Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Scalable Pharmaceutical Intermediate Production
Advanced Iridium-Catalyzed Asymmetric Hydrogenation for Scalable Pharmaceutical Intermediate Production
The landscape of chiral synthesis in the pharmaceutical industry is constantly evolving, driven by the relentless demand for higher purity intermediates and more sustainable manufacturing processes. A significant technological advancement in this domain is detailed in Chinese Patent CN111410604A, which discloses a novel method for the asymmetric hydrogenation of alkenoic acid compounds. This patent introduces a specialized class of iridium-phosphine oxazoline complexes that serve as highly efficient chiral catalysts, capable of transforming prochiral alkenoic acids into valuable chiral products with exceptional enantioselectivity. For R&D directors and process chemists, this technology represents a critical opportunity to access complex chiral building blocks that were previously difficult to synthesize with high optical purity. The core innovation lies in the specific structural design of the catalyst, which leverages strong coordination between the iridium center and the carboxyl group of the substrate to direct the stereochemical outcome of the hydrogenation reaction.
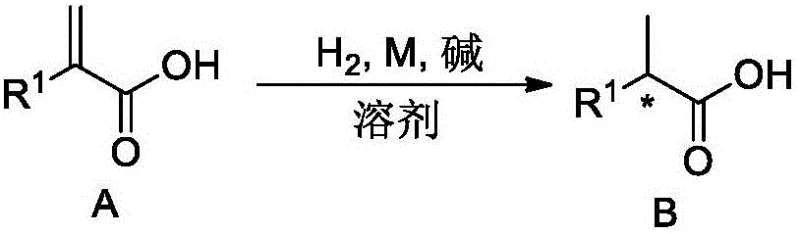
For procurement managers and supply chain heads, the implications of this patent extend beyond mere chemical curiosity; it offers a pathway to cost reduction in pharmaceutical intermediate manufacturing by streamlining synthetic routes. Traditional methods for generating chiral centers often rely on resolution techniques that inherently waste 50% of the material or require stoichiometric amounts of chiral auxiliaries, both of which drive up costs and environmental burden. In contrast, the catalytic asymmetric hydrogenation described in this patent utilizes sub-stoichiometric amounts of the iridium complex (catalyst loading as low as 0.00001 to 0.01 molar equivalents in the general description, though examples use S/C ratios around 400), making it economically viable for large-scale production. By adopting this technology, manufacturers can secure a reliable pharmaceutical intermediate supplier status, ensuring the continuous availability of high-value chiral acids needed for the synthesis of advanced therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydrogenation of functionalized alkenes, particularly those containing free carboxylic acid groups, has presented significant challenges for process chemists. Conventional rhodium-based catalysts, while effective for simple olefins, often suffer from poor activity or selectivity when faced with the coordinating ability of free carboxyl groups, which can poison the metal center or lead to non-selective background reactions. Furthermore, many existing protocols require harsh reaction conditions, such as extremely high hydrogen pressures or elevated temperatures, which pose safety risks and increase energy consumption in a commercial setting. The lack of robust catalysts that can tolerate diverse functional groups without extensive protection-deprotection sequences has frequently resulted in lengthy, multi-step synthetic routes. These inefficiencies not only inflate the cost of goods sold (COGS) but also extend the lead time for bringing new drug candidates to clinical trials, creating a bottleneck in the development pipeline that the industry is eager to resolve.
The Novel Approach
The technology outlined in Patent CN111410604A addresses these痛点 by introducing a tailored iridium-phosphine oxazoline (Ir-PHOX) catalyst system specifically designed to interact favorably with alkenoic acid substrates. Unlike traditional systems, this novel approach exploits the carboxyl group as a directing group, facilitating a coordinated interaction that enhances both reaction rate and stereocontrol. The patent demonstrates that by modifying the substituents on the oxazoline ring (R groups such as isopropyl or tert-butyl) and the counterions (such as BArF or PF6), the catalyst's performance can be finely tuned to achieve enantiomeric excess (ee) values exceeding 90% for specific substrates like the biphenyl derivative A-1. This level of precision allows for the direct synthesis of chiral acids like Compound B-1 without the need for downstream resolution, significantly simplifying the process flow. The ability to operate under moderate hydrogen pressures (e.g., 10 atm) and temperatures (50°C) further underscores the practicality of this method for commercial scale-up of complex pharmaceutical intermediates.
