy-intensive cryogenic cooling required for liquid ammonia reactions, the overall utility consumption is drastically lowered. The high yield of 87% means that less raw material is wasted per kilogram of finished product, directly improving the cost basis for high-purity ALD precursor production. Additionally, the simplified purification process reduces the load on vacuum sublimation equipment, extending maintenance intervals and lowering capital depreciation costs per unit produced.
- Enhanced Supply Chain Reliability: Sourcing strontium hydride and hexamethyldisilazane is generally more straightforward and less regulated than procuring large quantities of liquid ammonia or specialized organolithium reagents. This accessibility reduces the risk of supply disruptions due to regulatory changes or transportation restrictions on hazardous materials. The robustness of the reaction conditions, which tolerate a range of reflux temperatures and times without compromising the 5N metal purity, allows for greater flexibility in manufacturing scheduling. This operational resilience ensures that delivery commitments to semiconductor fabs can be met consistently, even in the face of minor variations in raw material batches or utility availability.
- Scalability and Environmental Compliance: The absence of halide byproducts and the minimal generation of hazardous waste make this process inherently more environmentally friendly and easier to scale. Traditional methods often generate significant amounts of salt waste and require complex effluent treatment for ammonia residues, whereas this route primarily produces volatile silazanes that can be recovered or treated with standard abatement systems. The ability to scale from gram-scale laboratory synthesis to multi-kilogram commercial production without altering the fundamental chemistry supports rapid capacity expansion. This scalability is essential for meeting the growing demand for strontium-containing dielectrics in advanced memory devices without incurring prohibitive environmental compliance costs.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this patented synthesis method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this precursor into their existing ALD processes. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation.
Q: What is the metal purity achievable with this new synthesis method?
A: The patented process achieves a metal purity as high as 5N (99.999%), verified by ICP-OES, which is critical for preventing defects in atomic layer deposition of SrO thin films.
Q: How does this method avoid solvent coordination issues found in older techniques?
A: By using hexamethyldisilazane as both solvent and reactant, the method ensures no solvent molecules remain coordinated to the strontium center in the final product, unlike THF-based methods.
Q: What is the typical yield for bis(triisopropylcyclopentadienyl)strontium using this route?
A: Optimization of reaction time and stoichiometry allows for yields up to 87%, significantly higher than the approximately 50% yield observed in traditional liquid ammonia methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bis(triisopropylcyclopentadienyl)strontium Supplier
As the demand for high-performance electronic materials continues to surge, partnering with a manufacturer who understands the nuances of complex organometallic synthesis is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize advanced analytical techniques to verify that every batch meets the 5N metal purity standards required for next-generation semiconductor applications. We are dedicated to supporting your R&D and production goals with materials that deliver reliable performance in every deposition cycle.
We invite you to engage with our technical procurement team to discuss how our optimized synthesis capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing processes can reduce your total cost of ownership. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that our bis(triisopropylcyclopentadienyl)strontium aligns perfectly with your technical and commercial objectives.
