Advanced Manufacturing of Pyrimidinyl-4-Aminopyrazole Compounds for LRRK2 Inhibitors
Advanced Manufacturing of Pyrimidinyl-4-Aminopyrazole Compounds for LRRK2 Inhibitors
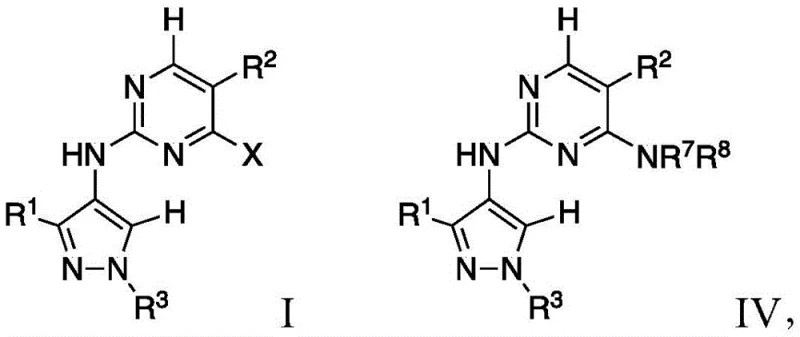
The pharmaceutical industry is constantly seeking robust and scalable synthetic routes for complex heterocyclic compounds, particularly those targeting neurodegenerative diseases. Patent CN112088003A, filed in December 2020, introduces a significant advancement in the preparation of pyrimidinyl-4-aminopyrazole compounds, specifically designed as inhibitors for Leucine-rich repeat kinase 2 (LRRK2). This kinase has been identified as a key therapeutic target for Parkinson's disease, driving substantial demand for high-quality intermediates. The disclosed technology offers a streamlined pathway to generate compounds of Formula I and IV, addressing critical challenges in regioselectivity and yield that have plagued previous methodologies. By leveraging specific Lewis acid catalysts and optimized reaction conditions, this process ensures the preferential formation of the desired N-1 substituted products over unwanted regioisomers. For procurement leaders and R&D directors, understanding the nuances of this patented chemistry is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality for clinical and commercial programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing pyrimidinyl-aminopyrazole scaffolds often suffer from poor regiocontrol during the nucleophilic aromatic substitution (SNAr) steps. In conventional processes, the coupling of aminopyrazoles with dihalo-pyrimidines frequently results in a mixture of N-1 and N-2 substituted regioisomers, necessitating complex and costly purification steps to isolate the active pharmaceutical ingredient. Furthermore, older methods may rely on harsh reaction conditions or expensive transition metal catalysts that introduce heavy metal impurities, complicating the regulatory approval process for final drug substances. The lack of selectivity not only reduces the overall yield but also increases the environmental footprint due to higher solvent consumption and waste generation. For supply chain managers, these inefficiencies translate into unpredictable lead times and volatile pricing, making it difficult to maintain a stable inventory of high-purity OLED material or pharmaceutical precursors. Consequently, there is a pressing need for a methodology that inherently suppresses side reactions and simplifies downstream processing.
The Novel Approach
The process outlined in CN112088003A overcomes these historical barriers through a meticulously designed sequence that prioritizes regioselectivity and operational simplicity. By employing specific Lewis acids such as zinc chloride (ZnCl2) during the coupling phase, the reaction kinetics are tuned to favor the displacement of the leaving group at the 2-position of the pyrimidine ring by the 4-amino group of the pyrazole. This strategic intervention effectively minimizes the formation of undesired isomers, thereby enhancing the crude purity of the intermediate. Additionally, the patent describes versatile pathways for functionalizing the pyrazole nitrogen, including alkylation and acylation strategies that are compatible with a wide range of substituents. This flexibility allows manufacturers to access diverse analog libraries without redesigning the entire synthetic route. From a commercial perspective, this novel approach facilitates cost reduction in API manufacturing by reducing the number of purification cycles and improving the overall mass balance of the process, ensuring a more sustainable and economically viable production model.
