Optimizing Asenapine Intermediate Production: A Technical Breakthrough in Refining Efficiency
Optimizing Asenapine Intermediate Production: A Technical Breakthrough in Refining Efficiency
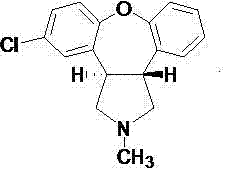
The global demand for central nervous system (CNS) therapeutics continues to drive rigorous innovation in the synthesis of key pharmaceutical intermediates. Specifically, the antipsychotic agent Asenapine, known commercially as SAPHRIS, requires high-purity precursors to meet stringent regulatory standards for clinical application. A pivotal advancement in this domain is documented in patent CN103772401A, which introduces a novel refining method for the critical intermediate 11-chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenzo[2,3:6,7]oxepino[4,5-c]pyrryl-1-one. This patent addresses a significant bottleneck in the existing manufacturing landscape, where traditional purification protocols often result in suboptimal yields and complex waste streams. By shifting the paradigm from laborious liquid-liquid extractions to a streamlined solvent-based crystallization and filtration approach, this technology offers a robust pathway for enhancing both the economic and operational efficiency of Asenapine production lines.
The significance of this technological leap cannot be overstated for stakeholders managing the supply chains of psychotropic medications. Asenapine functions as a broad-spectrum antagonist for norepinephrine, serotonin, and dopamine, making its synthesis chemically demanding due to the complexity of the dibenzoxepino-pyrrole scaffold. The intermediate in question serves not only as a direct precursor but also plays a role in the management of specific synthetic impurities, such as Impurity E. Consequently, the ability to refine this molecule with high fidelity directly impacts the overall quality profile of the final Active Pharmaceutical Ingredient (API). The data presented in the patent suggests that through careful solvent selection, specifically utilizing an ethyl acetate system, manufacturers can achieve near-quantitative recovery rates, a metric that fundamentally alters the cost structure of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN103772401A, the industry standard for processing this intermediate relied heavily on methodologies disclosed in earlier literature, such as US Patent 4145434A. These conventional approaches typically involved a multi-step workup procedure characterized by the use of methanol as a reaction solvent, followed by acidification with hydrochloric acid and subsequent liquid-liquid extraction using organic solvents like ethyl acetate. While chemically feasible, this traditional route is plagued by inherent inefficiencies that accumulate significantly at scale. The reliance on pH-dependent extraction introduces multiple points of failure, including the formation of stable emulsions that trap product, incomplete phase separation, and substantial product loss into the aqueous waste streams. Furthermore, the reported yields for these legacy methods hover around 72% to 76%, indicating that nearly a quarter of the valuable synthetic material is discarded during purification. This loss is not merely a financial burden but also an environmental one, as it necessitates the processing of larger batches of starting materials to achieve the same output, thereby increasing the load on waste treatment facilities and consuming more energy and raw resources.
The Novel Approach
In stark contrast to the cumbersome extraction protocols of the past, the novel approach outlined in the patent leverages the differential solubility properties of the target compound and its impurities in specific organic solvent systems. The core innovation lies in the substitution of the complex acid-base workup with a direct dissolution and filtration strategy using ethyl acetate. By dissolving the crude reaction mixture in ethyl acetate at controlled temperatures, typically around 30°C, the process allows for the selective retention of the desired intermediate in the solution phase while insoluble inorganic salts and polymeric byproducts are removed via simple filtration. This eliminates the need for aqueous washes and pH adjustments that often degrade sensitive heterocyclic structures. The result is a dramatic improvement in process mass intensity (PMI) and a yield surge to approximately 98%. This method transforms the purification step from a yield-limiting bottleneck into a high-efficiency recovery operation, ensuring that the maximum amount of synthesized material proceeds to the next stage of API manufacturing.
Mechanistic Insights into Solvent-Driven Purification Dynamics
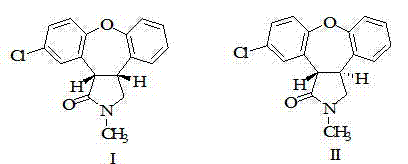
To fully appreciate the technical merit of this refining method, one must analyze the physicochemical interactions between the solute and the solvent matrix. The intermediate, 11-chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenzo[2,3:6,7]oxepino[4,5-c]pyrryl-1-one, possesses a rigid polycyclic structure with specific polar and non-polar regions. In the conventional methanol-water-acid systems, the protonation of the nitrogen atom increases water solubility, but the subsequent neutralization and extraction steps are kinetically slow and thermodynamically prone to equilibrium losses. The ethyl acetate system operates on a different principle: it exploits the moderate polarity of ethyl acetate to maintain the neutral form of the intermediate in solution while excluding highly polar or ionic impurities that might arise from the preceding Grignard-type or cyclization reactions. The absence of water in the primary isolation step prevents hydrolysis side reactions that could compromise the integrity of the oxepino ring system. Moreover, the data indicates that this solvent environment is particularly effective at managing the ratio of isomers (labeled I and II in the structural analysis), ensuring that the final solid maintains a consistent stereochemical profile required for downstream biological activity.
Furthermore, the control of impurities is achieved through a combination of solubility thresholds and physical filtration. The patent data highlights that alternative solvent systems, such as methylene dichloride-water-sulfuric acid, resulted in significantly lower chemical purity (80.23%) despite decent yields. This suggests that while dichloromethane is a good solvent, the acidic aqueous workup reintroduces impurities or fails to separate polar byproducts effectively. In contrast, the ethyl acetate system achieves a chemical purity of 98.5%. This high level of purity is likely due to the fact that many polar degradation products and inorganic residues remain insoluble in ethyl acetate and are removed in the filter cake before the solvent is evaporated. This "negative purification" mechanism—removing what you don't want rather than extracting what you do—is inherently more robust and less sensitive to minor variations in operating parameters, making it ideal for maintaining batch-to-batch consistency in a GMP environment.