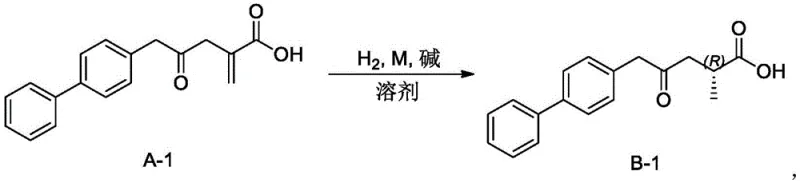
Mechanistic Insights into Iridium-Phosphine Oxazoline Catalyzed Hydrogenation
To fully appreciate the value of this technology for high-purity pharmaceutical intermediate production, one must understand the underlying mechanistic principles that govern its success. The iridium-phosphine oxazoline complex functions through a mechanism where the metal center first coordinates with the olefinic double bond of the substrate. Crucially, the presence of the carboxylic acid moiety allows for an additional coordination interaction, effectively locking the substrate into a specific orientation relative to the chiral ligand environment. This bidentate-like coordination ensures that the subsequent oxidative addition of hydrogen and migratory insertion steps occur from a single facial direction, thereby establishing the new chiral center with high fidelity. The steric bulk provided by the substituents on the oxazoline ring (as seen in the catalyst structures where R = iPr or tBu) creates a chiral pocket that sterically hinders one face of the alkene, forcing the hydride transfer to occur exclusively from the less hindered face. This precise spatial control is what enables the observed high enantioselectivity, minimizing the formation of the unwanted enantiomer and reducing the burden on purification processes.
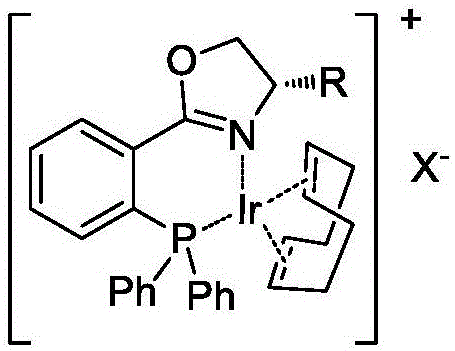
Furthermore, the choice of counterion and solvent plays a pivotal role in modulating the catalyst's activity and stability. The patent highlights the use of weakly coordinating anions like BArF (tetrakis[3,5-bis(trifluoromethyl)phenyl]borate), which help to maintain the cationic nature of the active iridium species, thereby enhancing its electrophilicity and ability to activate the hydrogen molecule. In terms of impurity control, the high selectivity of the catalyst means that side reactions such as over-reduction or isomerization are minimized, leading to a cleaner crude reaction profile. This is particularly important for regulatory compliance in the pharmaceutical sector, where impurity profiles must be strictly controlled. The robustness of the catalyst system against various functional groups (phenyl, biphenyl, benzophenone) suggests that it can be applied to a wide array of structurally diverse intermediates, providing a versatile platform for process development teams looking to optimize their synthetic strategies for maximum efficiency and yield.
How to Synthesize Chiral Alkenoic Acid Derivatives Efficiently
Implementing this asymmetric hydrogenation technology requires a systematic approach to ensure reproducibility and optimal performance. The process begins with the preparation of the alkenoic acid substrate, which can be synthesized via a Grignard reaction between appropriate precursors, followed by careful workup to ensure the absence of metal residues that might interfere with the sensitive iridium catalyst. Once the substrate is prepared, the hydrogenation is typically conducted in a sealed vessel under an inert atmosphere to prevent catalyst deactivation by oxygen. The reaction mixture, comprising the substrate, the iridium-phosphine oxazoline catalyst, a base such as triethylamine, and a solvent like methanol, is then pressurized with hydrogen. While the patent provides specific examples using a substrate-to-catalyst ratio of 400:1, process optimization may allow for even lower loadings to further enhance economic viability. Detailed standard operating procedures regarding temperature ramping, pressure maintenance, and quenching protocols are essential for transferring this laboratory-scale success to pilot and production scales.
- Prepare the alkenoic acid substrate (e.g., Compound A-1) via Grignard reaction between 4-methoxy-2-methylene-4-oxobutanoic acid and 2-([1,1'-biphenyl]-4-yl)acetic acid derivatives.
- Load the substrate into a hydrogenation vessel with an iridium-phosphine oxazoline catalyst (such as catalyst 7a or 7b) and an organic base like triethylamine in methanol.