Mechanistic Insights into Regioselective SNAr Coupling
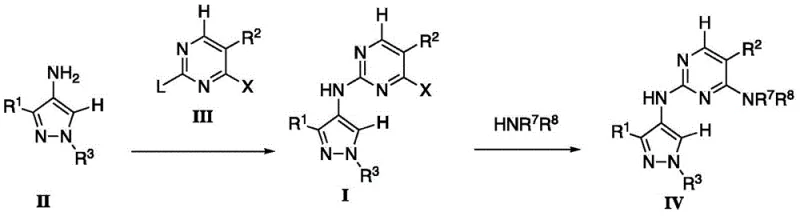
The core innovation of this technology lies in the mechanistic control exerted during the coupling of the aminopyrazole intermediate (Formula II) with the pyrimidine electrophile (Formula III). The reaction proceeds via a nucleophilic aromatic substitution mechanism where the nucleophilicity of the exocyclic amine is carefully modulated. In the presence of a Lewis acid, the electron density of the pyrimidine ring is altered, making the carbon bearing the leaving group 'L' more susceptible to nucleophilic attack compared to the carbon bearing substituent 'X'. This differential reactivity is crucial for achieving the specific connectivity required for LRRK2 inhibition. The patent explicitly notes that the coupling preferentially results in the substitution of the 'L' group, preserving the 'X' group for subsequent functionalization if necessary. This level of control is paramount for R&D teams aiming to optimize structure-activity relationships (SAR) without being hindered by inseparable impurities. Understanding this mechanism allows chemists to fine-tune reaction parameters such as temperature and stoichiometry to maximize efficiency.
Furthermore, the process incorporates robust impurity control mechanisms inherent to the choice of reagents and solvents. For instance, the use of polar aprotic solvents and specific bases helps to solubilize the intermediates while maintaining the stability of the sensitive functional groups. The reduction of nitro groups to amines, a preceding step in the synthesis, is conducted using catalytic hydrogenation with palladium on carbon, a method known for its cleanliness and scalability. This step avoids the generation of stoichiometric metal waste associated with chemical reductions using iron or tin. By integrating these clean technologies, the overall impurity profile of the final product is significantly improved, meeting the stringent purity specifications required for clinical trials. The ability to predict and manage impurity formation is a key differentiator for any contract development and manufacturing organization (CDMO) aiming to support late-stage drug development programs.
How to Synthesize Pyrimidinyl-4-Aminopyrazole Efficiently
The synthesis of these complex heterocycles involves a logical progression of transformations that build molecular complexity while maintaining high fidelity. The process begins with the preparation of the pyrazole core, followed by functionalization and final coupling. Detailed operational procedures provided in the patent examples demonstrate the feasibility of executing these reactions on a multi-kilogram scale, providing a clear roadmap for technology transfer. The following guide outlines the critical stages involved in transforming simple starting materials into the advanced intermediates described in the disclosure. For technical teams evaluating this route, it is important to note that the standardized synthetic steps见下方的指南 ensure reproducibility and safety across different manufacturing sites.
- Perform N-1 alkylation of 3-substituted-4-nitro-1H-pyrazole using an alkylating agent and base to form the N-1 substituted intermediate.
- Reduce the nitro group of the intermediate via catalytic hydrogenation using Pd/C to generate the 4-aminopyrazole derivative.
