Advanced Water-Soluble Copolymer Immobilized L-Proline Catalyst for Scalable Asymmetric Synthesis
Advanced Water-Soluble Copolymer Immobilized L-Proline Catalyst for Scalable Asymmetric Synthesis
The landscape of green chemistry and sustainable pharmaceutical manufacturing is undergoing a significant transformation, driven by the urgent need to replace toxic organic solvents with benign alternatives like water. A pivotal advancement in this domain is detailed in Chinese patent CN110698618A, which discloses a novel water-soluble copolymer immobilized L-proline catalyst. This innovation addresses the longstanding challenge of performing efficient asymmetric organocatalysis in aqueous media, a feat traditionally hindered by the poor solubility of organic substrates and the instability of catalysts in water. By leveraging reversible addition-fragmentation transfer (RAFT) living radical polymerization, the inventors have constructed an amphiphilic block copolymer that not only solubilizes the chiral L-proline catalyst but also creates a unique microenvironment that enhances reactivity and selectivity. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a paradigm shift towards more cost-effective and environmentally compliant synthetic routes.
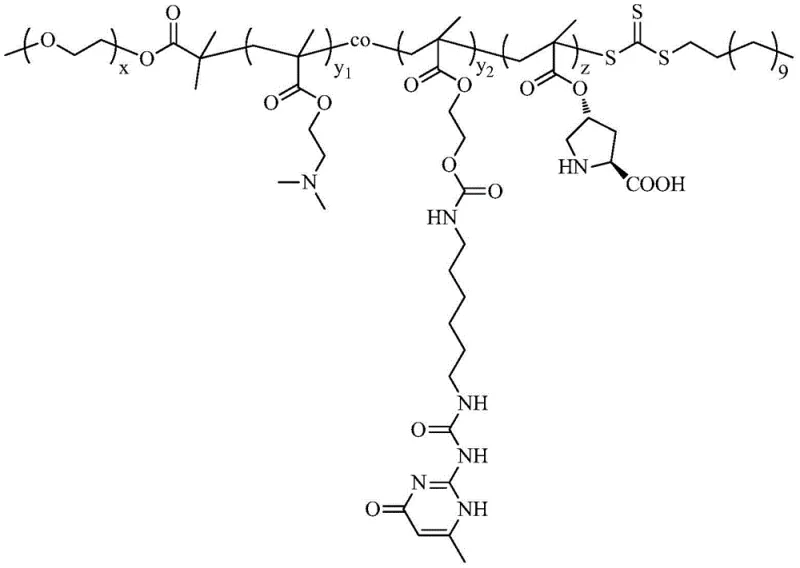
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, L-proline and its derivatives have been the gold standard for catalyzing asymmetric aldol condensations, a fundamental reaction for constructing carbon-carbon bonds in complex molecule synthesis. However, the conventional application of these organocatalysts relies heavily on polar aprotic organic solvents such as dimethylformamide (DMF), dimethyl sulfoxide (DMSO), or chloroform. These solvents pose severe environmental and safety hazards, requiring expensive disposal protocols and rigorous containment systems to protect worker health. Furthermore, homogeneous L-proline catalysts are notoriously difficult to separate from the reaction mixture, often necessitating complex chromatographic purification steps that drastically reduce overall yield and increase production costs. In an industrial setting, the inability to recycle the catalyst leads to substantial material waste, making large-scale processes economically unviable and environmentally unsustainable. Additionally, when attempts are made to run these reactions in water without modification, the hydrophobic nature of typical organic substrates prevents effective interaction with the catalyst, resulting in sluggish reaction rates and poor stereocontrol.
The Novel Approach
The technology described in patent CN110698618A offers a sophisticated solution by engineering a "foldable" polymer architecture that functions as a artificial enzyme. Instead of using free L-proline, the catalyst immobilizes the chiral unit onto a dual-hydrophilic and hydrophobic block copolymer backbone. The key innovation lies in the incorporation of ureidopyrimidinone (UPy) units, which possess the ability to form strong quadruple hydrogen bonds. In an aqueous environment, these UPy groups drive the self-assembly of the polymer chains into micelles with a hydrophobic core and a hydrophilic shell. This core-shell structure effectively creates a "nanoreactor" where hydrophobic reactants are concentrated within the cavity, in close proximity to the immobilized L-proline active sites. This biomimetic approach not only solubilizes the reactants in water but also provides a confined space that enhances the stereochemical outcome of the reaction. For companies focused on cost reduction in API manufacturing, this method eliminates the need for volatile organic compounds (VOCs) and simplifies downstream processing, offering a clear pathway to greener and cheaper production.
