Scalable Synthesis of Tyrosine Semicarbazide Hydrazone for Advanced Protease Stabilizer Manufacturing
Scalable Synthesis of Tyrosine Semicarbazide Hydrazone for Advanced Protease Stabilizer Manufacturing
The development of environmentally friendly and highly efficient protease stabilizers has become a critical focus in the modern detergent industry, driving a shift away from traditional boron-based compounds which pose environmental toxicity concerns. Patent CN108530322B introduces a groundbreaking synthetic methodology for producing tyrosine semicarbazide hydrazone hydrochloride, a key intermediate that serves as a superior alternative for stabilizing proteases in liquid detergent formulations. This technology addresses the longstanding chemical challenge of handling reactive aminoaldehydes by utilizing a stable semicarbazide protection strategy, thereby enabling the production of high-purity intermediates suitable for complex peptide coupling. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a significant leap forward in process safety and scalability, offering a robust pathway to manufacture next-generation bio-enzymatic additives without the logistical burdens associated with cryogenic chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing tyrosinal and its derivatives, which are essential precursors for protease stabilizers, have historically relied on aggressive and hazardous reduction protocols. Specifically, existing literature describes the reduction of amides to aldehydes using lithium aluminum hydride or diisobutylaluminum hydride (DIBAL-H) at extremely low temperatures, typically around -78°C. These organoaluminum catalysts are not only pyrophoric and dangerous to handle on a large scale but also necessitate expensive cryogenic infrastructure that drastically increases capital expenditure and operational costs. Furthermore, the resulting aminoaldehyde products are inherently unstable; the free amino group tends to react intramolecularly with the aldehyde group to form Schiff bases, leading to product degradation and inconsistent quality. This instability makes storage, transport, and subsequent chemical modification exceptionally difficult, creating a bottleneck for the reliable supply of high-quality protease stabilizer intermediates.
The Novel Approach
The innovative process disclosed in the patent circumvents these critical limitations by employing a mild, six-step synthetic route that utilizes readily available reagents and avoids extreme conditions. Instead of dangerous organometallic reducers, the process leverages sodium borohydride for the reduction of esters to alcohols and a TEMPO-catalyzed sodium hypochlorite system for the subsequent oxidation to aldehydes. This approach allows reactions to proceed at near-ambient temperatures (0°C to 35°C), significantly enhancing operational safety and energy efficiency. Crucially, the strategy involves protecting the reactive aldehyde moiety immediately upon formation by reacting it with semicarbazide. This converts the unstable aldehyde into a crystalline, stable semicarbazide hydrazone derivative that is resistant to degradation and compatible with standard peptide coupling conditions. By integrating dual-protection strategies for the amino and phenolic groups using Boc chemistry, the method ensures high regioselectivity and purity throughout the synthesis.
Mechanistic Insights into Boc-Protection and TEMPO Oxidation
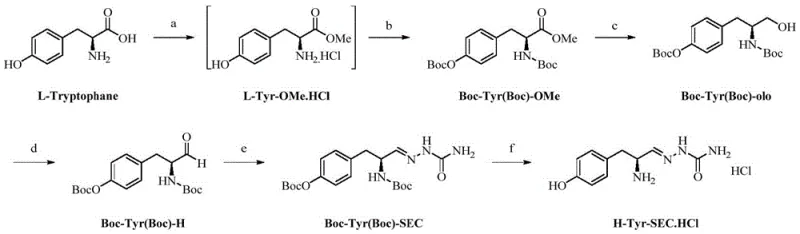
The core chemical ingenuity of this process lies in its sophisticated protection group strategy and the selective oxidation mechanism. The synthesis begins with the simultaneous protection of both the alpha-amino group and the phenolic hydroxyl group of tyrosine using di-tert-butyl dicarbonate (Boc2O). This dual-protection is vital because it prevents side reactions during the subsequent reduction and oxidation steps, ensuring that the reactivity is directed solely at the carboxylic acid derivative. Following the reduction of the methyl ester to a primary alcohol using sodium borohydride, the process employs a catalytic oxidation cycle involving 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) and sodium bromide. In this mechanism, TEMPO acts as a stable nitroxyl radical mediator that selectively oxidizes the primary alcohol to the aldehyde using sodium hypochlorite as the terminal oxidant. This catalytic cycle is highly efficient and prevents over-oxidation to the carboxylic acid, a common pitfall in aldehyde synthesis.
Once the fragile aldehyde intermediate (Boc-Tyr(Boc)-H) is generated, it is immediately trapped by semicarbazide to form the hydrazone linkage. This derivatization is thermodynamically favorable and effectively 'locks' the aldehyde in a stable form, preventing the nucleophilic attack by the nearby amino group that would otherwise lead to cyclization or polymerization. The final deprotection step utilizes hydrogen chloride in methanol to cleave the acid-labile Boc groups, yielding the target tyrosine semicarbazide hydrazone hydrochloride salt. This salt form is not only chemically stable for long-term storage but also possesses the necessary solubility and reactivity profiles to function effectively as a building block for the 'X-B1-B0-H' type protease stabilizers described in related art. The entire sequence demonstrates a masterful balance of reactivity control and functional group tolerance.
