Advanced Solid-Phase Synthesis of Atosiban Acetate for Commercial Scalability and Purity
Introduction to Advanced Atosiban Acetate Manufacturing
The pharmaceutical landscape for oxytocin antagonists has evolved significantly with the introduction of robust solid-phase peptide synthesis (SPPS) methodologies. Patent CN101357937A discloses a highly efficient preparation method for Atosiban Acetate, a critical active pharmaceutical ingredient used to delay imminent preterm birth. This technology represents a paradigm shift from traditional solution-phase or Boc-chemistry routes, leveraging Fmoc-protected amino acids and Rink Amide resins to streamline the production workflow. By anchoring the peptide chain to a solid support, the process facilitates easy washing and removal of excess reagents, thereby enhancing the overall purity profile of the intermediate. The strategic implementation of this protocol allows manufacturers to achieve a total recovery rate exceeding 18.5%, demonstrating its viability for high-demand clinical supply chains.
Furthermore, the structural integrity of the final nonapeptide is meticulously preserved through controlled coupling conditions and a specialized oxidative cyclization step. The process begins with the selection of high-quality Rink Amide AM or MBHA resins, which serve as the foundational scaffold for the sequential addition of amino acid monomers. This approach not only simplifies the operational complexity but also aligns with modern green chemistry principles by minimizing the generation of hazardous byproducts. For global procurement teams, understanding the nuances of this patented pathway is essential for securing a reliable Atosiban Acetate supplier capable of meeting stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptides like Atosiban relied heavily on Boc (tert-butyloxycarbonyl) chemistry, as exemplified by earlier patents such as USP 4,504,469. These legacy methods typically utilized chloromethyl resins and required harsh cleavage conditions involving anhydrous liquid ammonia or hydrogen fluoride (HF). The reliance on HF presents severe safety hazards, necessitating specialized equipment and rigorous containment protocols that drastically inflate capital expenditure and operational costs. Moreover, the production cycle for Boc-based synthesis is inherently longer due to the multiple neutralization and washing steps required to remove acidic byproducts. The environmental footprint of these conventional routes is also substantial, generating significant quantities of toxic three-waste streams that require expensive treatment before disposal, making them less attractive for modern sustainable manufacturing initiatives.
The Novel Approach
In contrast, the novel approach detailed in CN101357937A adopts Fmoc (9-fluorenylmethoxycarbonyl) chemistry, which operates under much milder basic conditions for deprotection. This shift eliminates the need for dangerous hydrofluoric acid, replacing it with a trifluoroacetic acid (TFA) based cleavage cocktail that is easier to handle and neutralize. The use of Rink Amide resins allows for the direct formation of the C-terminal amide, which is crucial for the biological activity of Atosiban, without requiring additional amidation steps post-cleavage. By integrating mercaptopropionic acid (Map) as the N-terminal capping group directly on the resin, the process ensures precise stoichiometry and reduces the risk of truncation sequences. This streamlined workflow not only shortens the production lead time but also significantly lowers the barrier to entry for scaling up production to meet global pharmaceutical demands.
Mechanistic Insights into Fmoc-Based Solid Phase Peptide Synthesis
The core of this manufacturing breakthrough lies in the iterative coupling mechanism facilitated by uranium-based condensing agents such as TBTU (O-(Benzotriazol-1-yl)-N,N,N',N'-tetramethyluronium tetrafluoroborate) in the presence of HOBt (1-Hydroxybenzotriazole). During each cycle, the Fmoc protecting group is removed using a piperidine solution, exposing the free amine on the growing peptide chain. Subsequently, the incoming Fmoc-amino acid is activated by TBTU to form a highly reactive ester intermediate, which rapidly reacts with the resin-bound amine to form a stable peptide bond. This activation strategy minimizes racemization, a critical quality attribute for chiral pharmaceutical intermediates, ensuring that the stereochemical integrity of residues like D-Tyr and L-Cys is maintained throughout the synthesis. The efficiency of this coupling is further enhanced by the use of N-methylmorpholine (NMM) as a base, which scavenges protons and drives the reaction equilibrium towards product formation.
Following the assembly of the linear nonapeptide sequence, the process transitions to a critical cleavage and cyclization phase. The protected peptide resin is treated with a cocktail comprising TFA, HBr, acetic acid, triisopropylsilane (TIS), and ethanedithiol (EDT). This mixture serves a dual purpose: it cleaves the peptide from the acid-labile Rink Amide linker and simultaneously removes side-chain protecting groups such as Trt (trityl) and Boc (tert-butoxycarbonyl). The inclusion of scavengers like TIS and EDT is vital for preventing the alkylation of sensitive residues like Cysteine and Tyrosine by carbocation byproducts generated during acidolysis. Once the linear reduced peptide is precipitated and isolated, it undergoes an air-oxidation step where the pH is carefully adjusted to between 7.5 and 10.0. Under these alkaline conditions, the thiol groups of the Cysteine residue and the terminal Mercaptopropionic acid undergo spontaneous oxidation to form the essential intramolecular disulfide bridge, locking the peptide into its bioactive conformation.