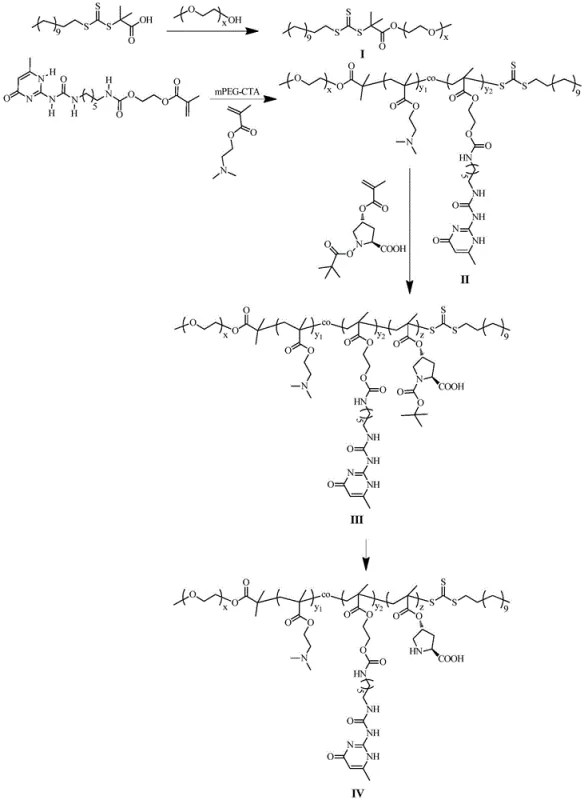
Mechanistic Insights into RAFT Polymerization and Micellar Catalysis
The synthesis of this advanced catalyst relies on the precision of RAFT polymerization, a technique that allows for strict control over molecular weight and polydispersity. The process begins with the esterification of a trithiocarbonate chain transfer agent (CTA) with methoxy polyethylene glycol (mPEG) to create a macro-CTA. This hydrophilic initiator is then used to polymerize a mixture of 2-(dimethylamino)ethyl methacrylate (DMAEMA) and a UPy-functionalized methacrylate monomer. The resulting amphiphilic block serves as the scaffold. Subsequently, a third block containing protected L-proline (Boc-ProlA) is added via chain extension. The final step involves the selective removal of the tert-butoxycarbonyl (Boc) protecting group using trifluoroacetic acid, revealing the free secondary amine of the proline residue which is essential for catalytic activity. This modular synthesis ensures that the catalytic sites are uniformly distributed along the polymer chain, preventing aggregation and ensuring consistent performance across batches.
The catalytic mechanism is deeply rooted in the thermoresponsive nature of the copolymer. The DMAEMA segments impart a lower critical solution temperature (LCST) behavior, meaning the polymer's solubility changes dramatically with temperature. Below the LCST (around 40°C), the polymer chains are extended and hydrophilic. As the temperature rises above this threshold during the reaction, the chains collapse and aggregate, strengthening the hydrophobic interactions within the micelle core. This dynamic folding-unfolding behavior is crucial; it allows the micelles to adapt their structure, facilitating the entry of substrates and the release of products while maintaining the integrity of the hydrophobic pocket. The UPy hydrogen bonds act as physical cross-links, stabilizing the micelle structure against dilution and shear forces, which explains the catalyst's exceptional stability and reusability. This level of structural control is what differentiates this system from simple surfactant-based emulsions, providing a robust platform for high-precision synthesis.
How to Synthesize Water-Soluble Copolymer Immobilized L-Proline Catalyst Efficiently
The preparation of this high-performance organocatalyst follows a streamlined, multi-step protocol that is amenable to scale-up. The process integrates standard organic synthesis techniques with controlled polymerization methods, ensuring reproducibility and high purity. The initial formation of the macro-chain transfer agent sets the foundation for the block architecture, followed by sequential monomer additions to build the functional domains. The final deprotection step is critical for activating the catalytic centers without degrading the polymer backbone. For technical teams looking to implement this technology, the detailed standardized synthesis steps are outlined in the guide below, which breaks down the specific molar ratios, solvent choices, and reaction conditions required to achieve the optimal molecular weight distribution and catalytic activity described in the patent literature.
- Synthesize the macromolecular chain transfer agent (mPEG-CTA) by reacting S-1-dodecyl-S′-(α,α′-dimethyl-α″-acetic acid) trithiocarbonate with polyethylene glycol monomethyl ether using EDC and DMAP in dichloromethane.
- Perform reversible addition-fragmentation transfer (RAFT) polymerization of DMAEMA and UPy-HEMA monomers initiated by mPEG-CTA in DMSO with AIBN to form the amphiphilic block copolymer backbone.