- Pressurize the system with hydrogen gas (0.5-10 MPa) and stir at temperatures between 20°C to 50°C for approximately 18 hours to achieve high enantioselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this iridium-catalyzed asymmetric hydrogenation process offers substantial strategic advantages for organizations focused on cost reduction in fine chemical manufacturing. By replacing inefficient resolution steps or multi-step chiral pool syntheses with a direct catalytic hydrogenation, companies can significantly reduce the number of unit operations required, leading to lower capital expenditure and operational costs. The high atom economy of hydrogenation reactions, where hydrogen is the only reagent added across the double bond, aligns perfectly with green chemistry principles, potentially reducing waste disposal costs and improving the overall environmental footprint of the manufacturing process. Moreover, the ability to produce chiral intermediates with high optical purity directly from achiral starting materials mitigates the risk of supply chain disruptions associated with sourcing scarce natural chiral building blocks, thereby enhancing supply security.
- Cost Reduction in Manufacturing: The primary economic driver for this technology is the elimination of material loss inherent in classical resolution techniques. Since the catalyst directs the formation of the desired enantiomer with high selectivity (up to 92% ee as demonstrated), the theoretical yield is nearly doubled compared to a kinetic resolution which caps at 50%. Additionally, the catalyst loading can be optimized to very low levels, minimizing the cost contribution of the precious metal iridium. The use of common solvents like methanol and mild reaction conditions further reduces energy consumption and solvent recovery costs, contributing to a leaner and more cost-effective production model that improves margin potential for high-volume intermediates.
- Enhanced Supply Chain Reliability: Dependence on complex natural extracts or limited chiral pool materials often introduces volatility into the supply chain. This synthetic approach relies on readily available petrochemical-derived starting materials, such as biphenyl acetic acid derivatives and simple acrylates, which are produced on a massive global scale. This shift to synthetic, catalytic routes ensures a more stable and predictable supply of critical intermediates, reducing the risk of shortages that can delay drug development timelines. The robustness of the catalyst system also implies a longer shelf-life and easier handling, simplifying logistics and inventory management for procurement teams managing global supply networks.
- Scalability and Environmental Compliance: The reaction conditions specified in the patent, such as hydrogen pressures around 10 atm and temperatures near 50°C, are well within the operating parameters of standard industrial hydrogenation reactors. This compatibility facilitates a smoother transition from laboratory discovery to commercial production without the need for specialized high-pressure equipment that would require significant capital investment. Furthermore, the high selectivity reduces the generation of isomeric impurities, simplifying wastewater treatment and solvent recycling processes. This alignment with environmental regulations supports corporate sustainability goals and ensures long-term operational continuity in increasingly regulated markets.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patent is crucial for stakeholders evaluating its potential integration into their manufacturing portfolios. The following questions address common inquiries regarding catalyst performance, substrate scope, and operational parameters, providing clarity on how this technology can be leveraged to solve specific process challenges. These insights are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Which catalyst variants provide the highest enantioselectivity for biphenyl-substituted alkenoic acids?
A: According to the experimental data in Patent CN111410604A, catalyst variants 7a (R=iPr) and 7b (R=tBu) demonstrated superior performance, achieving enantiomeric excess (ee) values of 92% and 91% respectively for the hydrogenation of compound A-1.
Q: What are the typical reaction conditions for this asymmetric hydrogenation process?
A: The process operates under relatively mild conditions, utilizing hydrogen pressures ranging from 0.5 to 10 MPa (typically 10 atm in examples) and temperatures between room temperature (20-30°C) and 50°C, using solvents such as methanol or dichloromethane with an organic base additive.
Q: Can this methodology be applied to substrates other than biphenyl derivatives?
A: Yes, the patent claims broad substrate scope where R1 can be phenyl, substituted phenyl, naphthyl, benzophenone, or C1-C6 alkyl groups, indicating the versatility of the iridium-phosphine oxazoline catalyst system for various pharmaceutical intermediate structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Alkenoic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the iridium-phosphine oxazoline system described in Patent CN111410604A. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate such innovative laboratory processes into robust, commercial-scale manufacturing operations. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot plant to full-scale manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent quality standards, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Whether you require custom synthesis of complex chiral acids or optimization of existing hydrogenation processes, our facility is equipped to handle the complexities of modern pharmaceutical manufacturing with precision and reliability.
We invite you to collaborate with us to explore how this asymmetric hydrogenation technology can enhance your supply chain and reduce your overall production costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating the tangible economic benefits of switching to this catalytic route. Please contact our technical procurement team today to request specific COA data for similar chiral intermediates and to discuss route feasibility assessments for your target molecules. Let us be your partner in driving innovation and efficiency in your pharmaceutical supply chain.