How to Synthesize Tyrosine Semicarbazide Hydrazone Efficiently
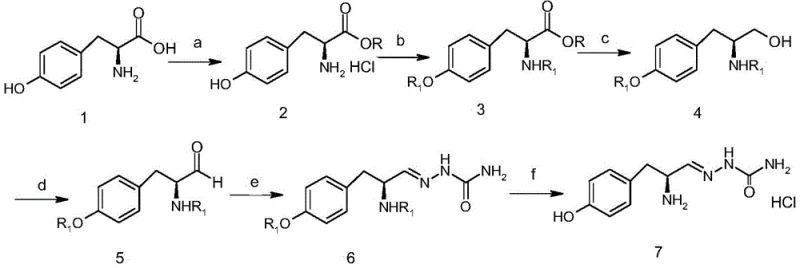
The synthesis of this high-value intermediate is designed for industrial feasibility, prioritizing telescoped operations to minimize unit operations and solvent usage. The process initiates with the esterification of L-tyrosine, followed by in-situ protection, reduction, and oxidation without the need for isolating solid intermediates until the final step. This continuous flow of reactivity reduces the physical footprint required for manufacturing and lowers the risk of material loss during transfer and purification. The detailed standardized synthetic steps, including specific molar ratios, temperature controls, and workup procedures for each of the six stages, are outlined below to guide process engineers in replicating this high-yield protocol.
- Esterify L-Tyrosine using thionyl chloride and methanol to form the methyl ester hydrochloride intermediate.
- Protect both the amino and phenolic hydroxyl groups using di-tert-butyl dicarbonate (Boc2O) to yield the di-Boc protected ester.
- Reduce the ester group to a primary alcohol using sodium borohydride in methanol under mild conditions.
- Oxidize the alcohol to the corresponding aldehyde using a TEMPO-catalyzed sodium hypochlorite system.
- React the unstable aldehyde intermediate with semicarbazide to form the stable semicarbazide hydrazone derivative.
- Remove the Boc protecting groups using hydrogen chloride in methanol to obtain the final hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound economic and logistical benefits that extend beyond simple yield improvements. By eliminating the dependency on cryogenic conditions and pyrophoric reagents, manufacturers can utilize standard stainless steel reactors without the need for specialized low-temperature cooling systems, resulting in substantially reduced capital investment and maintenance costs. The use of commodity chemicals such as sodium borohydride and bleach (sodium hypochlorite) replaces expensive and supply-constrained organometallic reagents, insulating the production cost from volatile market fluctuations associated with specialty catalysts. Furthermore, the ability to telescope multiple steps without intermediate purification drastically cuts down on solvent consumption and waste disposal costs, aligning the manufacturing process with increasingly stringent environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The replacement of hazardous, low-temperature reduction steps with ambient temperature sodium borohydride reductions eliminates the high energy costs associated with maintaining -78°C environments. Additionally, the avoidance of expensive chromatography purification for intermediates, relying instead on simple phase separations and concentration, leads to significant savings in both solvent procurement and waste treatment. The overall process efficiency is enhanced by the high atom economy of the TEMPO oxidation system, which minimizes the generation of heavy metal waste streams often associated with traditional oxidation methods like Swern or PCC.
- Enhanced Supply Chain Reliability: All raw materials utilized in this six-step sequence, including L-tyrosine, di-tert-butyl dicarbonate, and semicarbazide, are bulk commodity chemicals available from multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague processes relying on proprietary or exotic catalysts. The stability of the final hydrochloride salt product ensures a longer shelf life and simplifies logistics, allowing for larger batch production runs and strategic stockpiling without the fear of rapid degradation, thus guaranteeing consistent availability for downstream detergent formulators.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, as evidenced by the successful execution of hundred-gram scale experiments described in the patent data which maintained high purity and yield. The absence of heavy metals and the use of aqueous workups facilitate easier compliance with environmental discharge standards. The telescoped nature of the synthesis reduces the total volume of organic solvents required per kilogram of product, lowering the facility's overall carbon footprint and simplifying the permitting process for capacity expansion in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of tyrosine semicarbazide hydrazone hydrochloride. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational advantages and chemical properties of this intermediate for potential partners and technical evaluators.
Q: Why is tyrosine semicarbazide hydrazone used instead of free tyrosinal in protease stabilizers?
A: Free aminoaldehydes like tyrosinal are chemically unstable and prone to self-reaction (Schiff base formation) or degradation. The semicarbazide hydrazone derivative acts as a stable 'masked' aldehyde that can be easily regenerated in situ or reacted directly with peptide sequences without side reactions, ensuring the stability of the final protease stabilizer.
Q: What are the safety advantages of this synthesis route compared to prior art?
A: Conventional methods rely on hazardous reducing agents like lithium aluminum hydride or diisobutylaluminum hydride (DIBAL-H) which require cryogenic conditions (-78°C) and pose significant fire risks. This novel process utilizes sodium borohydride and TEMPO-catalyzed oxidation, which operate at near-ambient temperatures (0-35°C), drastically improving operational safety and eliminating the need for specialized low-temperature infrastructure.
Q: Does this process require purification of intermediates between steps?
A: No, one of the key commercial advantages of this method is that the intermediates (such as the ester, alcohol, and aldehyde forms) do not require isolation or fine purification. They can be telescoped directly into the subsequent reaction steps, which significantly reduces solvent consumption, minimizes material loss, and shortens the overall production cycle time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tyrosine Semicarbazide Hydrazone Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the formulation of advanced enzymatic detergents and pharmaceutical stabilizers. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch, guaranteeing that our tyrosine derivatives meet the exacting standards required for protease stabilization applications.
We invite you to collaborate with us to optimize your supply chain for protease stabilizer ingredients. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized synthetic route can reduce your total landed cost. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation, and let us support your innovation in the personal care and fine chemical sectors.