- Extend the polymer chain via a second RAFT reaction using N-tert-butoxycarbonyl methacryloyl-4-hydroxy-L-proline (Boc-ProlA) as the monomer to introduce the catalytic proline units.
- Remove the Boc protecting group from the L-proline units by treating the copolymer with trifluoroacetic acid (TFA) in dichloromethane to activate the final water-soluble catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this water-soluble copolymer catalyst offers tangible strategic benefits beyond mere technical performance. The primary advantage lies in the drastic simplification of the manufacturing workflow. By enabling reactions to proceed in pure water, facilities can eliminate the procurement, storage, and disposal costs associated with large volumes of hazardous organic solvents. This shift not only reduces the regulatory burden related to VOC emissions but also lowers insurance and safety compliance costs. Furthermore, the heterogeneous nature of the catalyst system—where the catalyst forms distinct micellar particles—allows for easier separation from the product compared to homogeneous systems. This translates to reduced energy consumption during purification and higher overall throughput, directly impacting the bottom line through operational efficiency.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive solvent recovery systems and the reduction of waste treatment fees. Since the catalyst operates in water, the need for complex distillation columns to recover solvents like DMF or DMSO is removed. Additionally, the high recyclability of the catalyst means that the effective cost per kilogram of product is significantly lowered over time, as the same batch of catalyst can be utilized for multiple production runs without significant loss of activity. This durability reduces the frequency of raw material ordering and minimizes inventory holding costs for expensive chiral catalysts.
- Enhanced Supply Chain Reliability: Relying on water as the primary reaction medium mitigates risks associated with the supply volatility of petrochemical-derived solvents. Water is universally available and inexpensive, insulating the production process from market fluctuations in solvent pricing. Moreover, the robustness of the polymer-supported catalyst ensures consistent quality output, reducing the risk of batch failures that can disrupt delivery schedules. The ability to maintain high enantiomeric excess over multiple cycles ensures that the supply of chiral intermediates remains stable and predictable, which is critical for meeting the stringent quality agreements of downstream pharmaceutical clients.
- Scalability and Environmental Compliance: From a scalability perspective, the aqueous nature of the reaction simplifies heat management, as water has a high heat capacity, reducing the risk of thermal runaways in large reactors. This safety profile facilitates easier scale-up from pilot plant to commercial production volumes. Environmentally, the process aligns perfectly with green chemistry principles, generating an aqueous waste stream that is far easier to treat than organic solvent waste. This compliance with increasingly strict environmental regulations future-proofs the manufacturing process, avoiding potential fines and ensuring long-term operational continuity in regions with tight emission controls.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copolymer catalyst system. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these factors is essential for evaluating the feasibility of integrating this catalytic system into existing production lines for chiral intermediate synthesis.
Q: How does the copolymer catalyst achieve high stereoselectivity in an aqueous environment?
A: The catalyst utilizes ureidopyrimidinone (UPy) groups that form quadruple hydrogen bonds, causing the polymer to self-assemble into a core-shell micelle structure in water. This creates a hydrophobic cavity that acts as a nanoreactor, concentrating hydrophobic reactants near the chiral L-proline active sites, thereby mimicking enzymatic conditions to achieve high enantiomeric excess (up to 90% ee).
Q: What are the recyclability characteristics of this immobilized L-proline catalyst?
A: Unlike traditional homogeneous L-proline catalysts which are difficult to recover, this amphiphilic copolymer catalyst demonstrates excellent recycling performance. Experimental data indicates that the catalyst maintains high conversion rates and stereoselectivity over at least nine consecutive reaction cycles, significantly reducing material consumption and waste generation.
Q: What are the optimal reaction conditions for asymmetric aldol condensation using this catalyst?
A: The catalyst exhibits thermoresponsive behavior with a lower critical solution temperature (LCST) around 40°C. Optimal catalytic performance, balancing conversion and selectivity, is achieved at 45°C with a catalyst loading of 5 mol% relative to the aldehyde substrate in pure water, eliminating the need for toxic organic solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Proline Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organocatalysis in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts like the water-soluble copolymer L-proline catalyst can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise measurement of enantiomeric excess and residual metal content, guaranteeing that every batch meets the highest global standards. We understand that transitioning to new catalytic technologies requires confidence in both the chemistry and the supply partner.
We invite you to collaborate with our technical team to explore how this green catalytic solution can optimize your specific synthetic routes. By leveraging our expertise in polymer chemistry and asymmetric synthesis, we can provide a Customized Cost-Saving Analysis tailored to your project's unique constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced catalytic platforms can drive efficiency and sustainability in your supply chain.