How to Synthesize 11-chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenzo[2,3:6,7]oxepino[4,5-c]pyrryl-1-one Efficiently
The implementation of this refining protocol requires precise adherence to solvent volumes and temperature controls to replicate the high yields reported in the patent literature. The process begins after the completion of the precursor synthesis, where the reaction mixture is concentrated to remove volatile components like methanol. The critical step involves the addition of ethyl acetate, which acts as both a solvent for the product and a washing medium for impurities. Operators must ensure that the dissolution is complete at mild temperatures to avoid thermal degradation, followed by a rigorous filtration step to clarify the solution. The final isolation is achieved by removing the ethyl acetate under reduced pressure, yielding a solid that requires minimal further processing. For a detailed breakdown of the specific operational parameters, reagent ratios, and safety considerations, please refer to the standardized synthesis guide below.
- Complete the precursor synthesis reaction and remove the initial reaction solvent (methanol) under reduced pressure to obtain the crude residue.
- Dissolve the crude residue in ethyl acetate at controlled temperatures (approx. 30°C) to ensure full solubilization of the target intermediate while leaving insoluble impurities behind.
- Perform hot filtration to remove insoluble particulates, followed by solvent removal from the filtrate to isolate the high-purity solid product with yields approaching 98%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refining technology translates directly into tangible bottom-line improvements and risk mitigation. The most immediate impact is the drastic reduction in raw material costs per kilogram of output. By elevating the yield from the historical average of roughly 72% to nearly 98%, the effective consumption of expensive starting materials is cut by more than a quarter. This efficiency gain does not require capital investment in new reactors but rather a modification of the downstream processing recipe, allowing for rapid integration into existing facilities. Additionally, the elimination of complex aqueous workups reduces the volume of wastewater generated, lowering the operational expenditures associated with environmental compliance and waste disposal. The simplicity of the process also reduces the man-hours required for execution, freeing up production capacity for other high-value campaigns.
- Cost Reduction in Manufacturing: The shift to an ethyl acetate-based refining system eliminates the need for large volumes of mineral acids and bases, which are not only consumable costs but also pose corrosion risks to equipment. More importantly, the near-quantitative yield means that the cost of goods sold (COGS) for this intermediate is significantly depressed. In a competitive generic pharmaceutical market, where margin compression is constant, a 20%+ improvement in yield at a key intermediate stage provides a substantial buffer against raw material price volatility. This cost advantage can be passed down the supply chain or retained as increased margin, offering strategic flexibility in pricing negotiations with API customers.
- Enhanced Supply Chain Reliability: The solvents utilized in this novel process, primarily ethyl acetate and methanol, are commodity chemicals with robust global supply networks. Unlike specialized reagents or catalysts that may face geopolitical supply constraints, these solvents are readily available from multiple vendors, reducing the risk of production stoppages due to material shortages. Furthermore, the simplified process flow reduces the cycle time for each batch. By removing time-consuming extraction and phase-separation steps, the turnaround time between batches is shortened, allowing for a more responsive supply chain that can adapt quickly to fluctuations in market demand for Asenapine maleate.
- Scalability and Environmental Compliance: From a scale-up perspective, the new method is inherently safer and more manageable. Liquid-liquid extractions on a multi-ton scale often present engineering challenges related to mixing efficiency and phase separation times. Replacing this with a dissolution-filtration-evaporation sequence simplifies the unit operations, making the transfer from pilot plant to commercial manufacturing seamless. Environmentally, the reduction in aqueous waste streams containing organic residues simplifies effluent treatment. The ability to recover and recycle ethyl acetate further enhances the green chemistry profile of the process, aligning with the increasingly strict sustainability mandates imposed by regulatory bodies and corporate ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential licensees and manufacturing partners. Understanding these nuances is critical for R&D teams evaluating process validation and for procurement officers assessing supplier capabilities.
Q: How does the ethyl acetate system improve yield compared to traditional methods?
A: Traditional methods involving acid-base extractions often suffer from product loss in aqueous phases and emulsion formation. The ethyl acetate system simplifies the workflow to dissolution and filtration, minimizing mechanical and solubility losses, thereby increasing yield from approximately 72% to 98%.
Q: What is the chemical purity achievable with this new refining method?
A: The optimized ethyl acetate solvent system consistently achieves a chemical purity of 98.5%, effectively controlling isomeric impurities and polar byproducts without the need for complex chromatographic separation.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process utilizes common, industrially available solvents like ethyl acetate and avoids hazardous or expensive reagents. The simplicity of the unit operations (dissolution, filtration, evaporation) makes it highly suitable for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 11-chloro-2,3,3a,12b-tetrahydro-2-methyl-1H-dibenzo[2,3:6,7]oxepino[4,5-c]pyrryl-1-one Supplier
The technical potential of the ethyl acetate refining method represents a significant opportunity for optimizing the production of Asenapine intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovative processes to life. Our facility is equipped with rigorous QC labs and stringent purity specifications that ensure every batch of intermediate meets the exacting standards necessary for CNS drug manufacturing. We understand that the transition from a laboratory patent to a commercial reality requires not just chemical knowledge but deep engineering expertise, which our team delivers consistently to our global partners.
We invite you to engage with our technical procurement team to discuss how this high-yield refining route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the specific economic benefits for your operation. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver this critical intermediate with the reliability and quality your organization demands.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