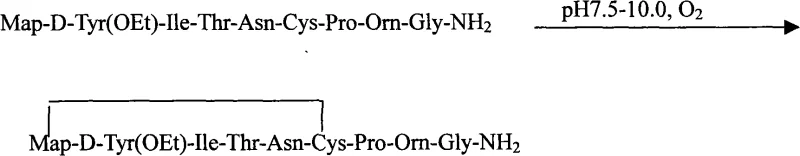
How to Synthesize Atosiban Acetate Efficiently
The synthesis of Atosiban Acetate via this solid-phase route requires precise control over reaction parameters to maximize yield and minimize impurities. The process initiates with the swelling of Rink Amide resin in DMF, followed by the sequential coupling of amino acids starting from Glycine at the C-terminus and extending to the N-terminal Mercaptopropionic acid. Each coupling step is monitored to ensure completeness, typically utilizing a molar excess of activated amino acids relative to the resin loading capacity. The detailed standard operating procedures for resin pretreatment, coupling cycles, and final cleavage are critical for reproducibility. For a comprehensive breakdown of the specific reagent ratios, reaction times, and temperature controls required for each step of this synthesis, please refer to the standardized guide below.
- Swelling and Deprotection: Treat Rink Amide resin with DMF and piperidine to prepare the active amine sites for coupling.
- Sequential Coupling: Iteratively couple Fmoc-protected amino acids (Gly, Orn, Pro, Cys, Asn, Thr, Ile, D-Tyr) and Mercaptopropionic acid using TBTU/HOBt activation.
- Cleavage and Cyclization: Cleave the peptide from the resin using a TFA-based cocktail, precipitate the crude linear peptide, and perform air oxidation at pH 7.5-10.0 to form the disulfide bridge.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the Fmoc-SPPS route described in CN101357937A offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials; Fmoc-protected amino acids are commodity chemicals with robust global availability, unlike some specialized reagents required for older Boc protocols. This abundance ensures supply continuity and mitigates the risk of production stoppages due to raw material shortages. Furthermore, the elimination of hazardous reagents like hydrogen fluoride reduces the regulatory burden and insurance costs associated with manufacturing, translating into a more stable and predictable cost structure for the final API. The ability to produce high-purity intermediates with fewer purification steps also means a faster time-to-market for generic versions of Atosiban, providing a competitive edge in the pharmaceutical marketplace.
- Cost Reduction in Manufacturing: The transition to Fmoc chemistry eliminates the need for expensive and dangerous infrastructure required for handling anhydrous hydrogen fluoride. By utilizing standard glass-lined or stainless steel reactors compatible with TFA cleavage, manufacturers can significantly lower capital expenditure. Additionally, the high coupling efficiency of the TBTU/HOBt system reduces the consumption of expensive amino acid monomers, as fewer recoupling steps are needed to drive reactions to completion. The simplified downstream processing, which avoids complex extraction procedures associated with liquid ammonia cleavage, further contributes to substantial cost savings in labor and utility consumption.
- Enhanced Supply Chain Reliability: The reliance on commercially available Rink Amide resins and standard Fmoc-amino acids creates a resilient supply chain that is less susceptible to geopolitical or logistical disruptions. Since these materials are produced by multiple vendors globally, procurement teams have the flexibility to qualify alternative sources, ensuring uninterrupted production schedules. The robustness of the solid-phase method also allows for better inventory management, as the resin-bound intermediates are stable and can be stored if necessary, providing a buffer against demand fluctuations. This reliability is crucial for maintaining the continuous supply of life-saving tocolytic agents to hospitals and clinics worldwide.
- Scalability and Environmental Compliance: From an environmental perspective, this method generates significantly less hazardous waste compared to traditional solution-phase synthesis. The solid-phase nature of the reaction allows for the efficient recycling of solvents like DMF and DCM, and the absence of heavy metal catalysts simplifies wastewater treatment. The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-kilogram commercial production without significant re-engineering of the process parameters. This scalability ensures that manufacturers can rapidly ramp up production volumes to meet surging market demand while remaining compliant with increasingly stringent environmental regulations regarding solvent emissions and chemical disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the solid-phase synthesis of Atosiban Acetate. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on yield expectations, safety protocols, and purification strategies. Understanding these details is vital for technical teams evaluating the feasibility of adopting this manufacturing route for their own production facilities or for sourcing partners who utilize this specific technology.
Q: What are the advantages of the Fmoc route over the traditional Boc route for Atosiban?
A: The Fmoc route described in CN101357937A eliminates the need for hazardous hydrogen fluoride (HF) and liquid ammonia used in Boc chemistry. It utilizes milder TFA cleavage conditions, significantly improving operator safety and reducing three-waste pollution while maintaining high yields.
Q: How is the critical disulfide bond formed in this synthesis?
A: After cleavage from the resin, the reduced linear peptide is dissolved in water. The pH is adjusted to 7.5-10.0 using ammonium hydroxide, and air is blown through the solution to facilitate spontaneous oxidation, forming the intramolecular disulfide bridge between the cysteine and mercaptopropionic acid residues.
Q: Is this solid-phase method suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method is convenient for industrialization. It uses commercially available Rink Amide resins and standard Fmoc amino acids, achieving a total recovery of over 18.5% with simplified purification steps compared to older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Atosiban Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality peptide intermediates in the development of effective tocolytic therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric requirements of global pharmaceutical partners. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify the identity and potency of every batch. Our commitment to excellence ensures that the Atosiban Acetate we supply meets the highest international standards for safety and efficacy, supporting your drug development and commercialization goals with unwavering reliability.
We invite you to collaborate with us to optimize your supply chain for this vital medication. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can enhance your project's success and deliver value to your organization through superior chemical solutions.