- Couple the aminopyrazole with a 2,4-disubstituted pyrimidine using a Lewis acid catalyst to achieve regioselective formation of the final compound.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthetic route offers tangible benefits for procurement and supply chain stakeholders who are tasked with managing costs and ensuring continuity of supply. The elimination of complex chromatographic purifications in favor of crystallization-based workups significantly reduces processing time and solvent usage. This simplification directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering utility costs and waste disposal fees. Moreover, the reagents utilized, such as zinc chloride and common organic solvents like dichloromethane and ethyl acetate, are readily available from global chemical suppliers, mitigating the risk of raw material shortages. This accessibility enhances supply chain reliability, allowing manufacturers to plan production schedules with greater confidence and reduce lead time for high-purity pharmaceutical intermediates. The robustness of the chemistry also means that batch-to-batch variability is minimized, ensuring consistent quality for downstream customers.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiencies by maximizing atom economy and minimizing the need for expensive chromatographic separations. By relying on crystallization for purification, the method reduces the consumption of high-grade solvents and silica gel, which are major cost drivers in fine chemical synthesis. Additionally, the high regioselectivity reduces the loss of valuable starting materials to side products, effectively increasing the overall yield of the desired isomer. These factors combine to lower the cost of goods sold (COGS), making the final API more competitive in the marketplace. The avoidance of precious metal catalysts in the coupling step further contributes to substantial cost savings, as there is no need for specialized metal scavenging resins or extensive testing for residual metals.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals and standard unit operations ensures that the supply chain remains resilient against market fluctuations. Unlike processes that depend on bespoke reagents or proprietary catalysts, this route utilizes materials that can be sourced from multiple vendors, preventing single points of failure. The scalability demonstrated in the patent examples, ranging from gram to kilogram quantities, proves that the chemistry can be seamlessly transferred from pilot plants to commercial-scale reactors. This scalability is critical for meeting the surging demand for Parkinson's disease therapeutics without compromising on delivery timelines. Furthermore, the stability of the intermediates allows for safer storage and transportation, reducing logistical complexities.
- Scalability and Environmental Compliance: The synthetic pathway is designed with green chemistry principles in mind, facilitating easier compliance with increasingly strict environmental regulations. The use of catalytic hydrogenation instead of stoichiometric metal reductions minimizes the generation of hazardous solid waste. Solvent recovery systems can be easily integrated into the process to recycle dichloromethane and ethyl acetate, further reducing the environmental footprint. The high purity of the crude product reduces the burden on wastewater treatment facilities, as fewer organic impurities need to be degraded. This alignment with sustainability goals not only protects the environment but also enhances the corporate social responsibility profile of the manufacturing entity, appealing to eco-conscious investors and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these pyrimidinyl-4-aminopyrazole compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is vital for partners looking to integrate this technology into their existing pipelines. The responses cover aspects ranging from reaction mechanisms to scalability potential, offering a comprehensive overview for decision-makers.
Q: What is the primary therapeutic application of these pyrimidinyl-4-aminopyrazole compounds?
A: These compounds serve as potent inhibitors of Leucine-rich repeat kinase 2 (LRRK2), making them critical intermediates for developing treatments for Parkinson's disease and other LRRK2-mediated disorders.
Q: How does this patent process improve upon conventional synthesis methods?
A: The disclosed process utilizes specific Lewis acid catalysts and controlled reaction conditions to ensure high regioselectivity during the coupling step, significantly reducing the formation of unwanted N-2 substituted regioisomers common in traditional methods.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the patent provides detailed examples demonstrating the synthesis on a kilogram scale (e.g., 15 kg batches), utilizing robust reagents and standard purification techniques like crystallization, which indicates strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidinyl-4-Aminopyrazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for next-generation therapeutics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to marketplace. We are committed to delivering high-purity pharmaceutical intermediates that meet rigorous quality standards, supported by our state-of-the-art analytical capabilities and stringent purity specifications. Our rigorous QC labs employ advanced techniques to verify the identity and purity of every batch, guaranteeing that the materials you receive are fit for purpose. Whether you require custom synthesis or technology transfer services, our infrastructure is designed to support your growth and innovation.
We invite you to collaborate with us to leverage the advantages of this patented process for your LRRK2 inhibitor programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. By partnering with us, you gain access to deep process knowledge and a commitment to excellence that drives value throughout your supply chain. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the development of life-saving medicines.
